Abstract
Introduction:
Intestinal electric stimulation (IES) is proposed as a potential tool for the treatment of morbid obesity. Our earlier study showed that IES with one pair of electrodes accelerated intestinal transit and decreased fat absorption in a segment of the jejunum in anesthetized rats. The aims of this study were to assess the effects of IES on whole gut transit and fat absorption in conscious rats, to examine the effects of multi-pairs IES and to explore the cholinergic mechanism behind the effects of IES.
Methods:
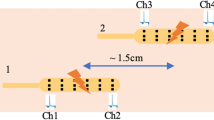
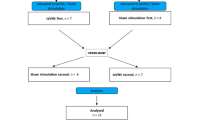
Thirty-eight male rats implanted with serosal electrodes were randomized into five groups: control without IES, two- or three-pairs IES with short pulses, atropine and atropine plus IES. The whole gut transit and fat remained and emptied from the gut were analyzed after continuous 2-h IES.
Results:
Two- and three-pairs IES significantly accelerated phenol red (PR, marker used for transit) excretion (analysis of variance (ANOVA), P<0.001). No significant difference was found between two- and three-pairs IES. Two-pairs IES significantly increased the excretion of fat (P<0.05). Atropine significantly blocked the accelerated transit induced by IES (ANOVA, P<0.001). Correlation was found between the percentage of PR and fat retained in the whole gut (r=0.497, P<0.01).
Conclusions:
IES accelerates whole gut transit and promotes fat excrement in conscious rats, and these effects are mediated through the cholinergic nerves. These findings are in support of the concept that IES may be a promising treatment option for obesity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
World Health Organisation. Diet Nutrition and the prevention of chronic diseases Report of a joint WHO/FAO expert consultation. WHO: Geneva, 2003 (WHO Technical Report Series, No. 916).
Cullen JJ, Kelly KA . The future of intestinal pacing. Gastroenterol Clin North Am 1994; 23: 391–402.
Abo M, Liang J, Qian L, Chen JD . Distension-induced myoelectrical dysrhythmia and effect of intestinal pacing in dogs. Dig Dis Sci 2000; 45: 129–135.
Chen JD, Lin HC . Electrical pacing accelerates intestinal transit slowed by fat-induced ileal brake. Dig Dis Sci 2003; 48: 251–256.
Sun Y, Qin C, Foreman RD, Chen JD . Intestinal electric stimulation modulates neuronal activity in the nucleus of the solitary tract in rats. Neurosci Lett 2005; 385: 64–69.
Xu J, McNearney TA, Chen JD . Gastric/Intestinal electrical stimulation modulates appetite regulatory peptide hormones in the stomach and duodenum in rats. Obes Surg 2007; 173: 406–413.
Yin J, Zhang J, Chen JD . Inhibitory Effects of Intestinal Electrical Stimulation on Food Intake, Weight Loss and Gastric Emptying in Rats. Am J Physiol Regul Integr Comp Physiol 2007; 293: R78–R82.
Sun Y, Chen J . Intestinal electric stimulation decreases fat absorption in rats: therapeutic potential for obesity. Obes Res 2004; 12: 1235–1242.
Dole VP . A relation between non-esterified fatty acids in plasma and the metabolism of glucose. J Clin Invest 1956; 35: 150–154.
Miller MS, Galligan JJ, Burks TF . Accurate measurement of intestinal transit in the rat. J Pharmacol Methods 1981; 6: 211–217.
Friedman MI, Ramirez I, Tordoff MG . Gastric emptying of ingested fat emulsion in rats: implications for studies of fat-induced satiety. Am J Physiol 1996; 270: R688–R692.
O'Connell PR, Kelly KA . Enteric transit and absorption after canine ileostomy. Effect of pacing. Arch Surg 1987; 122: 1011–1017.
Sawchuk A, Nogami W, Goto S, Yount J, Grosfeld JA, Lohmuller J et al. Reverse electrical pacing improves intestinal absorption and transit time. Surgery 1986; 100: 454–460.
Soper NJ, Geisler KL, Sarr MG, Kelly KA, Zinsmeister AR . Regulation of canine jejunal transit. Am J Physiol 1990; 259: G928–G933.
Amaris MA, Rashev PZ, Mintchev MP, Bowes KL . Microprocessor controlled movement of solid colonic content using sequential neural electrical stimulation. Gut 2002; 50: 475–479.
Zhang J, Tang M, Chen JD . Gastric electrical stimulation for obesity: the need for a new device using wider pulses. Obesity 2009; 17: 474–480.
Sanders KM . A case for interstitial cells of Cajal as pacemakers and mediators of neurotransmission in the gastrointestinal tract. Gastroent 1996; 111: 451–492.
Reiser SB, Weiser HF, Schusdziarra V, Siewert JR . Effect of pacing on small intestinal motor activity and hormonal response in dogs. Dig Dis Sci 1989; 34: 579–584.
Liu S, Liu J, Chen JD . Neural mechanisms involved in the inhibition of intestinal motility induced by intestinal electrical stimulation in conscious dogs. Neurogastroenterol Motil 2006; 18: 62–68.
Phillips RJ, Powley TL . Tension and stretch receptors in gastrointestinal smooth muscle: re-evaluating vagal mechanoreceptor electrophysiology. Brain Res Rev 2000; 34: 1–26.
Greenberg D . Intestinal satiety. In: Smith GP (ed). Satiation: From gut to brain. Oxford University Press: New York, 1998. pp 40–70.
Ekstrom J . Sensitization of the rat parotid gland to secretagogues following either parasympathetic denervation, sympathetic denervation, or decentralization. Acta Physiol Scand 1980; 108: 253–261.
Sundin T, Dahlstrom A . The sympathetic innervation of the urinary bladder and urethra in the normal state and after parasympathetic denervation of the spinal root level. Scand J Urol Nephrol 1973; 7: 131–449.
Agellon LB, Toth MJ, Thomson AB . Intracellular lipid binding proteins of the small intestine. Mol Cell Biochem 2002; 239: 79–82.
Cheng D, Nelson TC, Chen J, Walker SG, Wardwell-Swanson J, Meegalla R et al. Identification of acylcoenzyme A: monoacylglycerol acyltransferase 3, an intestinal specific enzyme implicated in dietary fat absorption. J Biol Chem 2003; 278: 13611–13614.
Ouyang H, Yin J, Chen JD . Gastric or intestinal electrical stimulation-induced increase in gastric volume is correlated with reduced food intake. Scand J Gastroenterol 2006; 41: 1261–1266.
Yin J, Ouyang H, Chen JD . Potential of intestinal electrical stimulation for obesity: a preliminary canine study. Obesity 2007; 15: 1133–1138.
Yin J, Zhang J, Chen JD . Inhibitory effects of intestinal electrical stimulation on food intake, weight loss and gastric emptying in rats. Am J Physiol Regul Integr Comp Physiol 2007; 293: R78–R82.
Acknowledgements
We would like to thank Professor Bela Szabo for his advice and technical supports. This study was partially supported by a grant from the National Institutes of Health (DK063733).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, Y., Chen, J. Intestinal electric stimulation accelerates whole gut transit and promotes fat excrement in conscious rats. Int J Obes 33, 817–823 (2009). https://doi.org/10.1038/ijo.2009.123
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2009.123
Keywords
This article is cited by
-
Intestinal electrical stimulation attenuates hyperglycemia and prevents loss of pancreatic β cells in type 2 diabetic Goto–Kakizaki rats
Nutrition & Diabetes (2019)
-
Hypoglycemic Effects of Intestinal Electrical Stimulation by Enhancing Nutrient-Stimulated Secretion of GLP-1 in Rats
Obesity Surgery (2018)
-
Pulse Width-Dependent Effects of Intestinal Electrical Stimulation for Obesity: Role of Gastrointestinal Motility and Hormones
Obesity Surgery (2017)
-
An Optimized IES Method and Its Inhibitory Effects and Mechanisms on Food Intake and Body Weight in Diet-Induced Obese Rats: IES for Obesity
Obesity Surgery (2017)
-
Electroacupuncture inhibits weight gain in diet-induced obese rats by activating hypothalamicLKB1-AMPK signaling
BMC Complementary and Alternative Medicine (2015)