Abstract
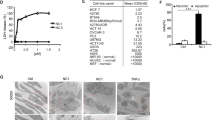
To evaluate the utility of tumor necrosis factor–related apoptosis–inducing ligand (TRAIL) as a cancer therapeutic, we created leucine zipper (LZ) forms of human (hu) and murine (mu) TRAIL to promote and stabilize the formation of trimers. Both were biologically active, inducing apoptosis of both human and murine target cells in vitro with similar specific activities. In contrast to the fulminant hepatotoxicity of LZ–huCD95L in vivo, administration of either LZ–huTRAIL or LZ–muTRAIL did not seem toxic to normal tissues of mice. Finally, repeated treatments with LZ–huTRAIL actively suppressed growth of the TRAIL–sensitive human mammary adenocarcinoma cell line MDA–231 in CB.17 (SCID) mice, and histologic examination of tumors from SCID mice treated with LZ–huTRAIL demonstrated clear areas of apoptotic necrosis within 9–12 hours of injection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Smith, C.A., Farrah, T. & Goodwin, R.G. The TNF receptor superfamily of cellular and viral proteins: activation, costimulation, and death. Cell 76, 959–962 (1994).
Cosman, D. A family of ligands for the TNF receptor superfamily. Stem Cells 12, 440–455 (1994).
Armitage, R.J. Tumor necrosis factor receptor superfamily members and their ligands. Curr. Opin. Immunol. 6, 407–413 (1994).
Peter, M.E., Ehret, A., Berndt, C. & Krammer, P.H. AIDS and the death receptors. Br. Med. Bull. 53, 604– 616 (1997).
Cohen, J. Cytokines as mediators of graft–versus–host disease. Bone Marrow Transplant. 3, 193–197 (1988).
Fisher, G.H. et al. Dominant interfering Fas gene mutations impair apoptosis in a human autoimmune lymphoproliferative syndrome. Cell 81, 935–946 (1995).
Wiley, S.R. et al. Identification and characterization of a new member of the TNF family that induces apoptosis. Immunity 3, 673–682 (1995).
Griffith, T.S., Chin, W.A., Jackson, G.C., Lynch, D.H. & Kubin, M.Z. Intracellular regulation of TRAIL–induced apoptosis in human melanoma cells. J. Immunol. 161, 2833–2840.
Pitti, R.M. et al. Induction of apoptosis by Apo–2 ligand, a new member of the tumor necrosis factor cytokine family. J. Biol. Chem. 271, 12687–12690 (1996).
Mariani, S.M., Matiba, B., Armandola, E.A. & Krammer, P.H. Interleukin 1 β–converting enzyme related proteases/caspases are involved in TRAIL–induced apoptosis of myeloma and leukemia cells. J. Cell. Biol. 137, 221–229 (1997).
Pan, G. et al. An antagonist decoy receptor and a death domain–containing receptor for TRAIL. Science 277, 815– 818 (1997).
Pan, G. et al. The receptor for the cytotoxic ligand TRAIL. Science 276, 111–113 (1997).
Sheridan, J.P. et al. Control of TRAIL–induced apoptosis by a family of signaling and decoy receptors. Science 277, 818– 821 (1997).
Walczak, H. et al. TRAIL–R2: a novel apoptosis–mediating receptor for TRAIL. EMBO J. 16, 5386– 5397 (1997).
Degli–Esposti, M.A. et al. Cloning and characterization of TRAIL–R3, a novel member of the emerging TRAIL receptor family. J. Exp. Med. 186, 1165–1170 (1997).
Degli–Esposti, M.A. et al. The novel receptor TRAIL–R4 induces NF–κB and protects against TRAIL–mediated apoptosis, yet retains an incomplete death domain. Immunity 7, 813– 820 (1997).
Marsters, S.A. et al. A novel receptor for Apo2L/TRAIL contains a truncated death domain. Curr. Biol. 7, 1003– 1006 (1997).
Fanslow, W.C. et al. Structural characteristics of CD40 ligand that determine biological function. Semin. Immunol. 6, 267– 278 (1994).
Schneider, P. et al. Conversion of membrane–bound Fas(CD95) ligand to its soluble form is associated with downregulation of its proapoptotic activity and loss of liver toxicity. J. Exp. Med. 187, 1205–1213 (1998).
Ogasawara, J. et al. Lethal effect of the anti–Fas antibody in mice. Nature 364, 806–809 (1993).
Tanaka, M., Suda, T., Yatomi, T., Nakamura, N. & Nagata, S. Lethal effect of recombinant human Fas ligand in mice pretreated with Propionibacterium acnes. J. Immunol. 158, 2303–2309 (1997).
Revel, M. & Schattner, A. Interferons: cytokines in autoimmunity. Ciba. Found. Symp. 129, 223– 233 (1987).
Cerami, A. & Beutler, B. The role of cachectin/TNF in endotoxic shock and cachexia. Immunol. Today 9, 28–31 (1988).
Fiers, W. Tumor necrosis factor. Characterization at the molecular, cellular and in vivo level. FEBS Lett. 285, 199– 212 (1991).
Elliott, M.J. et al. Randomised double–blind comparison of chimeric monoclonal antibody to tumour necrosis factor alpha (cA2) versus placebo in rheumatoid arthritis. Lancet 344, 1105– 1110 (1994).
Moreland, L.W. et al. Treatment of rheumatoid arthritis with a recombinant human tumor necrosis factor receptor (p75)–Fc fusion protein. N. Engl. J. Med. 337, 141–147 (1997).
Zheng, L.X. et al. Induction of apoptosis in mature T cells by tumour necrosis factor. Nature 377, 348– 351 (1995).
Vandenabeele, P., Declercq, W., Vanhaesebroeck, B., Grooten, J. & Fiers, W. Both TNF receptors are required for TNF–mediated induction of apoptosis in PC60 cells. J. Immunol. 154, 2904–2913 (1995).
Brouckaert, P.G., Leroux–Roels, G.G., Guisez, Y., Tavernier, J. & Fiers, W. In vivo anti–tumour activity of recombinant human and murine TNF, alone and in combination with murine IFN–γ, on a syngeneic murine melanoma. Int. J. Cancer 38, 763–769 (1986).
Havell, E.A., Fiers, W. & North, R.J. The antitumor function of tumor necrosis factor d(TNF). I. Therapeutic action of TNF against an established murine sarcoma is indirect, immunologically dependent, and limited by severe toxicity. J. Exp. Med. 167, 1067–1085 (1988).
Alderson, M.R. et al. Fas ligand mediates activation–induced cell death in human T lymphocytes. J. Exp. Med. 181, 71–77 (1995).
Dhein, J., Walczak, H., Bäumler, C., Debatin, K.–M. & Krammer, P.H. Autocrine T–cell suicide mediated by APO–1/(Fas/CD95). Nature 373, 438–441 (1995).
Brunner, T. et al. Cell–autonomous Fas (CD95)/Fas–ligand interaction mediates activation–induced apoptosis in T–cell hybridomas. Nature 373, 441–444 (1995).
Ju, S.–T. et al. Fas(CD95)/FasL interactions required for programmed cell death after T–cell activation. Nature 373, 444–448 (1995).
Lowin, B., Hahne, M., Mattmann, C. & Tschopp, J. Cytolytic T–cell–cytotoxicity is mediated through perforin and Fas ligand pathways. Nature 370, 650–653 (1994).
Kägi, D. et al. Cytotoxicity mediated by T cells and natural killer cells is greatly impaired in perforin–deficient mice. Nature 369, 31–37 (1994).
Galle, P.R. et al. Involvement of the CD95 (APO–1/Fas) receptor and ligand in liver damage. J. Exp. Med. 182, 1223– 1230 (1995).
Kondo, T., Suda, T., Fukuyama, H., Adachi, M. & Nagata, S. Essential roles of the Fas ligand in the development of hepatitis. Nature Med. 3, 409– 413 (1997).
Griffith, T.S., Brunner, T., Fletcher, S.M., Green, D.R. & Ferguson, T.A. Fas ligand–induced apoptosis as a mechanism of immune privilege. Science 270, 1189–1192 (1995).
Bellgrau, D. et al. A role for CD95 ligand in preventing graft rejection. Nature 377, 630–632 (1995).
French, L.E. et al. Fas and Fas ligand in embryos and adult mice: ligand expression in several immune–privileged tissues and coexpression in adult tissues characterized by apoptotic cell turnover. J. Cell. Biol. 133, 335–343 (1996).
Hahne, M. et al. Melanoma cell expression of Fas(Apo–1/CD95) ligand: implications for tumor immune escape. Science 274, 1363–1366 (1996).
Strand, S. et al. Lymphocyte apoptosis induced by CD95 (APO–1/Fas) ligand–expressing tumor cells—a mechanism of immune evasion? Nature Med. 2, 1361–1366 (1996).
Matiba, B., Mariani, S.M. & Krammer, P.H. The CD95 system and the death of a lymphocyte. Semin. Immunol. 9, 59–68 (1997).
Nagata, S. Apoptosis by death factor. Cell 88, 355– 365 (1997).
Emery, J.G. et al. Osteoprotegerin is a receptor for the cytotoxic ligand TRAIL. J. Biol. Chem. 273, 14363– 14367 (1998).
Acknowledgements
The authors thank D. Williams, K. Mohler and M. Widmer for their critical comments and A. Aumell for her expert editorial help in the production of this manuscript. H. Walczak is supported by the AIDS Stipend Program of the Bundesministerium fur Forschung und Technologie (German Ministry for Research and Technology).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Walczak, H., Miller, R., Ariail, K. et al. Tumoricidal activity of tumor necrosis factor–related apoptosis–inducing ligand in vivo. Nat Med 5, 157–163 (1999). https://doi.org/10.1038/5517
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/5517
This article is cited by
-
Exosome-based delivery strategies for tumor therapy: an update on modification, loading, and clinical application
Journal of Nanobiotechnology (2024)
-
The E3 ubiquitin ligase Itch regulates death receptor and cholesterol trafficking to affect TRAIL-mediated apoptosis
Cell Death & Disease (2024)
-
Venetoclax efficacy on acute myeloid leukemia is enhanced by the combination with butyrate
Scientific Reports (2024)
-
Harnessing TRAIL-induced cell death for cancer therapy: a long walk with thrilling discoveries
Cell Death & Differentiation (2023)
-
TRAIL-mediated signaling in bladder cancer: realization of clinical efficacy of TRAIL-based therapeutics in medical oncology
Medical Oncology (2023)