Abstract
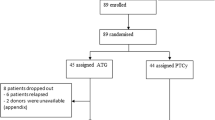
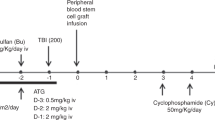
By retrospective analysis of 88 patients from the British Society of Blood and Marrow Transplantation registry, we investigated the effect of in vivo T-cell depletion in HLA-identical sibling reduced-intensity conditioning (RIC) allografts for adult AML by comparing patients who received alemtuzumab with those without alemtuzumab conditioning. Both groups were equivalent for age, sex, karyotype and disease status at transplant. With a median follow-up of 27 months (3–72 months) and 48 months (7–72 months), the 2- and 5-year overall survival, with or without alemtuzumab, is 60 and 60% (P=0.80) and 61 and 53%, respectively (P=0.85). The 2-year non-relapse mortality is 12% with alemtuzumab, and 17% without alemtuzumab (P=0.49). The 2-year relapse rate is 35% with alemtuzumab compared with 19% without alemtuzumab (P=0.28). Grades II–IV acute GVHD occurred in 22% (8/37) without alemtuzumab compared with 14% (7/51) given alemtuzumab (P=0.25). Extensive chronic GVHD occurred in 47% (14/30) not given alemtuzumab compared with 4% (2/45) who were given alemtuzumab (P=0.001). Among evaluable patients, the risk of infections was higher in those treated with alemtuzumab compared with those not treated with alemtuzumab (79 vs 57%, respectively, P=0.02). In conclusion, alemtuzumab has a beneficial effect by reducing chronic GVHD without affecting overall survival. Further studies are warranted before alemtuzumab can be recommended as standard in RIC allografts for AML.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Dick JE, Lapidot T . Biology of normal and acute myeloid leukemia stem cells. Int J Hematol 2005; 82: 389–396.
Bonnet D, Dick JE . Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med 1997; 3: 730–737.
Grimwade D, Walker H, Oliver F, Wheatley K, Harrison C, Harrison G et al. The importance of diagnostic cytogenetics on outcome in AML: analysis of 1612 patients entered into the MRC AML 10 trial. The Medical Research Council Adult and Children's Leukaemia Working Parties. Blood 1998; 92: 2322–2333.
Burnett AK, Wheatley K, Goldstone AH, Stevens RF, Hann IM, Rees JH et al. The value of allogeneic bone marrow transplant in patients with acute myeloid leukaemia at differing risk of relapse: results of the UK MRC AML 10 trial. Br J Haematol 2002; 118: 385–400.
Knapper S, Burnett AK, Littlewood T, Kell WJ, Agrawal S, Chopra R et al. A phase 2 trial of the FLT3 inhibitor lestaurtinib (CEP701) as first-line treatment for older patients with acute myeloid leukemia not considered fit for intensive chemotherapy. Blood 2006; 108: 3262–3270.
Lancet JE, Gojo I, Gotlib J, Feldman EJ, Greer J, Liesveld JL et al. A phase 2 study of the farnesyltransferase inhibitor tipifarnib in poor-risk and elderly patients with previously untreated acute myelogenous leukemia. Blood 2007; 109: 1387–1394.
Faderl S, Gandhi V, O’Brien S, Bonate P, Cortes J, Estey E et al. Results of a phase 1–2 study of clofarabine in combination with cytarabine (ara-C) in relapsed and refractory acute leukemias. Blood 2005; 105: 940–947.
Sharma S, Nadrous HF, Peters SG, Tefferi A, Litzow MR, Aubry MC et al. Pulmonary complications in adult blood and marrow transplant recipients: autopsy findings. Chest 2005; 128: 1385–1392.
Slavin S, Nagler A, Naparstek E, Kapelushnik Y, Aker M, Cividalli G et al. Nonmyeloablative stem cell transplantation and cell therapy as an alternative to conventional bone marrow transplantation with lethal cytoreduction for the treatment of malignant and nonmalignant hematologic diseases. Blood 1998; 91: 756–763.
Giralt S, Estey E, Albitar M, van Besien K, Rondon G, Anderlini P et al. Engraftment of allogeneic hematopoietic progenitor cells with purine analog-containing chemotherapy: harnessing graft-versus-leukemia without myeloablative therapy. Blood 1997; 89: 4531–4536.
Sandmaier BM, McSweeney P, Yu C, Storb R . Nonmyeloablative transplants: preclinical and clinical results. Semin Oncol 2000; 27: 78–81.
Kottaridis PD, Milligan DW, Chopra R, Chakraverty RK, Chakrabarti S, Robinson S et al. In vivo CAMPATH-1H prevents graft-versus-host disease following nonmyeloablative stem cell transplantation. Blood 2000; 96: 2419–2425.
Chakraverty R, Robinson S, Peggs K, Kottaridis PD, Watts MJ, Ings SJ et al. Excessive T cell depletion of peripheral blood stem cells has an adverse effect upon outcome following allogeneic stem cell transplantation. Bone Marrow Transplant 2001; 28: 827–834.
Chakraverty R, Sykes M . The role of antigen-presenting cells in triggering GVHD and GVL. Blood 2007; 110: 9–17.
Hale G, Waldmann H . CAMPATH-1 monoclonal antibodies in bone marrow transplantation. J Hematother 1994; 3: 15–31.
Fang L, Fehse B, Engel M, Zander A, Kroger N . Antithymocyte globulin induces ex vivo and in vivo depletion of myeloid and plasmacytoid dendritic cells. Transplantation 2005; 79: 369–371.
Dubey S, Nityanand S . Involvement of Fas and TNF pathways in the induction of apoptosis of T cells by antithymocyte globulin. Ann Hematol 2003; 82: 496–499.
Morris EC, Rebello P, Thomson KJ, Peggs KS, Kyriakou C, Goldstone AH et al. Pharmacokinetics of alemtuzumab used for in vivo and in vitro T-cell depletion in allogeneic transplantations: relevance for early adoptive immunotherapy and infectious complications. Blood 2003; 102: 404–406.
Bunjes D, Hertenstein B, Wiesneth M, Stefanic M, Novotny J, Duncker C et al. In vivo/ex vivo T cell depletion reduces the morbidity of allogeneic bone marrow transplantation in patients with acute leukaemias in first remission without increasing the risk of treatment failure: comparison with cyclosporin/methotrexate. Bone Marrow Transplant 1995; 15: 563–568.
Perez-Simon JA, Kottaridis PD, Martino R, Craddock C, Caballero D, Chopra R et al. Nonmyeloablative transplantation with or without alemtuzumab: comparison between 2 prospective studies in patients with lymphoproliferative disorders. Blood 2002; 100: 3121–3127.
Hale G, Jacobs P, Wood L, Fibbe WE, Barge R, Novitzky N et al. CD52 antibodies for prevention of graft-versus-host disease and graft rejection following transplantation of allogeneic peripheral blood stem cells. Bone Marrow Transplant 2000; 26: 69–76.
Kottaridis PD, Milligan DW, Chopra R, Chakraverty RK, Chakrabarti S, Robinson S et al. In vivo CAMPATH-1H prevents GvHD following nonmyeloablative stem-cell transplantation. Cytotherapy 2001; 3: 197–201.
Chakraverty R, Peggs K, Chopra R, Milligan DW, Kottaridis PD, Verfuerth S et al. Limiting transplantation-related mortality following unrelated donor stem cell transplantation by using a nonmyeloablative conditioning regimen. Blood 2002; 99: 1071–1078.
Delgado J, Thomson K, Russell N, Ewing J, Stewart W, Cook G et al. Results of alemtuzumab-based reduced-intensity allogeneic transplantation for chronic lymphocytic leukemia: a British Society of Blood and Marrow Transplantation Study. Blood 2006; 107: 1724–1730.
Tauro S, Craddock C, Peggs K, Begum G, Mahendra P, Cook G et al. Allogeneic stem-cell transplantation using a reduced-intensity conditioning regimen has the capacity to produce durable remissions and long-term disease-free survival in patients with high-risk acute myeloid leukemia and myelodysplasia. J Clin Oncol 2005; 23: 9387–9393.
D’Sa S, Peggs K, Pizzey A, Verfuerth S, Thuraisundaram D, Watts M et al. T- and B-cell immune reconstitution and clinical outcome in patients with multiple myeloma receiving T-cell-depleted, reduced-intensity allogeneic stem cell transplantation with an alemtuzumab-containing conditioning regimen followed by escalated donor lymphocyte infusions. Br J Haematol 2003; 123: 309–322.
Chakrabarti S, Avivi I, Mackinnon S, Ward K, Kottaridis PD, Osman H et al. Respiratory virus infections in transplant recipients after reduced-intensity conditioning with Campath-1H: high incidence but low mortality. Br J Haematol 2002; 119: 1125–1132.
Avivi I, Chakrabarti S, Milligan DW, Waldmann H, Hale G, Osman H et al. Incidence and outcome of adenovirus disease in transplant recipients after reduced-intensity conditioning with alemtuzumab. Biol Blood Marrow Transplant 2004; 10: 186–194.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J et al. 1994 Consensus conference on acute GVHD grading. Bone Marrow Transplant 1995; 15: 825–828.
Shulman HM, Sullivan KM, Weiden PL, McDonald GB, Striker GE, Sale GE et al. Chronic graft-versus-host syndrome in man. A long-term clinicopathologic study of 20 Seattle patients. Am J Med 1980; 69: 204–217.
Barnes DW, Corp MJ, Loutit JF, Neal FE . Treatment of murine leukaemia with X rays and homologous bone marrow; preliminary communication. Br Med J 1956; 2: 626–627.
Gale RP, Horowitz MM, Ash RC, Champlin RE, Goldman JM, Rimm AA et al. Identical-twin bone marrow transplants for leukemia. Ann Intern Med 1994; 120: 646–652.
Gorin NC, Labopin M, Fouillard L, Meloni G, Frassoni F, Iriondo A et al. Retrospective evaluation of autologous bone marrow transplantation vs allogeneic bone marrow transplantation from an HLA identical related donor in acute myelocytic leukemia. A study of the European Cooperative Group for Blood and Marrow Transplantation (EBMT). Bone Marrow Transplant 1996; 18: 111–117.
Weiden PL, Sullivan KM, Flournoy N, Storb R, Thomas ED . Antileukemic effect of chronic graft-versus-host disease: contribution to improved survival after allogeneic marrow transplantation. N Engl J Med 1981; 304: 1529–1533.
Horowitz MM, Gale RP, Sondel PM, Goldman JM, Kersey J, Kolb HJ et al. Graft-versus-leukemia reactions after bone marrow transplantation. Blood 1990; 75: 555–562.
Davies JK, Taussig DC, Oakervee H, Davies AJ, Agrawal SG, Gribben JG et al. Long-term follow-up after reduced-intensity conditioning allogeneic transplantation for acute myeloid leukemia/myelodysplastic syndrome: late CNS relapses despite graft-versus-host disease. J Clin Oncol 2006; 24: e23–e25.
Teshima T, Ordemann R, Reddy P, Gagin S, Liu C, Cooke KR et al. Acute graft-versus-host disease does not require alloantigen expression on host epithelium. Nat Med 2002; 8: 575–581.
Reddy P, Ferrara JL . Immunobiology of acute graft-versus-host disease. Blood Rev 2003; 17: 187–194.
Shah AJ, Kapoor N, Crooks GM, Weinberg KI, Azim HA, Killen R et al. The effects of Campath 1H upon graft-versus-host disease, infection, relapse, and immune reconstitution in recipients of pediatric unrelated transplants. Biol Blood Marrow Transplant 2007; 13: 584–593.
Barge RM, Starrenburg CW, Falkenburg JH, Fibbe WE, Marijt EW, Willemze R . Long-term follow-up of myeloablative allogeneic stem cell transplantation using Campath ‘in the bag’ as T-cell depletion: the Leiden experience. Bone Marrow Transplant 2006; 37: 1129–1134.
Lamba R, Carrum G, Myers GD, Bollard CM, Krance RA, Heslop HE et al. Cytomegalovirus (CMV) infections and CMV-specific cellular immune reconstitution following reduced intensity conditioning allogeneic stem cell transplantation with alemtuzumab. Bone Marrow Transplant 2005; 36: 797–802.
Acknowledgements
This study was performed on behalf of the Clinical Trials Committee of the BSBMT. We thank all of the data managers and transplantation physicians at the following participating centres for providing data and responding to additional requests: University Hospital of Wales, Cardiff; Leeds Teaching Hospitals NHS Trust; Sheffield Teaching Hospitals NHS Trust; Christie Hospital, Manchester; Addenbrookes Hospital, Cambridge; Nottingham City Hospital; The Royal Marsden Hospital, Sutton, Surrey; Royal Infirmary of Edinburgh; Birmingham Heartlands Hospital; St George's Hospital, London; John Radcliffe Hospital, Oxford; Manchester Royal Infirmary; St Bartholomew's Hospital, London; Queen Elizabeth Hospital, Birmingham; Southampton General Hospital; Hammersmith Hospital, London; Glasgow Royal Infirmary; Newcastle-upon-Tyne Hospitals; Bristol Childrens Hospital. AJP and TJL participate as members of the Oxford Biomedical Research Centre.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Malladi, R., Peniket, A., Littlewood, T. et al. Alemtuzumab markedly reduces chronic GVHD without affecting overall survival in reduced-intensity conditioning sibling allo-SCT for adults with AML. Bone Marrow Transplant 43, 709–715 (2009). https://doi.org/10.1038/bmt.2008.375
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2008.375
Keywords
This article is cited by
-
Population Pharmacokinetics of Alemtuzumab (Campath) in Pediatric Hematopoietic Cell Transplantation: Towards Individualized Dosing to Improve Outcome
Clinical Pharmacokinetics (2019)
-
Impact of pre-transplant co-morbidities on outcome after alemtuzumab-based reduced intensity conditioning allo-SCT in elderly patients: A British Society of Blood and Marrow Transplantation study
Bone Marrow Transplantation (2015)
-
IL15 levels on day 7 after hematopoietic cell transplantation predict chronic GVHD
Bone Marrow Transplantation (2013)
-
Methotrexate dose delivery is more important than ciclosporin level in graft-versus-host disease prophylaxis following T-replete reduced-intensity sibling allogeneic stem cell transplant
International Journal of Hematology (2011)
-
Alemtuzumab for the prevention and treatment of graft-versus-host disease
International Journal of Hematology (2011)