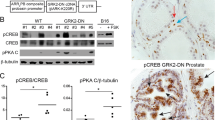
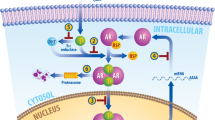
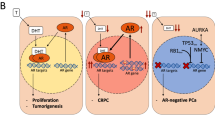
Abstract
Prostate cancer progresses from a hormone-sensitive, androgen-dependent stage to a hormone-refractory, androgen-independent tumor. The androgen receptor pathway functions in these androgen-independent tumors despite anti-androgen therapy. In our LAPC-4 prostate cancer model, androgen-independent sublines expressed higher levels of the HER-2/neu receptor tyrosine kinase than their androgen-dependent counterparts. Forced overexpression of HER-2/neu in androgen-dependent prostate cancer cells allowed ligand-independent growth. HER-2/neu activated the androgen receptor pathway in the absence of ligand and synergized with low levels of androgen to 'superactivate' the pathway. By modulating the response to low doses of androgen, a tyrosine kinase receptor can restore androgen receptor function to prostate cancer cells, a finding directly related to the clinical progression of prostate cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Talpin, M.E. et al. Mutation of the androgen-receptor gene in metastatic androgen-independent prostate cancer. N. Engl. J. Med. 332, 1393 –1398 (1995).
Gaddipati, J.P. et al. Frequent detection of codon 877 mutation in the androgen receptor gene in advanced prostate cancers. Cancer Res. 54, 2861–2864 (1994).
Visakorpi, T. et al. In vivo amplification of the androgen receptor gene and progression of human prostate cancer. Nature Genet. 9, 401–406 (1995).
Veldscholte, J. et al. The androgen receptor in LNCaP cells contains a mutation in the ligand binding domain which affects steroid binding characteristics and response to antiandrogens. J. Steroid Biochem. Mol. Biol. 41, 665–669 (1992).
Ignar-Trowbridge, D.M. et al. Coupling of dual signaling pathways: epidermal growth factor action involves the estrogen receptor. Proc. Natl. Acad. Sci. USA 89, 4658–4662 ( 1992).
Power, R.F., Mani, S.K., Codina, J., Conneely, O.M. & O'Malley, B.W. Dopaminergic and ligand-independent activation of steroid hormone receptors. Science 254, 1636–1639 (1991).
Aronica, S.M. & Katzenellenbogen, B.S. Stimulation of estrogen receptor-mediated transcription and alteration in the phosphorylation state of the rat uterine estrogen receptor by estrogen, cyclic adenosine monophosphate, and insulin-like growth factor-I. Mol. Endocrinol. 7, 743–752 (1993).
Denner, L.A., Weigel, N.L., Maxwell, B.L., Schrader, W.T., & O'Malley, B.W. Regulation of progesterone receptor-mediated transcription by phosphorylation. Science 250, 1740–1743 (1990).
Kato, S. et al. Activation of the estrogen receptor through phosphorylation by mitogen-activated protein kinase. Science 270, 1491–1494 (1995).
Culig, Z. et al. Androgen receptor activation in prostatic tumor cell lines by insulin-like growth factor-I, keratinocyte growth factor, and epiderman growth factor. Cancer Res. 54, 5474– 5478 (1994).
Slamon, D.J. et al. Studies of the Her-2/neu proto-oncogene in human breast and ovarian cancer. Science 244, 707– 712 (1989).
Adnane, J. et al. Proto-oncogene amplification and human breast tumor phenotype. Oncogene 4, 1389–1395 (1989).
Zeillinger, R. et al. HER-2 amplification, steroid receptors and epidermal growth factor receptor in primary breast cancer. Oncogene 4, 109–114 (1989).
Borg, A. et al. ERBB2 amplification is associated with tamoxifen resistance in steroid-receptor positive breast cancer. Cancer Lett. 81, 137–144 (1994).
Leitzel, K. et al. Elevated serum c-erbB-2 antigen levels and decreased response to hormone therapy of breast cancer. J. Clin. Oncol. 13, 1129–1135 (1995).
Pietras, R.J. et al. HER-2 tyrosine kinase pathway targets estrogen receptor and promotes hormone-independent growth in human breast cancer cells. Oncogene 10, 2435–2446 ( 1995).
Ware, J.L., Maygarden, S.J., Koontz, W.W. Jr. & Strom, S.C. Immunohistochemical detection of c-erbB-2 protein in human benign and neoplastic prostate. Hum. Pathol. 22, 254–258 (1991).
Robinson, D., He, F., Preglow, T. & Kung, H.J. A tyrosine kinase profile of prostate carcinoma. Proc. Natl. Acad. Sci. USA 93, 5958–5962 (1996).
Lyne, J.C. et al. Tissue expression of neu differentiation factor/heregulin and its receptor complex in prostate cancer and its biological effects on prostate cancer cells in vitro. Cancer J. Sci. Am. 3, 21–30 (1997).
Mellon, K. et al. p53, c-erbB-2 and the epidermal growth factor receptor in the benign and malignant prostate. J. Urol. 147, 496–499 (1992).
Kuhn, E.J., Kurnot, R.A., Sesterhenn, I.A., Chang, E.H. & Moul, J.W. Expression of the c-erb-B-2 (HER-2/neu) oncoprotein in human prostatic carcinoma. J. Urol. 150, 1427–1433 (1993).
Sadasivan, R. et al. Overexpression of HER-2/Neu may be an indicator of poor prognosis in prostate cancer. J. Urol. 150, 126– 131 (1993).
Ross, J.S. et al. Prognostic significance of HER-2/neu gene amplification status by fluorescence in situ hybridization of prostate carcinoma. Cancer 79, 2162–2170 ( 1997).
Arai, Y., Tatsuhiro, T. & Yoshida, O. c-erbB-2 oncoprotein: a potential biomarker of advanced prostate cancer. Prostate 30, 195– 201 (1997).
Klein, K.A. et al. Progression of metastatic human prostate cancer to androgen independence in immunodeficient SCID mice. Nature Med. 3, 402–408 (1997).
Romijn, J.C., Verkoelen, C.F. & Schroeder, F.H. Application of the MTT assay to human prostate cancer cell lines in vitro: establishment of test conditions and assessment of hormone-stimulated growth and drug-induced cytostatic and cytotoxic effects. Prostate 12, 99–110 (1988).
Nagabhushan, M. et al. CWR22: the first human prostate cancer xenograft with strongly androgen-dependent and relapsed strains both in vivo and in soft agar. Cancer Res. 56, 3042–3046 (1996).
Kyprianou, N., English, HF. & Isaacs, JT. Programmed cell death during regression of PC-82 human prostate cancer following androgen ablation. Cancer Res. 50, 3748–3753 (1990).
Wolf, D.A., Schulz, P. & Fittler, F. Transcriptional regulation of prostate kallikrein-like genes by androgen. Mol. Endocrinol. 6, 753 –762 (1992).
Cleutjens, K.B.J.M. et al. An androgen response element in a far upstream enhancer region is essential for high, androgen-regulated activity of the prostate-specific antigen promoter. Mol. Endocrinol. 11, 148 –161 (1997).
Schurr, E.R. et al. Prostate-specific antigen expression is regulated by an upstream enhancer. J. Biol. Chem. 271, 7043– 7051 (1996).
Pang, S. et al. Identification of a positive regulatory element responsible for tissue-specific expression of prostate-specific antigen. Cancer Res. 57, 495–499 ( 1997).
Wang, E.H., Zou, S. & Tjian, R. TAFII250-dependent transcription of cyclin A is directed by ATF activator proteins. Genes Dev. 11, 2658– 2669 (1997).
Riegman, P.H.J., Vlietstra, R.J., van der Korput, J.A.G.M., Brinkmann, A.O. & Trapman, J. The promoter of the prostate-specific antigen gene contains a functional androgen responsive element. Mol. Endocrinol. 5, 1921– 1930 (1991).
Chan, J.M. et al. Plasma insulin-like growth factor-I and prostate cancer risk: a prospective study. Science 279, 563– 566 (1998).
Burfeind, P., Chernicky, C.L., Rininsland, F. & Ilan, J. Antisense RNA to the type I insulin-like growth factor receptor suppresses tumor growth and prevents invasion by rat prostate cancer cell in vivo . Proc. Natl. Acad. Sci. USA 93, 7263 –7268 (1996).
Ben-Levy, R., Paterson, H.F., Marshall, C.J. & Yarden, Y. A single autophosphorylation site confers oncogenicity to the Neu/ErbB-2 receptor and enables coupling to the MAP kinase pathway. EMBO J. 13, 3302–3311 (1994).
Bunone, G., Briand, P.-A., Miksicek, R.J. & Picard, D. Activation of the unliganded estrogen receptor by EGF involves the MAP kinase pathway and direct phosphorylation. EMBO J. 15, 2174–2183 (1996).
Janknecht, R. & Hunter, T. A growing coactivator network. Nature 383, 22–23 ( 1996).
Glass, C.K., Rose, D.W. & Rosenfeld, M.G. Nuclear receptor coactivators. Curr. Opin. Cell. Biol. 9, 222–232 ( 1997).
Cobleigh, M.A. et al. Efficacy and safety of HerceptinTM (humanized anti-HER2 antibody) as a single agent in 222 women with HER2 overexpression who relapsed following chemotherapy for metastatic breast cancer. Progr/Proc. Am. Soc. Clin. Oncol. 17, 97a (1998).
Slamon, D. et al. Addition of Herceptin (humanized anti-HER2 antibody) to first line chemotherapy for HER2 overexpressing metastatic breast cancer (HER2+/MBC) markedly increases anticancer activity: a randomized, multinational controlled phase III trial. Progr/Proc. Am. Soc. Clin. Oncol. 17, 98a (1998).
Chazin, V.R., Kaleko, M., Miller, A.D. & Slamon, D.J. Transformation mediated by the human HER-2 gene independent of the epidermal growth factor receptor. Oncogene 7, 1859– 1866 (1992).
Emami, K.H. & Carey, M. A synergistic increase in potency of a multimerized VP16 transcriptional activation domain. EMBO J. 11, 5005–5012 ( 1992).
Raitano, A.B., Halpern, J.R., Hambuch, T.M. & Sawyers, C.L. The Bcr-Abl leukemia oncogene activates Jun Kinase and requires Jun for transformation. Proc. Natl. Acad. Sci. USA 92, 11746– 50 (1995).
Acknowledgements
We thank J. Redula and C. Tran for assistance with animal experiments, A. Raitano for advice during the early phases of this work, D. Slamon and A. Belldegrun for reagents and D. Reese for discussions. This work was supported by grants from CaP CURE, the Margaret Early Trust and the James S. McDonnell Foundation and NIH #GM08042.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Craft, N., Shostak, Y., Carey, M. et al. A mechanism for hormone-independent prostate cancer through modulation of androgen receptor signaling by the HER-2/neu tyrosine kinase. Nat Med 5, 280–285 (1999). https://doi.org/10.1038/6495
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/6495
This article is cited by
-
Endosomal recycling inhibitors downregulate the androgen receptor and synergise with enzalutamide
Investigational New Drugs (2024)
-
The extent of androgen receptor and HER2 expression allows for targeted therapy in most cases of salivary duct carcinoma: analysis of clinical and histopathological data in a tertiary care center
European Archives of Oto-Rhino-Laryngology (2024)
-
Circulating tumor cell gene expression and plasma AR gene copy number as biomarkers for castration-resistant prostate cancer patients treated with cabazitaxel
BMC Medicine (2022)
-
RNF8 up-regulates AR/ARV7 action to contribute to advanced prostate cancer progression
Cell Death & Disease (2022)
-
Androgen receptor phosphorylation at serine 81 and serine 213 in castrate-resistant prostate cancer
Prostate Cancer and Prostatic Diseases (2020)