Abstract
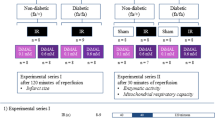
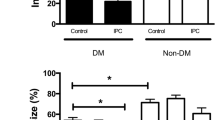
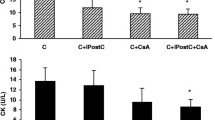
Sarcolemmal ATP-sensitive potassium (KATP) channels have been mentioned to participate in preconditioning protection. Since these channels are altered in diabetes, it would be possible that preconditioning does not develop in diabetic (D) hearts. The purpose of this study was to assess whether early (EP) and late (LP) ischemic preconditioning protect diabetic hearts against stunning in a conscious diabetic sheep model and whether diabetes might have altered KATP channel functioning. Sheep received alloxan monohydrate (1 g) and were ascribed to three experimental groups: control (DC, 12 min of ischemia (I) followed by 2 h of reperfusion (R)), early preconditioning (DEP, six 5 min I – 5 min R periods were performed before the 12 min I) and late preconditioning (DLP, same as DEP except that the preconditioning stimulus was performed 24 h before the 12 min I). Regional mechanics during reperfusion was evaluated as the percent recovery of wall thickening fraction (%WTH) expressed as percentage of basal values (100%) and KATP behaviour was indirectly assessed by monophasic action potential duration (MAPD) and sensitivity to glibenclamide blockade (0.1 and 0.4 mg/Kg). The results were compared to those obtained in normal (N) sheep. EP and LP protected against stunning in normal sheep (%WTH: NC = 63 ± 3.7, NLP = 80 ± 5**, NEP equals; 78 ± 3*, *p < 0.05 and **p < 0.01 against NC) whereas contrary results occurred in diabetic ones, where DLP (%WTH = 60 ± 4) afforded a similar recovery to DC (%WTH = 54 ± 5) and DEP surprisingly worsened instead of improving mechanical function (%WTH = 38 ± 6, p < 0.01 against DC). KATP channel behaviour appeared altered in diabetic hearts as shown by MAPD during ischemia in normal sheep (153 ± 9 msec) compared to diabetic ones (128 ± 11 msec, p < 0.05) and by the sensitivity to glibenclamide (while 0.4 mg/Kg blocked action potential shortening in normal and diabetic animals, 0.1 mg/Kg completely blocked KATP in diabetic but not in normal hearts, p ( 0.05). A sarcolemmal KATP channel dysfunction might afford a primary approach to explain the absence of ischemic preconditioning protection against stunning in diabetic sheep.
Similar content being viewed by others
References
Murry CE, Jennings RB, Reimer KA: Preconditioning with ischemia: A delay of lethal cell injury in ischemic myocardium. Circulation 74: 1124-1136, 1986
Schott RJ, Rohmann S, Braun ER, Schaper W: Ischemic preconditioning reduces infarct size in swine myocardium. Circ Res 66: 1133-1142, 1990
Cohen MV, Liu GS, Downey JM: Preconditioning causes improved wall motion as well as smaller infarcts after transient coronary occlusion in rabbits. Circulation 84: 341-349, 1991
Shiki K, Hearse DJ: Preconditioning of ischemic myocardium: Reperfusion-induced arrhythmias. Am J Physiol 253: H1470-H1476, 1987
Parrat J, Vegh A, Papp JG: Pronounced antiarrhythmic effects of ischaemic preconditioning — are there possibilities for pharmacological exploitation? Pharmacol Res 31: 225-234, 1995
Sun JZ, Tang XK, Knowlton AA, Park SW, Qiu Y, Bolli R: Late preconditioning against myocardial stunning. An endogenous protective mechanism that confers resistance to postichemic dysfunction 24 h after brief ischemia in conscious pigs. J Clin Invest 95: 388-403, 1995
Takano H, Tang XL, Bolli R: Differential role of KATP channels in late preconditioning protection against myocardial stunning and infarction in rabbits. Am J Physiol 279: H2350-H2359, 2000
Lascano Elena C, Negroni Jorge A, del Valle Héctor F, Crottogini Alberto J: Left ventricular regional systolic and diastolic function in conscious sheep undergoing ischemic preconditioning. Cardiovasc Res 41: 77-86, 1999
Yellon DM, Baxter GF, Garcia-Dorado D, Heusch G, Sumeray MS: Ischaemic preconditioning: Present position and future directions. Cardiovasc Res 37: 21-33, 1998
Liu Y, Thornton JD, Cohen MV, Downey JM, Schaffer SW: Streptozotocin-induced non-insulin-dependent diabetes protects the heart from infarction. Circulation 88: 1273-1278, 1993
Tosaki A, Engelman DT, Engelman RM, Das DK: The evolution of diabetic response to ischemia/reperfusion and preconditioning in isolated working rat hearts. Cardiovasc Res 31: 526-536, 1996
Bouchard JF, Lamontagne D: Protection afforded by preconditioning to the diabetic heart against ischaemic injury. Cardiovasc Res 37: 82-90, 1998
Kersten JR, Toller WG, Gross ER, Pagel PS, Warltier DC: Diabetes abolishes ischemic preconditioning: Role of glucose, insulin and osmolality. Am J Physiol 278: H1218-H1224, 2000
Pantos CI, Davos CH, Carageorgiou HC, Varonos DV, Cokkinos DV: Ischaemic preconditioning protects against myocardial dysfunction caused by ischaemia in isolated hypertrophied rat hearts. Basic Res Cardiol 91: 444-449, 1996
Moolman JA, Genade S, Tromp E, Opie LH, Lochner A: Ischaemic preconditioning does not protect hypertrophied myocardium against ischaemia. S Afr Med J 87: C151-C156, 1997
Fein SF, Sonnenblick EH: Diabetic cardiomyopathy. Cardiovasc Drugs Ther 8: 65-73, 1994
Paulson DJ: The diabetic heart is more sensitive to ischemic injury. Cardiovasc Res 34: 104-112, 1997
Feuvray D, Lopaschuk GD: Controversies on the sensitivity of the diabetic heart to ischemic injury: The sensitivity of the diabetic heart to ischemic injury is decreased. Cardiovasc Res 34: 113-120, 1997
Smith JM, Wahler GM: ATP-sensitive potassium channels are altered in ventricular myocytes from diabetic rats. Mol Cell Biochem 158: 43-51, 1996
Magyar J, Rusznák Z, Szentesi P, Szücs G, Kovács L: Action potential and potassium currents in rat ventricular muscle during experimental diabetes. J Moll Cell Cardiol 24: 841-853, 1992
Miura H, Breu M, Gutterman DD: Diabetes mellitus impairs ATP-sensitive potassium channel function of human coronary arterioles (Abstract). Circulation 96: 1-17, 1997
Gopalakrishnan M, Janis RA, Triggle DJ: ATP sensitive K+ channels: Pharmacologic properties, regulation and therapeutic potential. Drug Dev Res 28: 95-127, 1993
Gross GJ, Fryer RM: Sarcolemmal vs. mitochondrial ATP-sensitive K+ channels and myocardial preconditioning. Circ Res 84: 973-979, 1999
Kloner RA, Bolli R, Marban E, Reinlib L, Braunwald E et al.: Medical and cellular implications of stunning, hibernation and preconditioning. An NHKBI workshop. Circulation 97: 1848-1867, 1998
Ferdinandy P, Szilvassy Z, Baxter GF: Adaptation to myocardial stress in disease states: Is preconditioning a healthy heart phenomenon? Trend Pharmacol Sci 19: 223-229, 1998
Regan TJ, Ettinger PI, Khan MI et al.: Altered myocardial function and metabolism in chronic diabetes mellitus without ischemia in dogs. Circulation Res 35: 222-237, 1974
McCandless EL, Woodward BA, Dye JA: Alloxan diabetes in sheep under fasting and non-fasting conditions. Am J Physiol 154: 94-106, 1948
del Valle HF, Lascano EC, Negroni JA, Crottogini AJ: Glibenclamide effects on reperfusion-induced malignant arrhythmias and left ventricular mechanical recovery from stunning in conscious sheep. Cardiovasc Res 50: 474-485, 2001
Wilkinson JS: Spontaneous diabetes in domestic animals. Vet Rev Annot 3: 69-96, 1957
Shimoni Y, Firek L, Severson D, Giles W: Short-term diabetes alters K+ currents in rat ventricular myocytes. Circ Res 74: 620-628, 1957
Aomine M, Yamato T: Electrophysiological properties of ventricular muscle obtained from spontaneously diabetic mice. Exp Anim 49: 23-33, 2000
Ravingerova T, Stetka R, Pancza D, Ulicna O, Ziegelhöffer A, Styk J: Susceptibility to ischemia-induced arrhythmias and the effect of preconditioning in the diabetic rat heart. Physiol Res 49: 607-616, 2000
Tatsumi T, Matoba S, Kobara M et al.: Energy metabolism after ischemic preconditioning in streptozotocin-induced diabetic rat hearts. J Am Coll Cardiol 31: 707-715, 1998
Cleveland JC, Meldrum DR, Cain BS, Banerjee A, Harken AH: Oral sulfonylurea hypoglycemic agents prevent ischemic preconditioning in human myocardium. Two paradoxes revisited. Circulation 96: 29-32, 1997
Forrat R, Sebbag L, Wiernsperger N, Guidollet J, Renaud S, de Lorgeril M: Acute myocardial infarction in dogs with experimental diabetes. Cardiovasc Res 27: 1908-1912, 1993
Cole WC, McPherson, Sontag D: ATP-regulated K+ channels protect the myocardium against ischemia-reperfusion damage. Circ Res 69: 571-581, 1991
Shigematsu S, Sato T, Abe T et al.: Pharmacological evidence for the persistent activation of ATP-sensitive K+ channels in early phase of reperfusion and its protective role against myocardial stunning. Circulation 92: 2266-2275, 1995
Toyoda Y, Fries I, Parker RA, Levitsky S, McCully JD: Differential role of sarcolemmal and mitochondrial KATP channels in adenosine-enhanced ischemic preconditioning. Am J Physiol 279: H2694-H2703, 2000
Shimoni Y, Light PE, French RJ: Altered ATP sensitivity of ATP-dependent K+ channels in diabetic rat hearts. Am J Physiol Endocrinol Metab 275: E568-E576, 1998
Huuponen R: Adverse cardiovascular effects of Sulfonylurea drugs. Clinical significance. Med Toxicol 2: 190-209, 1987
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
del Valle, H.F., Lascano, E.C., Negroni, J.A. et al. Absence of ischemic preconditioning protection in diabetic sheep hearts: Role of sarcolemmal KATP channel dysfunction. Mol Cell Biochem 249, 21–30 (2003). https://doi.org/10.1023/A:1024797530152
Issue Date:
DOI: https://doi.org/10.1023/A:1024797530152