Abstract
Objective:
Increased use of non-invasive respiratory support (NRS) in the delivery room management of preterm neonates has resulted in delayed surfactant treatment, yet the short-term effects of this change are unknown. The aim of this study was to comparatively evaluate the use of surfactant and the short-term outcomes prior to and after the implementation of early routine use of NRS.
Study Design:
Eligible infants of <29 weeks gestational age (GA) admitted to a Canadian tertiary neonatal center during two time periods (2005 to 2008 and 2010 to 2013) were included in this retrospective cohort study. Timing of surfactant (prophylactic vs therapeutic) and short-term outcomes were compared between the two groups. Univariate and multivariate regression analysis was performed to determine the adjusted odds ratio (AOR) along with 95% confidence interval (CI) of receiving exogenous surfactant and developing bronchopulmonary dysplasia (BPD) using the later cohort as the reference group. Subgroup analyses were also performed for infants <26 and 26 to 286/7 weeks GA, respectively.
Results:
A total of 3980 and 5137 infants were included in the first and second time periods, respectively. There was no significant difference in overall surfactant utilization between the two time periods (AOR 1.00, 95% CI 0.89, 1.13). However, between 2005 and 2008, a lower proportion of neonates received therapeutic surfactant compared with the later cohort (47.1% vs 56.9%, P<0.01) but were more likely to receive prophylactic surfactant (52.9% vs 43.1%, P<0.01). BPD overall was significantly higher in the earlier cohort (AOR 1.19, 95% CI 1.07, 1.33), particularly among the <26 weeks gestation subgroup (AOR 1.34, 95% CI 1.08, 1.66).
Conclusion:
Early routine use of NRS did not impact overall surfactant utilization rate, although therapeutic surfactant administration rates were higher with a concomitant decrease in BPD rates.
Similar content being viewed by others
Introduction
Surfactant replacement therapy is now a well-established treatment for infants with respiratory distress syndrome (RDS). Several large meta-analyses, including the early clinical trials carried out in the 1980s and 1990s,1, 2 have shown that surfactant therapy reduces mortality with an approximate number needed to treat of 11 to prevent 1 neonatal death. These meta-analyses further confirmed that surfactant decreases the risk of pneumothorax, pulmonary interstitial emphysema and the combined outcome of death or bronchopulmonary dysplasia (BPD), primarily by reducing death rather than BPD.3, 4, 5, 6
Traditionally, surfactant is administered invasively with endotracheal intubation. Invasive surfactant delivery is accompanied by several possible complications, including bradycardia, hypoxemia and blockage of the endotracheal tube.7 In recent literature, less-invasive surfactant delivery has been associated with decreased use of mechanical ventilation and a lower likelihood of death or BPD but still carries with it the acute complications similar to invasive delivery.8
In order to reduce or eliminate invasive ventilation altogether, early non-invasive respiratory support (NRS) is increasingly used in the delivery room management of preterm neonates.9, 10 Many studies have suggested lower rates of BPD with NRS.10, 11, 12 A meta-analysis of seven trials, including two large randomized controlled trials of NRS vs prophylactic intubation, suggested that avoidance of endotracheal intubation had a beneficial impact of preventing BPD in infants <30 weeks gestational age (GA).13, 14, 15 This is of considerable importance given that BPD has been independently associated with poor neurological outcomes.16
The introduction of early NRS in the delivery room has led to questions regarding the ideal timing of surfactant administration. Recent systematic reviews have favored selective surfactant use (treatment of infants only after a period of observation for clinical signs and symptoms of RDS or respiratory failure) over prophylactic surfactant use (treatment at the time of birth or shortly thereafter to a selected group of infants at high risk of developing RDS based on GA)17 and early selective surfactant, as compared with delayed surfactant treatment.18, 19 With more consistent use of early NRS in the delivery room, it is important to understand the trends of surfactant use and the outcomes related to this evolution. Avoidance of endotracheal intubation and mechanical ventilation and increased use of NRS is likely to result in delayed use of surfactant. The use of early NRS may in fact eliminate the need for surfactant altogether in up to 50% of preterm neonates.20
During the implementation of the quality improvement project, Evidence-based Practice for Improving Quality in 2009, the use of NRS became increasingly adopted by neonatal units participating in the Canadian Neonatal Network (CNN).21 The aim of this study was to comparatively evaluate the use of surfactant therapy prior to the implementation of routine early use of NRS (2005 to 2008) and after its implementation (2010 to 2013) and to compare the short-term neonatal outcomes, including development of BPD, during the two time periods.
Methods
Study population
The CNN is a consortium of all tertiary-level neonatal units across the country. All newborn infants <29 weeks GA admitted to a CNN center between 1 January 2005 and 31 December 2008 and between 1 January 2010 and 31 December 2013 were evaluated for inclusion in this study. Infants who had major congenital anomalies, were outborn, were offered palliative care or were moribund at birth were excluded from the analysis.
Study design
This retrospective cohort study was conducted by extracting data from the CNN database from the two time periods described above. Data were extracted for the following categories: (1) Maternal data: age, gravity, parity, singleton or multiple gestation, antenatal steroids, maternal health problems (including diabetes mellitus, hypertension or urinary tract infection), Group B streptococcus status, prolonged rupture of membranes before delivery, maternal fever, chorioamnionitis, and intrapartum antibiotics; (2) Delivery and resuscitation measures: mode of delivery, presence of meconium, Apgar scores <7 at 5 min, requirement for resuscitation (positive-pressure ventilation and/or chest compressions); and (3) Neonatal data: GA, sex, birth weight, small for GA, Score for Neonatal Acute Physiology II, outborn/inborn, use of NRS including continuous positive airway pressure (CPAP), use of intubation and mechanical ventilation at birth, prophylactic (defined as surfactant administration ⩽30 min of life) or therapeutic (defined as administration after 30 min) surfactant administration, number of doses of surfactant, early use of caffeine (first 3 days of life), duration of endotracheal mechanical ventilation, duration of CPAP, duration of oxygen therapy, length of hospital stay and short-term outcomes (BPD, defined as need for oxygen at 36 weeks postmenstrual age (PMA)22 or at time of transfer to a level 2 unit, retinopathy of prematurity (ROP, stage ⩾3 according to international classification23), necrotizing enterocolitis (stage ⩾2 using Bell’s criteria24), sepsis (early and late onset), grade 3 or 4 intraventricular hemorrhage,25 mortality before discharge, air leak syndromes (pneumothorax, pneumomediastinum) and the need for chest tubes).
Statistical analysis
Maternal and neonatal baseline characteristics and short-term clinical outcomes were compared using Pearson’s Chi-square test for categorical variables and Student’s t-test for continuous variables. Non-parametric data were analyzed using the Wilcoxon’s Rank-Sum test. Univariate and multivariate analyses were conducted separately to assess the impact of the use of NRS on patient mortality and morbidity. Univariate and multivariate logistic regression analysis was performed using surfactant exposure and BPD as outcomes with maternal and neonatal factors as potential predictors. Adjustments were made for the following confounding variables found to be significantly different in a comparison of baseline characteristics: GA, sex, prolonged rupture of membranes, intrapartum antibiotics, maternal diabetes, antenatal steroid use, early caffeine use, Apgar score at 5 min of age, and maternal age (see Table 1). Chorioamnionitis was not adjusted for because of missing data in a large number of participants (approximately one-third). Odds ratios (ORs) with 95% confidence intervals (CIs) for surfactant utilization and BPD in the earlier time period compared with the later time period were generated from these analyses. Further, a subgroup analysis based on GA categories (<26 weeks and 26 to 286/7 weeks) was performed to determine whether the early use of NRS and delayed use of surfactant corresponded with similar trends in BPD across all GA groups. Statistical analyses were conducted using SAS V.9.3 (SAS Institute, Cary, NC, USA), and a P-value of 0.05 was considered significant.
Ethical approval
Data collection was approved by either research ethics boards or hospital quality improvement committees at each participating hospital. Approval for this particular study was obtained from Mount Sinai Hospital’s Research Ethics Board and the Executive Committee of the CNN.
Results
A total of 5579 and 6600 infants<29 weeks GA were admitted to CNN centers between 2005 and 2008 and between 2010 and 2013, respectively. After application of the exclusion criteria, 3980 infants born between 2005 and 2008 and 5137 infants between 2010 and 2013 remained in the analysis (Figure 1).
A comparison of maternal and neonatal characteristics was performed between the two groups (Table 1). Maternal characteristics that appeared to be significantly different overall were the presence of maternal diabetes, the use of antenatal steroids, rupture of membranes >24 h and the use of intrapartum antibiotics, all of which were higher in the later time period. Neonatal factors that were significantly different were GA, male sex, Apgar score at 5 min and early use of caffeine (first 3 days of birth). These characteristics were included in the logistic regression analysis for the overall study population and for subgroup analysis.
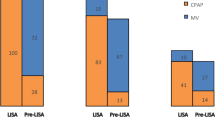
Table 2 shows the utilization of surfactant and ventilation resources during the two time periods. There was no significant difference in overall surfactant utilization between the groups (77.2% in 2005 to 2008 vs 76.1% in 2010 to 2013, P=0.23). However, compared with 2005 to 2008, more neonates received therapeutic surfactant in 2010 to 2013 (56.9% vs 47.1%, P<0.01) and less prophylactic surfactant (43.1% vs 52.9%, P<0.01). In addition, a lower proportion of neonates in the later era required two or more doses of surfactant (35.6% in 2010 to 2013 vs 39.6% in 2005 to 2008, P<0.01). Stratification into subgroups revealed differences in surfactant use for infants <26 weeks and 26 to 286/7 weeks. A higher proportion of infants born <26 weeks GA were administered surfactant in 2010 to 2013 compared with 2005 to 2008 (91.0% vs 85.3%; P <0.01) while the use of surfactant decreased for infants born 26 to 286/7 weeks (68.8% vs 73.6% in 2005 to 2008; P<0.01) (Table 2). In both the GA groups, there was increased utilization of therapeutic surfactant and fewer infants received ⩾2 doses in the later cohort (Table 2). The median days spent on invasive mechanical ventilation were less between 2010 and 2013 (6 vs 9 days in 2005 to 2008, P<0.01), while duration of CPAP increased (Table 2). However, neonates born between 2010 and 2013 appeared to require significantly more days on oxygen (27 vs 7 days between 2005 and 2008, P<0.01; Table 2).
The incidences of short-term outcomes of neonates born in both eras were compared as well (Table 3). Mortality, air leak syndrome, necrotizing enterocolitis, early-onset sepsis and grades 3 to 4 intraventricular hemorrhage were unchanged overall between these time periods, with the exception of mortality in infants <26 weeks GA, which decreased in 2010 to 2013. Despite there being no difference in the rate of air leak or pneumothorax (5.9% in 2005 to 2008 vs 5.7% in 2010 to 2013, P=0.68), babies born in the later era more frequently required chest tube insertion (3.4% vs 1.5% in 2005 to 2008, P<0.01). BPD at 36 weeks PMA, however, was significantly reduced in the more recent group (38.7% in 2010 to 2013 vs 42.0% in 2005 to 2008, P<0.01; Table 3). Nosocomial infections were also lower in the later time period (25.1% in 2010 to 2013 vs 28.6% in 2005 to 2008, P<0.01), although this reduction was statistically significant only for infants 26 to 286/7 weeks. The rate of ROP also decreased over time (13.5% in 2010 to 2013 vs 15.6% in 2005 to 2008; P=0.02). It should be noted that there was a significant amount of missing data on ROP, and therefore, the results should be interpreted with caution.
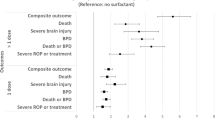
Univariate logistic regression analysis revealed that the odds of surfactant use in 2005 to 2008 compared with 2010 to 2013 was 1.06 (95% CI 0.96, 1.17), indicating no significant difference in overall surfactant use between the two time periods (Table 4). When adjusted for GA, sex, prolonged rupture of membranes, intrapartum antibiotics, maternal diabetes, antenatal steroid use, Apgar score at 5 min of age, early caffeine use and maternal age, the OR remained nonsignificant (adjusted OR (AOR) 1.00, 95% CI 0.89, 1.13). The odds of developing BPD at 36 weeks PMA was significantly higher in the earlier era (AOR 1.19, 95% CI 1.07, 1.33) when adjusted for the same confounding variables. However, in our subgroup analysis, the adjusted odds of surfactant administration in infants <26 and 26 to 286/7 weeks were 0.55 (95% CI 0.42, 0.73) and 1.14 (95% CI 1.00, 1.30), respectively. The odds of developing BPD at 36 weeks PMA was significantly higher in the earlier era (AOR 1.34, 95% CI 1.08, 1.66) for infants <26 weeks GA while a trend was noted for infants 26 to 286/7 weeks (AOR 1.13, 95% CI 0.99, 1.29) when adjusted for the same confounding variables.
Discussion
Prior to 2008, routine use of early NRS in the delivery room was not practiced. An initial Cochrane review of prophylactic vs rescue surfactant showed a lower risk of air leak and neonatal morbidity with prophylactic surfactant. However, when this review was updated to include trials where routine CPAP was used, the advantage of prophylactic surfactant administration was no longer apparent. In fact, routine application of CPAP lowered the risk of chronic lung disease or death.17 As a result, the practice of stabilization on CPAP in the delivery room was adopted in many countries, including Canada. The purpose of this study was to retrospectively analyze the utilization of surfactant in an era where routine CPAP was applied in the delivery room and compare the outcomes of these infants to those who were born before this practice was adopted. To our knowledge, this is the first study to describe the trend in surfactant utilization and outcomes in Canadian neonatal intensive care units since the adoption of early routine CPAP.
Our study shows that, in recent years, surfactant utilization did not appear to have changed overall despite the use of routine early delivery room stabilization on NRS. In fact, further examination revealed that overall surfactant use actually increased in infants <26 weeks GA, whereas usage declined in the 26 to 286/7 subgroup. Nonetheless, the shift towards decreased prophylactic surfactant use and increased therapeutic use was similar in both subgroups. Previous studies have shown that, although some preterm neonates may never need surfactant after early routine CPAP, between 38% and 67% of infants will need intubation and surfactant at some time during their clinical course, depending on the clinical criteria used to determine CPAP failure and need for surfactant.13, 14, 26 Physiologically, although early CPAP may help establish functional residual capacity, reduce work of breathing and improve gas exchange, primary surfactant deficiency is unchanged and thus surfactant therapy would still be required. This is particularly important in preterm neonates born close to the limits of viability who have a more significant surfactant deficiency. In these neonates, it appears that early NRS use may in fact necessitate the use of surfactant more often than in neonates born 26 to 286/7 weeks GA. Previous studies have shown that neonates at the limits of viability are rarely managed with NRS alone.27, 28 However, the use of early NRS is still associated with reductions in BPD and death in this age group14 and is likely still the preferred management choice. Interestingly, although the incidence of air leak or pneumothorax was unchanged in a time when more NRS and less surfactant was used overall, a higher proportion of babies born in the later time period required chest tube insertion. Unfortunately, it is difficult to determine the severity of the air leak syndrome and the indications for chest tube insertion are unknown.
As expected, the timing of surfactant administration did differ between the two time periods with an increased use of rescue surfactant in more recent years. In addition, fewer infants in both GA groups required repeated doses of surfactant in the 2010 to 2013 period. This may be related to the increased use of non-invasive ventilation and the use of higher positive-end expiratory pressures. The infants in the later time period also spent fewer days on mechanical ventilation, which again is in keeping with previous studies.13, 14 The decreased use of invasive mechanical ventilation may in part explain the reductions in BPD, as has been demonstrated in several publications previously.29, 30, 31 In these studies, both early extubation and shorter cumulative time spent on mechanical ventilation reduced the risk of BPD.
The infants in the later time period required more days of oxygen therapy (27 vs 7 days between 2005 and 2008, P<0.01). This was even more significant in infants born <26 weeks GA with a mean of 57 days spent on supplemental oxygen compared with 9 days in 2005 to 2008. This may be a result of earlier extubation practices with inadequate maintenance of functional residual capacity on CPAP or an increase in oxygen saturation targets. Regardless of this increased use of oxygen, there was actually a decreased rate of both ROP and BPD at 36 weeks PMA in 2010 to 2013 compared with 2005 to 2008, indicating that, despite this prolonged time on oxygen, the rates of these outcomes were unchanged. Of note, however, there was a significant amount of missing data on ROP and it is difficult to accurately make this conclusion.
Other short-term neonatal outcomes were also compared between the two time periods. There was no difference in overall mortality, necrotizing enterocolitis, early-onset sepsis and grade 3 to 4 intraventricular hemorrhage between the two time periods. Nosocomial infections appear to be higher in the earlier time period. This may not only be more reflective of evolving quality improvement practices in neonatal intensive care units following Evidence-based Practice for Improving Quality implementation but may also be related to decreased rates of ventilator-associated pneumonia caused by prolonged intubation and mechanical ventilation in the earlier time period.21
In this study, the incidence of BPD was significantly lower in the later era. This decrease was more pronounced among infants <26 weeks GA, while a more subtle decline was observed in infants 26 to 286/7 weeks GA. This was not observed in previous studies comparing prophylactic surfactant with rescue surfactant.13, 14, 17, 26, 32, 33 In this neonatal population, the rate of antenatal glucocorticoids was comparable to earlier studies.13, 14, 26 In addition, neonates appeared to spend more days in oxygen in the later time period, indicating that, despite more oxygen exposure, the risk of developing BPD was lower. However, other factors may have contributed to the decline in BPD. For instance, the use early caffeine significantly increased between the two time periods that have been previously reported to reduce BPD rates.34 The decreased rate of nosocomial infections from the initial time period to 2010 to 2013 may have also contributed to the decrease in BPD, especially in the older GA group, and the association between sepsis and subsequent development of BPD has been suggested in previous studies.35, 36 Finally, improved ventilation strategies between 2008 and 2010 may have contributed to a lower rate of BPD. This may include earlier extubation and less use of invasive mechanical ventilation; use of non-invasive ventilation techniques, such as non-invasive positive-pressure ventilation or non-invasive high-frequency ventilation; or the use of alternative mechanical ventilation modes, such as high-frequency oscillation or high-frequency jet ventilation. Although chorioamnionitis may contribute to the development of BPD,37 it was not adjusted for because of missing data. However, the rate of chorioamnionitis was higher in the later time period, and therefore, the decrease in BPD may actually be underestimated from this analysis.
The limitations of this study are that it is an observational study with data collected retrospectively. It is difficult to determine indications for certain procedures, such as chest tube insertion, as well as to determine whether variations in outcomes were in fact attributable to the implementation of early CPAP and rescue surfactant therapy. Information on other variables, such as oxygen saturation targets, type of NRS, type of surfactant or the criteria or technique used to administer surfactant, were not collected that may have important implications for the reported outcomes.
In summary, in an era where routine early CPAP is used with rescue surfactant, although surfactant utilization remains unchanged, the timing of surfactant has shifted to later rescue use. A lower incidence of BPD is also noted in this era, although other factors such as a high rate of antenatal steroid use, lower rates of nosocomial infection, improved ventilation strategies and early caffeine use may have also contributed to this improvement. Early CPAP with a strategy for rescue surfactant has been suggested as the optimal means of treating neonatal RDS while improving short-term outcomes, but the ideal timing for surfactant remains to be determined and may need to be individualized. In addition, based on recent data, it may be better to deliver surfactant less invasively through a catheter rather than through the endotracheal tube.8 This new delivery technique may alter the timing of surfactant and further improve short-term outcomes by avoiding intubation altogether.
Conclusions
Although it appears that early routine use of non-invasive ventilation has not led to alterations in overall surfactant use in very preterm infants, both the timing of surfactant administration and the overall use in specified GA subgroups (<26 and 26 to 280/7 weeks) has changed between the two time periods. In addition, there was an observed reduction in BPD in later years. Further studies are required to accurately determine whether the underlying cause of this reduction in BPD is related to the delayed use of invasive mechanical ventilation, early non-invasive ventilation use, changes in surfactant practices or a combination of these factors.
References
Collaborative European Multicenter Study Group. Surfactant replacement therapy for severe neonatal respiratory distress syndrome: an international randomized clinical trial. Pediatrics 1988; 82: 683–691.
Speer CP, Robertson B, Curstedt T, Halliday HL, Compagnone D, Gefeller O et al. Randomized European multicenter trial of surfactant replacement therapy for severe neonatal respiratory distress syndrome: single versus multiple doses of Curosurf. Pediatrics 1992; 89: 13–20.
Seger N, Soll R . Animal derived surfactant extract for treatment of respiratory distress syndrome. Cochrane Database Syst Rev 2009; (2): CD007836.
Soll RF . Synthetic surfactant for respiratory distress syndrome in preterm infants. Cochrane Database Syst Rev 2000; (2): CD001149.
Soll R, Ozek E . Prophylactic protein free synthetic surfactant for preventing morbidity and mortality in preterm infants. Cochrane Database Syst Rev 2010; (1): CD001079.
Soll RF . Prophylactic natural surfactant extract for preventing morbidity and mortality in preterm infants. Cochrane Database Syst Rev 2000; (2): CD000511.
Hentschel R, Jorch G . Acute side effects of surfactant treatment. J Perinat Med 2002; 30: 143–148.
Isayama T, Iwami H, McDonald S, Beyene J . Association of noninvasive ventilation strategies with mortality and bronchopulmonary dysplasia among preterm infants: a systematic review and meta-analysis. JAMA 2016; 316: 611–624.
Mahmoud RA, Roehr CC, Schmalisch G . Current methods of non-invasive ventilatory support for neonates. Paediatr Respir Rev 2011; 12: 196–205.
Bhandari V . The potential of non-invasive ventilation to decrease BPD. Semin Perinatol 2013; 37: 108–114.
Patel D, Greenough A . Does nasal CPAP reduce bronchopulmonary dysplasia (BPD)? Acta Paediatr 2008; 97: 1314–1317.
Hascoet JM, Espagne S, Hamon I . CPAP and the preterm infant: lessons from the COIN trial and other studies. Early Hum Dev 2008; 84: 791–793.
Morley CJ, Davis PG, Doyle LW, Brion LP, Hascoet JM, Carlin JB et al. Nasal CPAP or intubation at birth for very preterm infants. N Engl J Med 2008; 358: 700–708.
Finer NN, Carlo WA, Walsh MC, Rich W, Gantz MG, Laptook AR et al. Early CPAP versus surfactant in extremely preterm infants. N Engl J Med 2010; 362: 1970–1979.
Fischer HS, Bührer C . Avoiding endotracheal ventilation to prevent bronchopulmonary dysplasia: a meta-analysis. Pediatrics 2013; 132: e1351–e1360.
Schmidt B, Asztalos EV, Roberts RS, Robertson CM, Sauve RS, Whitfield MF et al. Impact of bronchopulmonary dysplasia, brain injury, and severe retinopathy on the outcome of extremely low-birth-weight infants at 18 months: results from the trial of indomethacin prophylaxis in preterms. JAMA 2003; 289: 1124–1129.
Rojas-Reyes MX, Morley CJ, Soll R . Prophylactic versus selective use of surfactant in preventing morbidity and mortality in preterm infants. Cochrane Database Syst Rev 2012; (3): CD000510.
Bahadue FL, Soll R . Early versus delayed selective surfactant treatment for neonatal respiratory distress syndrome. Cochrane Database Syst Rev 2012; 11: CD001456.
Isayama T, Chai-Adisaksopha C, McDonald SD . Noninvasive ventilation with vs without early surfactant to prevent chronic lung disease in preterm infants: a systematic review and meta-analysis. JAMA Pediatr 2015; 169: 731–739.
Blennow M, Bohlin K . Surfactant and noninvasive ventilation. Neonatology 2015; 107: 330–336.
Lee SK, Aziz K, Singhal N, Cronin CM, James A, Lee DS et al. Improving the quality of care for infants: a cluster randomized controlled trial. CMAJ 2009; 181: 469–476.
Shennan AT, Dunn MS, Ohlsson A, Lennox K, Hoskins EM . Abnormal pulmonary outcomes in premature infants: prediction from oxygen requirement in the neonatal period. Pediatrics 1988; 82: 527–532.
International Committee for the Classification of Retinopahty of Prematurity. The International Classification of Retinopathy of Prematurity revisited. Arch Ophthalmol 2005; 123: 991–999.
Bell MJ, Ternberg JL, Feigin RD, Keating JP, Marshall R, Barton L et al. Neonatal necrotizing enterocolitis. Therapeutic decisions based upon clinical staging. Ann Surg 1978; 187: 1–7.
Papile LA, Burstein J, Burstein R, Koffler H . Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1,500 gm. J Pediatr 1978; 92: 529–534.
Dunn MS, Kaempf J, de Klerk A, de Klerk R, Reilly M, Howard D et al. Randomized trial comparing 3 approaches to the initial respiratory management of preterm neonates. Pediatrics 2011; 128: e1069–e1076.
Mehler K, Grimme J, Abele J, Huenseler C, Roth B, Kribs A . Outcome of extremely low gestational age newborns after introduction of a revised protocol to assist preterm infants in their transition to extrauterine life. Acta Paediatr 2012; 101: 1232–1239.
Aly H, Massaro AN, Patel K, El-Mohandes AA . Is it safer to intubate premature infants in the delivery room? Pediatrics 2005; 115: 1660–1665.
Jensen EA, DeMauro SB, Kornhauser M, Aghai ZH, Greenspan JS, Dysart KC . Effects of multiple ventilation courses and duration of mechanical ventilation on respiratory outcomes in extremely low-birth-weight infants. JAMA Pediatr 2015; 169: 1011–1017.
Robbins M, Trittmann J, Martin E, Reber KM, Nelin L, Shepherd E . Early extubation attempts reduce length of stay in extremely preterm infants even if re-intubation is necessary. J Neonatal Perinatal Med 2015; 8: 91–97.
Berger J, Mehta P, Bucholz E, Dziura J, Bhandari V . Impact of early extubation and reintubation on the incidence of bronchopulmonary dysplasia in neonates. Am J Perinatol 2014; 31: 1063–1072.
Rojas MA, Lozano JM, Rojas MX, Laughon M, Bose CL, Rondon MA et al. Very early surfactant without mandatory ventilation in premature infants treated with early continuous positive airway pressure: a randomized, controlled trial. Pediatrics 2009; 123: 137–142.
Sandri F, Plavka R, Ancora G, Simeoni U, Stranak Z, Martinelli S et al. Prophylactic or early selective surfactant combined with nCPAP in very preterm infants. Pediatrics 2010; 125: e1402–e1409.
Schmidt B, Roberts RS, Davis P, Doyle LW, Barrington KJ, Ohlsson A et al. Caffeine therapy for apnea of prematurity. N Engl J Med 2006; 354: 2112–2121.
Ballard AR, Mallett LH, Pruszynski JE, Cantey JB . Chorioamnionitis and subsequent bronchopulmonary dysplasia in very-low-birth weight infants: a 25-year cohort. J Perinatol 2016; 36: 1045–1048.
Shah J, Jefferies AL, Yoon EW, Lee SK, Shah PS Canadian Neonatal Network. Risk factors and outcomes of late-onset bacterial sepsis in preterm neonates born at <32 weeks' gestation. Am J Perinatol 2015; 32: 675–682.
Hartling L, Liang Y, Lacaze-Masmonteil T . Chorioamnionitis as a risk factor for bronchopulmonary dysplasia: a systematic review and meta-analysis. Arch Dis Child Fetal Neonatal Ed 2012; 97: F8–F17.
Acknowledgements
We thank all the site investigators of the Canadian Neonatal Network (CNN). We also thank the data abstractors of the CNN, as well as the staff at the Maternal-Infant Care Research Centre at Mount Sinai Hospital, Toronto, ON, Canada for providing organizational and statistical support for this project. Although no specific funding has been received for this study, organizational support for the Canadian Neonatal Network was provided by the Maternal-Infant Care Research Centre (MiCare) at Mount Sinai Hospital in Toronto, ON, Canada. MiCare is supported by a Canadian Institutes of Health Research (CIHR) Team Grant (FRN87518).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Canadian Neonatal Network Investigators
Prakesh S Shah, MD, MSc (Director, Canadian Neonatal Network and site investigator), Mount Sinai Hospital, Toronto, ON, Canada; Adele Harrison, MD, MBChB, Victoria General Hospital, Victoria, BC, Canada; Anne Synnes, MDCM, MHSC, and Joseph Ting, MD, B.C. Women’s Hospital and Health Centre, Vancouver, BC, Canada; Zenon Cieslak, MD, Royal Columbian Hospital, New Westminster, BC, Canada; Rebecca Sherlock, MD, Surrey Memorial Hospital, Surrey, BC, Canada; Wendy Yee, MD, Foothills Medical Centre, Calgary, AB, Canada; Khalid Aziz, MBBS, MA, MEd, and Jennifer Toye, MD, Royal Alexandra Hospital, Edmonton, AB, Canada; Carlos Fajardo, MD, Alberta Children’s Hospital, Calgary, AB, Canada; Zarin Kalapesi, MD, Regina General Hospital, Regina, SK, Canada; Koravangattu Sankaran, MD, MBBS, and Sibasis Daspal, MD, Royal University Hospital, Saskatoon, SK, Canada; Mary Seshia, MBChB, Winnipeg Health Sciences Centre, Winnipeg, MB, Canada; Ruben Alvaro, MD, St. Boniface General Hospital, Winnipeg, MB, Canada; Amit Mukerji, MD, Hamilton Health Sciences Centre, Hamilton, ON, Canada; Orlando Da Silva, MD, MSc, London Health Sciences Centre, London, ON, Canada; Chuks Nwaesei, MD, Windsor Regional Hospital, Windsor, ON, Canada; Kyong-Soon Lee, MD, MSc, Hospital for Sick Children, Toronto, ON, Canada; Michael Dunn, MD, Sunnybrook Health Sciences Centre, Toronto, ON, Canada; Brigitte Lemyre, MD, Children’s Hospital of Eastern Ontario and Ottawa General Hospital, Ottawa, ON, Canada; Kimberly Dow, MD, Kingston General Hospital, Kingston, ON, Canada; Ermelinda Pelausa, MD, Jewish General Hospital, Montréal, QC, Canada; Keith Barrington, MBChB, Hôpital Sainte-Justine, Montréal, QC, Canada; Christine Drolet, MD, and Bruno Piedboeuf, MD, Centre Hospitalier Universitaire de Québec, Sainte Foy, QC, Canada; Martine Claveau, NNP, and Marc Beltempo, MD, McGill University Health Centre, Montréal, QC, Canada; Valerie Bertelle, MD, and Edith Masse, MD, Centre Hospitalier Universitaire de Sherbrooke, Sherbrooke, QC, Canada; Roderick Canning, MD, Moncton Hospital, Moncton, NB, Canada; Hala Makary, MD, Dr Everett Chalmers Hospital, Fredericton, NB, Canada; Cecil Ojah, MBBS, and Luis Monterrosa, MD, Saint John Regional Hospital, Saint John, NB, Canada; Akhil Deshpandey, MBBS, MRCPI, Janeway Children’s Health and Rehabilitation Centre, St John’s, NL, Canada; Jehier Afifi, MB BCh, MSc, IWK Health Centre, Halifax, NS, Canada; Andrzej Kajetanowicz, MD, Cape Breton Regional Hospital, Sydney, NS, Canada; Shoo K Lee, MBBS, PhD (Chairman, Canadian Neonatal Network), Mount Sinai Hospital, Toronto, ON, Canada.
Rights and permissions
About this article
Cite this article
Raghuram, K., Mukerji, A., Young, J. et al. Surfactant utilization and short-term outcomes in an era of non-invasive respiratory support in Canadian neonatal intensive care units. J Perinatol 37, 1017–1023 (2017). https://doi.org/10.1038/jp.2017.98
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2017.98