Abstract
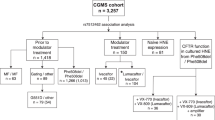
Cystic fibrosis (CF) is one of the most common life-shortening genetic disorders, and the CF transmembrane conductance regulator (CFTR) is the major causal gene. However, a substantial clinical variability among patients with identical CFTR genotypes suggests the presence of modifier genes. We tested the effect of four genes involved in Pseudomonas aeruginosa infection. Analysis of a primary cohort detected eight candidate polymorphisms that were genotyped in the secondary cohort of 1579 patients; lung function and age at first infection with P. aeruginosa were considered as the phenotypes. Both additive and codominant models were considered, adjusting for confounding variables but not for multiple comparisons. In the secondary cohort, heme oxygenase-1 (HMOX1) rs2071749 had the most significant effect on lung function in the pediatric group (P=0.01; Pcorrected=0.03), and complement factor 3 (C3) rs11569393 and HMOX1 rs2071746 in the adult groups (P=0.03 for both variants; Pcorrected=0.16, 0.09). No polymorphism of complement factor B (CFB) or toll-like receptor 4 (TLR4) had a significant modifying effect on lung function in either group. We have identified two genes that showed nominal association with disease severity among CF patients. However, because of the multiple comparisons made, further studies are required to confirm the interaction between these modifying genes and CFTR.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Bobadilla JL, Macek Jr M, Fine JP, Farrell PM . Cystic fibrosis: a worldwide analysis of CFTR mutations—correlation with incidence data and application to screening. Hum Mutat 2002; 19: 575–606.
Kerem E, Corey M, Kerem BS, Rommens J, Markiewicz D, Levison H et al. The relation between genotype and phenotype in cystic fibrosis—analysis of the most common mutation (ΔF508). N Engl J Med 1990; 323: 1517–1522.
Johansen HK, Nir M, Hoiby N, Koch C, Schwartz M . Severity of cystic fibrosis in patients homozygous and heterozygous for ΔF508 mutation. Lancet 1991; 337: 631–634.
Burke W, Aitken ML, Chen SH, Scott CR . Variable severity of pulmonary disease in adults with identical cystic fibrosis mutations. Chest 1992; 102: 506–509.
Santis G, Osborne L, Knight RA, Hodson ME . Independent genetic determinants of pancreatic and pulmonary status in cystic fibrosis. Lancet 1990; 336: 1081–1084.
Mekus F, Ballmann M, Bronsveld I, Bijman J, Veeze H, Tummler B . Categories of ΔF508 homozygous cystic fibrosis twin and sibling pairs with distinct phenotypic characteristics. Twin Res 2000; 3: 277–293.
Vanscoy LL, Blackman SM, Collaco JM, Bowers A, Lai T, Naughton K et al. Heritability of lung disease severity in cystic fibrosis. Am J Respir Crit Care Med 2007; 175: 1036–1043.
Drumm ML, Konstan MW, Schluchter MD, Handler A, Pace R, Zou F et al. Genetic modifiers of lung disease in cystic fibrosis. N Engl J Med 2005; 353: 1443–1453.
Dorfman R, Sandford A, Taylor C, Huang B, Frangolias D, Wang Y et al. Complex two-gene modulation of lung disease severity in children with cystic fibrosis. J Clin Invest 2008; 118: 1040–1049.
Hillian AD, Londono D, Dunn JM, Goddard KA, Pace RG, Knowles MR et al. Modulation of cystic fibrosis lung disease by variants in interleukin-8. Genes Immun 2008; 9: 501–508.
Taylor-Cousar JL, Zariwala MA, Burch LH, Pace RG, Drumm ML, Calloway H et al. Histo-blood group gene polymorphisms as potential genetic modifiers of infection and cystic fibrosis lung disease severity. PLoS One 2009; 4: e4270.
Gu Y, Harley IT, Henderson LB, Aronow BJ, Vietor I, Huber LA et al. Identification of IFRD1 as a modifier gene for cystic fibrosis lung disease. Nature 2009; 458: 1039–1042.
Hoiby N, Flensborg EW, Beck B, Friis B, Jacobsen SV, Jacobsen L . Pseudomonas aeruginosa infection in cystic fibrosis. Diagnostic and prognostic significance of Pseudomonas aeruginosa precipitins determined by means of crossed immunoelectrophoresis. Scand J Respir Dis 1977; 58: 65–79.
Wilmott RW, Tyson SL, Matthew DJ . Cystic fibrosis survival rates. The influences of allergy and Pseudomonas aeruginosa. Am J Dis Child 1985; 139: 669–671.
Currie AJ, Speert DP, Davidson DJ . Pseudomonas aeruginosa: role in the pathogenesis of the CF lung lesion. Semin Respir Crit Care Med 2003; 24: 671–680.
Mueller-Ortiz SL, Drouin SM, Wetsel RA . The alternative activation pathway and complement component C3 are critical for a protective immune response against Pseudomonas aeruginosa in a murine model of pneumonia. Infect Immun 2004; 72: 2899–2906.
Zhou H, Lu F, Latham C, Zander DS, Visner GA . Heme oxygenase-1 expression in human lungs with cystic fibrosis and cytoprotective effects against Pseudomonas aeruginosa in vitro. Am J Respir Crit Care Med 2004; 170: 633–640.
Nyholt DR . A simple correction for multiple testing for single-nucleotide polymorphisms in linkage disequilibrium with each other. Am J Hum Genet 2004; 74: 765–769.
Carlson CS, Eberle MA, Rieder MJ, Yi Q, Kruglyak L, Nickerson DA . Selecting a maximally informative set of single-nucleotide polymorphisms for association analyses using linkage disequilibrium. Am J Hum Genet 2004; 74: 106–120.
Horvath S, Xu X, Laird NM . The family based association test method: strategies for studying general genotype—phenotype associations. Eur J Hum Genet 2001; 9: 301–306.
Wang X, Dockery DW, Wypij D, Fay ME, Ferris Jr BG . Pulmonary function between 6 and 18 years of age. Pediatr Pulmonol 1993; 15: 75–88.
Hankinson JL, Odencrantz JR, Fedan KB . Spirometric reference values from a sample of the general U.S. population. Am J Respir Crit Care Med 1999; 159: 179–187.
Acknowledgements
We thank the CF Directors for their significant contribution and Clinic Coordinators at each CF center that enrolled subjects and ascertained phenotype data and DNA as part of the Canadian Cystic Fibrosis Genetic Modifier Study. We also thank all CF patients and their families. This work was supported by the Genome Canada through the Ontario Genomics Institute (2004-OGI-3-05 to PRD) and the Canadian Cystic Fibrosis Foundation (operating grants to PRD and AJS). DD is currently holding a Canada Research Chair on the Genetic Epidemiology of Complex Diseases and a Michael Smith Foundation for Health Research Career Scholar Award. AJS is the recipient of a Canada Research Chair in genetics and a Michael Smith Foundation for Health Research Senior Scholar Award.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Genes and Immunity website
Supplementary information
Rights and permissions
About this article
Cite this article
Park, J., Yung, R., Stefanowicz, D. et al. Cystic fibrosis modifier genes related to Pseudomonas aeruginosa infection. Genes Immun 12, 370–377 (2011). https://doi.org/10.1038/gene.2011.5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gene.2011.5
Keywords
This article is cited by
-
Significance of heme oxygenase-1(HMOX1) gene on fetal hemoglobin induction in sickle cell anemia patients
Scientific Reports (2020)
-
DNA methylation at modifier genes of lung disease severity is altered in cystic fibrosis
Clinical Epigenetics (2017)
-
Demographic, clinical, and laboratory parameters of cystic fibrosis during the last two decades: a comparative analysis
BMC Pulmonary Medicine (2015)
-
Polymorphisms in the glutathione pathway modulate cystic fibrosis severity: a cross-sectional study
BMC Medical Genetics (2014)
-
One thing leads to another: the cascade of obligations when researchers report genetic research results to study participants
European Journal of Human Genetics (2012)