Abstract
Objective:
To determine how adults in the United States view non-invasive prenatal testing using cell-free fetal DNA (cffDNA testing) in order to help estimate uptake.
Study Design:
A national sample of 1861 US-based adults was surveyed using a validated online survey instrument. The survey was administered by a commercial survey research company. Respondents were randomized to receive a survey about prenatal testing for trisomy 13 and 18 or trisomy 21. Participants were asked to select among testing modalities, including cffDNA testing, and rank the features of testing that they considered most important to decision making.
Result:
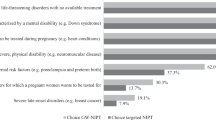
There was substantive interest in the use of cffDNA testing rather than traditional screening mechanisms, with a minority of respondents reporting that they would support the use of both methods in combination. The lower rates of false-negative and false-positive test results and the ability to use the test earlier in the pregnancy were the most highly rated benefits of cffDNA testing. Participants expressed strong support for diagnostic confirmation via invasive testing after a positive result from either screening or cffDNA testing. However, almost one-third of participants reported that they would not endorse the use of either invasive or non-invasive prenatal testing.
Conclusion:
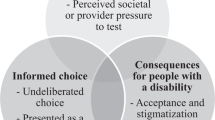
There appears to be support for uptake of non-invasive prenatal tests. Clinical guidelines should therefore go forward in providing guidance on how to integrate non-invasive methods into the current standard of care. However, our findings indicate that even when accuracy, which is rated by patients as the most important aspect of prenatal testing, is significantly improved over existing screening methods and testing is offered non-invasively, the number of individuals who reported that they would decline any testing remained the same. Attention should therefore be directed at ensuring that the right of informed refusal of prenatal testing is not impacted by new, non-invasive methods.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Zimmermann B, Hill M, Gemelos G, Demko Z, Banjevic M, Baner J et al Noninvasive prenatal aneuploidy testing of chromosomes 13, 18, 21, X, and Y, using targeted sequencing of polymorphic loci. Prenat Diagn 2012; 32 (13): 1233–1241.
Heger M . As rivals vie for share of noninvasive trisomy testing market, physicians see value for screening. GenomeWeb 2012. Available athttp://www.genomeweb.com/sequencing/rivals-vie-share-noninvasive-trisomy-testing-market-physicians-see-value-screeni
Allyse M, Sayres C, King S, Norton E, Cho K . Cell-free fetal DNA testing for fetal aneuploidy and beyond: Clinical integration challenges in the US context. Hum Reprod 2012; 27 (11): 3123–3131.
Sayres LC, Cho MK . Cell-free fetal nucleic acid testing: a review of the technology and its applications. Obstet Gynecol Surv 2011; 66 (7): 431.
Bianchi DW, Platt LD, Goldberg JD, Abuhamad AZ, Sehnert AJ, Rava RP . Genome-wide fetal aneuploidy detection by maternal plasma DNA sequencing. Obstet Gynecol 2012; 119 (5): 890–901.
Norton ME, Brar H, Weiss J, Karimi A, Laurent LC, Caughey AB et al Non-invasive chromosomal evaluation (NICE) study: results of a multicenter prospective cohort study for detection of fetal trisomy 21 and trisomy 18. Am J Obstet Gynecol 2012; 207 (2): 137 e1-8.
Agarwal A, Sayres LC, Cho MK, Cook-Deegan R, Chandrasekharan S . Commercial landscape of noninvasive prenatal testing in the united states. Prenat Diagn 2013; 33 (6): 521–531.
Tan W . Prenatal markets remain strong and fertile. Genet Eng News 2011; 31.
Garfield SS, Armstrong SO . Clinical and cost consequences of incorporating a novel non-invasive prenatal test into the diagnostic pathway for fetal trisomies. J Managed Care Med 2012; 15 (2): 34–41.
The American College of Obstetricians and Gynecologists Committee on Genetics, The Society for Maternal-Fetal Medicine Publications Committee. Committee Opinion Number 545, 2012. Available at http://www.acog.org/Resources_And_Publications/Committee_Opinions/Committee_on_Genetics/Noninvasive_Prenatal_Testing_for_Fetal_Aneuploidy (accessed January 2014).
Devers P, Cronister S, Ormond K, Facio F, Brasington C, Flodman P . Noninvasive prenatal testing/noninvasive prenatal diagnosis: the position of the national society of genetic counselors. J Genet Couns 2013; 22 (13): 291–295.
Kalfoglou AL, Doksum T, Bernhardt B, Geller G, LeRoy L, Mathews DJ et al. Opinions about new reproductive genetic technologies: Hopes and fears for our genetic future. Fertil Steril 2005; 83 (6): 1612–1621.
Tischler R, Hudgins L, Blumenfeld YJ, Greely HT, Ormond KE . Noninvasive prenatal diagnosis: pregnant women's interest and expected uptake. Prenat Diagn 2011; 31 (13): 1292–1299.
Zamerowski S, Lumley M, Arreola R, Dukes K, Sullivan L . Favorable attitudes toward testing for chromosomal abnormalities via analysis of fetal cells in maternal blood. Genet Med 2001; 3 (4): 301.
Lewis C, Hill M, Skirton H, Chitty LS . Non-invasive prenatal diagnosis for fetal sex determination: benefits and disadvantages from the service users' perspective. Eur J Hum Genet 2012; 20 (11): 1127–1133.
Sayres LC, Allyse M, Norton ME, Cho MK . Cell-free fetal DNA testing: a pilot study of obstetric healthcare provider attitudes toward clinical implementation. Prenat Diagn 2011; 31 (11): 1070–1076.
Ubel PA, Smith DM, Zikmund-Fisher BJ, Derry HA, McClure J, Stark A et al. Testing whether decision aids introduce cognitive biases: results of a randomized trial. Patient Educ Couns 2010; 80 (2): 158–163.
Farrimond R, Kelly E . Public viewpoints on new non-invasive prenatal genetic tests. Public Underst Sci 2013; 22 (6): 730–744.
Kelly SE, Farrimond HR . Non-invasive prenatal genetic testing: a study of public attitudes. Public Health Genomics 2012; 15 (2): 73–81.
Wald NJ, Bestwick JP . Incorporating DNA sequencing into current prenatal screening practice for Down's syndrome. PLoS One 2013; 8 (3): e58732.
Rose NC, Lagrave D, Hafen B, Jackson M . The impact of utilization of early aneuploidy screening on amniocenteses available for training in obstetrics and fetal medicine. Prenat Diagn 2013; 33 (3): 242–244.
Horsting JM, Dlouhy SR, Hanson K, Quaid K, Bai S, Hines KA . Genetic counselors' experience with cell-free fetal DNA testing as a prenatal screening option for aneuploidy. J Genet Couns 2013 (in press).
Musci TJ, Fairbrother G, Batey A, Bruursema J, Struble C, Song K . Non-invasive prenatal testing with cell-free DNA: US physician attitudes toward implementation in clinical practice. Prenat Diagn 2013; 33 (5): 424–428.
Norton ME, Rose NC, Benn P . Noninvasive prenatal testing for fetal aneuploidy: clinical assessment and a plea for restraint. Obstet Gynecol 2013; 121 (4): 847–850.
Chetty S, Garabedian MJ, Norton ME . Uptake of noninvasive prenatal testing (NIPT) in women following positive aneuploidy screening. Prenat Diagn 2013; 33 (6): 542–546.
Markens S, Browner C, Press N . 'Because of the risks': how US pregnant women account for refusing prenatal screening. Soc Sci Med 1999; 49: 359–369.
García E, Timmermans DR, van Leeuwen E . The impact of ethical beliefs on decisions about prenatal screening tests: earching for justification. Soc Sci Med 2008; 66 (3): 753–764.
Dahl K, Hvidman L, Jørgensen FS, Kesmodel US . Knowledge of prenatal screening and psychological management of test decisions. ltrasound. Obstet Gynecol 2011; 38 (2): 152–157.
Favre R, Moutel G, Duchange N, Vayssière C, Kohler M, Bouffet N et al. What about informed consent in first-trimester ultrasound screening for Down syndrome? Fetal Diagn Ther 2008; 23 (3): 173–184.
Ormond KE, Banuvar S, Daly A, Iris M, Minogue J, Elias S . Information preferences of high literacy pregnant women regarding informed consent models for genetic carrier screening. Patient Educ Couns 2009; 75 (2): 244–250.
Constantine M, Allyse M, Rockwood T, Wall M, De Vries R Imperfect informed consent for prenatal screening: lessons from the quad screen. Clin Ethics 2013 (in press).
Schmitz D, Netzer C, Henn W . An offer you can't refuse? Ethical implications of non-invasive prenatal diagnosis. Nat Rev Genet 2009; 10: 515.
Schmitz D, Henn W, Netzer C . Clinical review: Commentary: no risk, no objections? Ethical pitfalls of cell-free fetal DNA and RNA testing. BMJ 2009; 339: b2690.
Deans Z, Newson AJ . Should non-invasiveness change informed consent procedures for prenatal diagnosis? Health Care Anal 2011; 19 (2): 122–132.
van den Heuvel A, Chitty L, Dormandy E, Newson A, Deans Z, Attwood S et al. Will the introduction of non-invasive prenatal diagnostic testing erode informed choices? An experimental study of health care professionals. Patient Educ Couns 2010; 78 (1): 24–28.
Benn PA, Chapman AR . Ethical challenges in providing noninvasive prenatal diagnosis. Curr Opin Obstet Gynecol 2010; 22 (2): 128–134.
de Jong A, Dondorp WJ, Frints SG, de Die-Smulders CE, de Wert GM . Advances in prenatal screening: the ethical dimension. Nat Rev Genet 2011; 12 (9): 657–663.
Allyse MA, Sayres LC, Havard M, King JS, Greely HT, Hudgins L et al. Best ethical practices for clinicians and laboratories in the provision of non-invasive prenatal testing. Prenat Diagn 2013; 33 (7): 656–661.
Flessel M, Goldman S California prenatal screening program to include noninvasive testing. Acog.org 2013, http://www.acog.org/About_ACOG/ACOG_Departments/District_Newsletters/District_IX/September_2013/California_Prenatal_Screening_Program.
Natoli JL, Ackerman DL, McDermott S, Edwards JG . Prenatal diagnosis of down syndrome: a systematic review of termination rates (1995-2011). Prenat Diagn 2012; 32 (2): 142–153.
Skotko BG, Levine SP, Goldstein R . Having a son or daughter with Down syndrome: perspectives from mothers and fathers. Am J Med Genet A 2011; 155A (10): 2335–2347.
Skotko BG, Levine SP, Goldstein R . Self-perceptions from people with Down syndrome. Am J Med Genet A 2011; 155A (10): 2360–2369.
Sheets KB, Crissman BG, Feist CD, Sell SL, Johnson LR, Donahue KC et al. Practice guidelines for communicating a prenatal or postnatal diagnosis of down syndrome: recommendations of the national society of genetic counselors. J Genet Couns 2011; 20 (5): 432–441.
Kuppermann M, Learman LA, Gates E, Gregorich SE, Nease RF Jr, Lewis J et al. Beyond race or ethnicity and socioeconomic status: predictors of prenatal testing for Down syndrome. Obste Gynecol 2006; 107 (5): 1087.
Glasson EJ, Sullivan SG, Hussain R, Petterson BA, Montgomery PD, Bittles AH . The changing survival profile of people with Down's syndrome: implications for genetic counselling. Clin Genet 2002; 62 (5): 390–393.
Skotko BG, Kishnani PS, Capone GT Down Syndrome Diagnosis Study Group. Prenatal diagnosis of Down syndrome: how best to deliver the news. Am J Med Genet A 2009; 149A (11): 2361–2367.
Madeo AC, Biesecker BB, Brasington C, Erby LH, Peters KF . The relationship between the genetic counseling profession and the disability community: a commentary. Am J Med Genet A 2011; 155A (8): 1777–1785.
Acknowledgements
This work is funded by NIH grant P50 HG003389 (Center for Integrating Ethics and Genetic Research). Dr Cho is additionally supported by NIH grant 1 U54 RR024374-01A1 (Stanford Center for Clinical and Translational Education and Research).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Journal of Perinatology website
Supplementary information
Rights and permissions
About this article
Cite this article
Allyse, M., Sayres, L., Goodspeed, T. et al. Attitudes towards non-invasive prenatal testing for aneuploidy among US adults of reproductive age. J Perinatol 34, 429–434 (2014). https://doi.org/10.1038/jp.2014.30
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2014.30
Keywords
This article is cited by
-
Effects of an animated educational video on knowledge of cell-free DNA screening among Thai pregnant women: a randomized control trial
BMC Pregnancy and Childbirth (2023)
-
What do people think about genetics? A systematic review
Journal of Community Genetics (2019)
-
Attitudes Toward and Uptake of Prenatal Genetic Screening and Testing in Twin Pregnancies
Journal of Genetic Counseling (2018)
-
Genetic Counseling, Professional Values, and Habitus: An Analysis of Disability Narratives in Textbooks
Journal of Medical Humanities (2018)
-
“The Top Priority Is a Healthy Baby”: Narratives of Health, Disability, and Abortion in Online Pregnancy Forum Discussions in the US and China
Journal of Genetic Counseling (2017)