Abstract
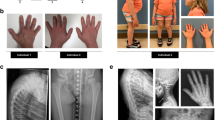
Perlecan is a large heparan sulfate (HS) proteoglycan present in all basement membranes and in some other tissues such as cartilage1,2, and is implicated in cell growth and differentiation3,4,5. Mice lacking the perlecan gene6,7 (Hspg2) have a severe chondrodysplasia with dyssegmental ossification of the spine and show radiographic, clinical and chondro-osseous morphology similar to a lethal autosomal recessive disorder in humans termed dyssegmental dysplasia, Silverman-Handmaker type (DDSH; MIM 224410). Here we report a homozygous, 89-bp duplication in exon 34 of HSPG2 in a pair of siblings with DDSH born to consanguineous parents, and heterozygous point mutations in the 5′ donor site of intron 52 and in the middle of exon 73 in a third, unrelated patient, causing skipping of the entire exons 52 and 73 of the HSPG2 transcript, respectively. These mutations are predicted to cause a frameshift, resulting in a truncated protein core. The cartilage matrix from these patients stained poorly with antibody specific for perlecan, but there was staining of intracellular inclusion bodies. Biochemically, truncated perlecan was not secreted by the patient fibroblasts, but was degraded to smaller fragments within the cells. Thus, DDSH is caused by a functional null mutation of HSPG2. Our findings demonstrate the critical role of perlecan in cartilage development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Noonan, D.M. et al. The complete sequence of perlecan, a basement membrane heparan sulfate proteoglycan, reveals extensive similarity with laminin A chain, low density lipoprotein-receptor, and the neural cell adhesion molecule. J. Biol. Chem. 266, 22939–22947 (1991).
SundarRaj, N., Fite, D., Ledbetter, S., Chakravarti, S. & Hassell, J.R. Perlecan is a component of cartilage matrix and promotes chondrocyte attachment. J. Cell Sci. 108, 2663–2672 (1995).
Aviezer, D. et al. Perlecan, basal lamina proteoglycan, promotes basic fibroblast growth factor-receptor binding, mitogenesis, and angiogenesis. Cell 79, 1005–1013 (1994).
Iozzo, R.V., Cohen, I.R., Grassel, S. & Murdoch, A.D. The biology of perlecan: the multifaceted heparan sulphate proteoglycan of basement membranes and pericellular matrices. Biochem. J. 302, 625–639 (1994).
Perrimon, N. & Bernfield, M. Specificities of heparan sulphate proteoglycans in developmental processes. Nature 404, 725–728 (2000).
Arikawa-Hirasawa, E., Watanabe, H., Takami, H., Hassell, J.R. & Yamada, Y. Perlecan is essential for cartilage and cephalic development. Nature Genet. 23, 354–358 (1999).
Costell, M. et al. Perlecan maintains the integrity of cartilage and some basement membranes. J. Cell Biol. 147, 1109–1122 (1999).
Aleck, K.A. et al. Dyssegmental dysplasias: clinical, radiographic, and morphologic evidence of heterogeneity. Am. J. Med. Genet. 27, 295–312 (1987).
Westvik, J. & Lachman, R.S. Coronal and sagittal clefts in skeletal dysplasias. Pediatr. Radiol. 28, 764–770 (1998).
Dodge, G.R. et al. Heparan sulfate proteoglycan of human colon: partial molecular cloning, cellular expression, and mapping of the gene (HSPG2) to the short arm of human chromosome 1. Genomics 10, 673–680 (1991).
Kallunki, P. et al. Cloning of human heparan sulfate proteoglycan core protein, assignment of the gene (HSPG2) to 1p36.1–p35 and identification of a BamHI restriction fragment length polymorphism. Genomics 11, 389–396 (1991).
Murdoch, A.D., Dodge, G.R., Cohen, I., Tuan, R.S. & Iozzo, R.V. Primary structure of the human heparan sulfate proteoglycan from basement membrane (HSPG2/perlecan). A chimeric molecule with multiple domains homologous to the low density lipoprotein receptor, laminin, neural cell adhesion molecules, and epidermal growth factor. J. Biol. Chem. 267, 8544–8557 (1992).
Kallunki, P. & Tryggvason, K. Human basement membrane heparan sulfate proteoglycan core protein: a 467-kD protein containing multiple domains resembling elements of the low density lipoprotein receptor, laminin, neural cell adhesion molecules, and epidermal growth factor. J. Cell Biol. 116, 559–571 (1992).
Cohen, I.R., Grassel, S., Murdoch, A.D. & Iozzo, R.V. Structural characterization of the complete human perlecan gene and its promoter. Proc. Natl. Acad. Sci. USA 90, 10404–10408 (1993).
Liu, W., Qian, C. & Francke, U. Silent mutation induces exon skipping of fibrillin-1 gene in Marfan syndrome. Nature Genet. 16, 328–329 (1997).
Celebi, J.T., Wanner, M., Ping, X.L., Zhang, H. & Peacocke, M. Association of splicing defects in PTEN leading to exon skipping or partial intron retention in Cowden syndrome. Hum. Genet. 107, 234–238 (2000).
Messiaen, L.M. et al. Exhaustive mutation analysis of the NF1 gene allows identification of 95% of mutations and reveals a high frequency of unusual splicing defects. Hum. Mutat. 15, 541–555 (2000).
Delot, E., Brodie, S.G., King, L.M., Wilcox, W.R. & Cohn, D.H. Physiological and pathological secretion of cartilage oligomeric matrix protein by cells in culture. J. Biol. Chem. 273, 26692–26697 (1998).
Hastbacka, J. et al. Atelosteogenesis type II is caused by mutations in the diastrophic dysplasia sulfate-transporter gene (DTDST): evidence for a phenotypic series involving three chondrodysplasias. Am. J. Hum. Genet. 58, 255–262 (1996).
Superti-Furga, A. et al. Achondrogenesis type IB is caused by mutations in the diastrophic dysplasia sulphate transporter gene. Nature Genet. 12, 100–102 (1996).
ul Haque, M.F. et al. Mutations in orthologous genes in human spondyloepimetaphyseal dysplasia and the brachymorphic mouse. Nature Genet. 20, 157–162 (1998).
Kurima, K. et al. A member of a family of sulfate-activating enzymes causes murine brachymorphism. Proc. Natl. Acad. Sci. USA 95, 8681–8685 (1998); erratum: 95, 12071 (1998).
Nicole, S. et al. Perlecan, the major proteoglycan of basement membranes, is altered in patients with Schwartz-Jampel syndrome (chondrodystrophic myotonia). Nature Genet. 26, 480–483 (2000).
David, G., Bai, X.M., Van der Schueren, B., Cassiman, J.J. & Van den Berghe, H. Developmental changes in heparan sulfate expression: in situ detection with mAbs. J. Cell Biol. 119, 961–975 (1992).
Brodie, S.G. et al. Radiographic and morphologic findings in a previously undescribed type of mesomelic dysplasia resembling atelosteogenesis type II. Am. J. Med. Genet. 80, 247–251 (1998).
Brodie, S.G., Lachman, R.S., McGovern, M.M., Mekikian, P.B. & Wilcox, W.R. Lethal osteosclerotic skeletal dysplasia with intracellular inclusion bodies. Am. J. Med. Genet. 83, 372–377 (1999).
Murdoch, A.D., Liu, B., Schwarting, R., Tuan, R.S. & Iozzo, R.V. Widespread expression of perlecan proteoglycan in basement membranes and extracellular matrices of human tissues as detected by a novel monoclonal antibody against domain III and by in situ hybridization. J. Histochem. Cytochem. 42, 239–249 (1994).
Gruber, H.E., Lachman, R.S. & Rimoin, D.L. Calcospherite (calcification nodule) size in the short rib polydactyly syndromes. Scanning Microsc. 4, 775–780 (1990).
Acknowledgements
We thank D. Rimoin, R. Lachman, E. Cukierman, E. Hoffman, C. Francomano, N. Ho, H. Watanabe, K. Kimata and H. Grand for comments; W. Cohen and J. King for referring the cases to the International Skeletal Dysplasia Registry; K. Yamada for anti-fibronectin antibodies; J. Couchman and R. Timpl for anti-perlecan antibodies; and B. Mekikian, K. Bolland and N. Hayes for technical support. W.R.W. was supported by NIH 5P01 HD22657. Some of this work was supported by grants from Seikagaku Corporation and from the Shriners of North America.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arikawa-Hirasawa, E., Wilcox, W., Le, A. et al. Dyssegmental dysplasia, Silverman-Handmaker type, is caused by functional null mutations of the perlecan gene. Nat Genet 27, 431–434 (2001). https://doi.org/10.1038/86941
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/86941
This article is cited by
-
Knockdown of hspg2 is associated with abnormal mandibular joint formation and neural crest cell dysfunction in zebrafish
BMC Developmental Biology (2021)
-
Spinal anesthesia in a patient with Schwartz–Jampel syndrome
JA Clinical Reports (2020)
-
Assembling custom side chains on proteoglycans to interrogate their function in living cells
Nature Communications (2020)
-
Exome sequencing identified rare variants in genes HSPG2 and ATP2B4 in a family segregating developmental dysplasia of the hip
BMC Medical Genetics (2017)