Abstract
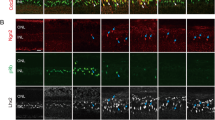
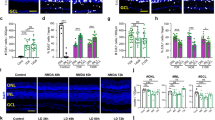
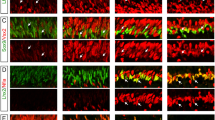
The retina of warm-blooded vertebrates is believed to be incapable of neural regeneration. Here we provide evidence that the retina of postnatal chickens has the potential to generate new neurons. In response to acute damage, numerous Müller glia re-entered the cell cycle, and shortly thereafter, expressed CASH-1, Pax6 and Chx10, transcription factors expressed by embryonic retinal progenitors. These progenitor-like cells transiently expressed neurofilament. Newly formed cells became distributed throughout the inner and outer nuclear layers of the retina, and remained for at least three weeks after damage. Some of these newly formed cells differentiated into retinal neurons, a few formed Müller glia, and most remained undifferentiated, with continued expression of Pax6 and Chx10. These cells continued to proliferate when grown in culture, with some differentiating into retinal neurons or Müller glia. We propose that, in response to damage, Müller glia in the retina are a potential source of neural regeneration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Raymond, P. A. & Hitchcock, P. F. Retinal regeneneration: common principles but diversity of mechanisms. Adv. Neurol. 72, 171–184 (1997).
Reh, T. A. & Levine, E. M. Multipotential stem cells and progenitors in the vertebrate retina. J. Neurobiol. 80, 206–220 (1988).
Johns, P. R. & Fernald, R. D. Genesis of rods in teleost fish retina. Nature 293, 141–142 (1981).
Johns, P. R. Formation of photoreceptors in larval and adult goldfish. J. Neurosci. 176, 343–357 (1982).
Goldman, S. A. & Nottebohm, F. Neuronal production, migration, and differentiation in a vocal control nucleus of the adult female canary brain. Proc. Natl. Acad. Sci. USA 80, 2390–2394 (1983).
Fischer, A. J. & Reh, T. A. Identification of a proliferating marginal zone of retinal progenitors in postnatal chickens. Dev. Biol. 220, 197–210 (2000).
Reynolds, B. A. & Weiss, S. Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science 255, 1707–1710 (1992).
Tropepe, V. et al. Retinal stem cells in the adult mouse eye. Science 287, 2032–2036 (2000).
Scharff, C., Kirn, J. R., Grossman, M., Macklis, J. D. & Nottenbohm, F. Targeted neuronal death affects replacement and vocal behavior in adult songbirds. Neuron 25, 481–492 (2000).
Magavi, S. S., Leavitt, B. R. & Macklis, J. D. Induction of neurogenesis in the neocortex of adult mice. Nature 405, 951–955 (2000).
Coulombre, J. L. & Coulombre, A. J. Regeneration of neural retina from pigmented epithelium in the chick embryo. Dev. Biol. 12, 79–92 (1965).
Park, C. M. & Hollenberg, M. J. Basic fibroblast growth factor induces retinal regeneration in vivo. Dev. Biol. 134, 201–205 (1989).
Willbold, E. & Layer, P. G. A hidden retina regenerative capacity from the chick ciliary margin is reactivated in vitro, that is accompanied by down-regulation of butyrylcholinesterase. Eur. J. Neurosci. 4, 210–220 (1991).
Reh, T.A. & Pittack, C. Transdifferentiation and retinal regeneration. Semin. Cell Biol. 6, 137–142 (1995).
Prada, C., Puga, J., Pérez-Méndez, L., Lóper, R. & Ramírez, G. Spatial and temporal patterns of neurogenesis in the chick retina. Eur. J. Neurosci. 3, 559–569 (1991).
Gould, E. & Tanapat, B. S. Lesion-induced proliferation of neuronal progenitors in the dentate gyrus of the adult rat. Neuroscience 80, 427–436 (1997).
Reh, T. A. Cell-specific regulation of neuronal production in the larval frog retina. J. Neurosci. 7, 3317–3324 (1987).
Barrington, M. J., Sattayasai, J., Zappia, J. & Ehrlich . Excitatory amino acids interfere with normal eye growth in posthatch chicks. Curr. Eye Res. 8, 781–792 (1989).
Ehrlich, D., Sattayasai, J., Zappia, J. & Barrington, M. in Myopia and the Control of Eye Growth—Symposium No. 155 (eds. Bock, G. R. & Widdows, K.) 63–84 (John Wiley & Sons, Chichester, England, 1990).
Tung, N. N., Morgan, I. G. & Ehrlich, D. A quantitative analysis of the effects of excitatory neurotoxins on retinal ganglion cells in the chick. Vis. Neurosci. 4, 217–223 (1990).
Fischer, A. J., Seltner, R. L. P., Poon, J. & Stell, W. K. Immunocytochemical characterization of NMDA and QA-induced excitotoxicity in the retina of chicks. J. Comp. Neurol. 393, 1–15 (1998).
Chadee, D. N. et al. Increased phosphorylation of histone H1 in mouse fibroblasts transformed with oncogenes or constitutively active mitogen-activated protein kinase kinase. J. Biol. Chem. 270, 20098–20105 (1995).
Mahadevan, L. C., Willis, A. C. & Baratt, M. S. Rapid histone H3 phosphorylation in response to growth factors, phorbol esters, okadaic acid, and protein synthesis inhibitors. Cell 65, 775–783 (1991).
Ajiro, K., Yoda, K., Utsumi, K. & Nishikawa, Y. Alteration of cell cycle-dependent histone phosphorylations by okadaic acid. Induction of mitosis-specific H3 phosphorylation and chromatin condensation in mammalian interphase cells. J. Biol. Chem. 271, 13197–13207 (1996).
Belecky-Adams, T. et al. Pax-6, Prox 1, and Chx10 homeobox gene expression correlates with phenotypic fate of retinal precursor cells. Invest. Ophthal. Vis. Sci. 38, 1293–1303 (1997).
Jasoni, C. L., Walker, M. B., Morrism M. D. & Reh T. A. A chicken acheate-scute homolog (CASH-1) is expressed in a temporally and spatially discrete manner in the developing nervous system. Development 120, 769–783 (1994).
Fischer, A. J., Wallman, J., Mertz, J. & Stell, W. K. Localization of retinoid binding proteins, receptors, and synthetic enzymes to the ocular tissues of the chick. J. Neurocytol. 58, 697–609 (1999).
Braisted, J. E., Essman, T. F. & Raymond, P. A. Selective regeneration of photoreceptors in goldfish retina. Development 122, 1427–1438 (1994).
Negishi, K. et al. Induction of immunoreactive proliferating cell nuclear antigen (PCNA) in goldfish retina following intravitreal injection with tunicamycin. Brain Res. Dev. Brain Res. 63, 71–83 (1991).
Oesterie, E. C. & Rubel, E. W. Hair cell generation in vestibular sensory receptor epithelia. Ann. NY Acad. Sci. 78, 34–46 (1996).
Doetsch, F., Caille, I., Lim, D. A., Garcia-Verdugo, J. M. & Alvarez-Buylla, A. Subventricular zone astrocytes are neural stem cells in the adult mammalian brain. Cell 11, 703–716 (1999).
Tomita, K., Nakanishi, S., Guillemot, F. & Kageyama, R. Mash1 promotes neuronal differentiation in the retina. Genes Cells 1, 765–774 (1996).
Takahashi, M., Palmer, T. D., Takahashi, J. & Gage, F. H. Widespread integration and survival of adult-derived neural progenitor cells in the developing optic retina. Mol. Cell. Neurosci. 12, 340–348 (1998).
Anchan, R. M., Reh, T. A., Angello, J., Balliet, A. & Walker, M. EGF and TGF-α stimulate retinal neuroepithelial cell proliferation in vitro. Neuron 6, 923–936 (1991).
Acknowledgements
We thank B. Dierks for technical assistance, and W.K. Stell, D. Raible and J. Hurley for comments on the manuscript. The BrdU, Pax6, vimentin, neurofilament and LEP-100 antibodies developed by S.J. Kaufman, A. Kawakami, J. Sanes, J. Wood and D.M. Fambrough, respectively, were obtained from the DSHB developed under auspices of the NICHD and maintained by the University of Iowa, Department of Biological Sciences, Iowa City, Iowa 52242. This work was supported by fellowships to A.J.F from the Alberta Heritage Foundation for Medical Research, the Medical Research Council of Canada, and the Fight for Sight Foundation Research Division of Prevent Blindness America, and by grants to T.A.R. from the National Science Foundation, NSF9604843 the Foundation Fighting Blindness and NIH NS28308.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fischer, A., Reh, T. Müller glia are a potential source of neural regeneration in the postnatal chicken retina. Nat Neurosci 4, 247–252 (2001). https://doi.org/10.1038/85090
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/85090
This article is cited by
-
Transcriptomics of CD29+/CD44+ cells isolated from hPSC retinal organoids reveals a single cell population with retinal progenitor and Müller glia characteristics
Scientific Reports (2023)
-
MAP4Ks inhibition promotes retinal neuron regeneration from Müller glia in adult mice
npj Regenerative Medicine (2023)
-
Bioengineering strategies for restoring vision
Nature Biomedical Engineering (2022)
-
Age- and cell cycle-related expression patterns of transcription factors and cell cycle regulators in Müller glia
Scientific Reports (2022)
-
Aquaporin-4 Removal from the Plasma Membrane of Human Müller Cells by AQP4-IgG from Patients with Neuromyelitis Optica Induces Changes in Cell Volume Homeostasis: the First Step of Retinal Injury?
Molecular Neurobiology (2021)