Abstract
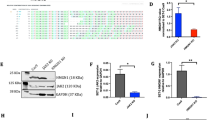
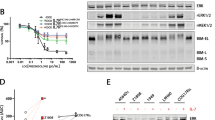
MLL-rearranged acute lymphoblastic leukemia (ALL) in infants is characterized by a poor clinical outcome and resistance to glucocorticoids (for example, prednisone and dexamethasone). As both the response to prednisolone in vitro and prednisone in vivo are predictive for clinical outcome, understanding and overcoming glucocorticoid resistance remains an essential step towards improving prognosis. Prednisolone-induced apoptosis depends on glucocorticoid-evoked Ca2+ fluxes from the endoplasmic reticulum towards the mitochondria. Here, we demonstrate that in MLL-rearranged infant ALL, over-expression of S100A8 and S100A9 is associated with failure to induce free-cytosolic Ca2+ and prednisolone resistance. Furthermore, we demonstrate that enforced expression of S100A8/S100A9 in prednisolone-sensitive MLL-rearranged ALL cells, rapidly leads to prednisolone resistance as a result of S100A8/S100A9 mediated suppression of prednisolone-induced free-cytosolic Ca2+ levels. In addition, the Src kinase inhibitor PP2 markedly sensitized MLL-rearranged ALL cells otherwise resistant to prednisolone, via downregulation of S100A8 and S100A9, which allowed prednisolone-induced Ca2+ fluxes to reach the mitochondria and trigger apoptosis. On the basis of this novel mechanism of prednisolone resistance, we propose that developing more specific S100A8/S100A9 inhibitors may well be beneficial for prednisolone-resistant MLL-rearranged infant ALL patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
References
Pui CH, Relling MV, Downing JR . Acute lymphoblastic leukemia. N Engl J Med 2004; 350: 1535–1548.
Greaves MF . Infant leukaemia biology, aetiology and treatment. Leukemia 1996; 10: 372–377.
Pieters R, Schrappe M, De Lorenzo P, Hann I, De Rossi G, Felice M et al. A treatment protocol for infants younger than 1 year with acute lymphoblastic leukaemia (Interfant-99): an observational study and a multicentre randomised trial. Lancet 2007; 370: 240–250.
Pieters R, den Boer ML, Durian M, Janka G, Schmiegelow K, Kaspers GJ et al. Relation between age, immunophenotype and in vitro drug resistance in 395 children with acute lymphoblastic leukemia--implications for treatment of infants. Leukemia 1998; 12: 1344–1348.
Riehm H, Reiter A, Schrappe M, Berthold F, Dopfer R, Gerein V et al. Corticosteroid-dependent reduction of leukocyte count in blood as a prognostic factor in acute lymphoblastic leukemia in childhood (therapy study ALL-BFM 83). Klin Padiatr 1987; 199: 151–160.
Dordelmann M, Reiter A, Borkhardt A, Ludwig WD, Gotz N, Viehmann S et al. Prednisone response is the strongest predictor of treatment outcome in infant acute lymphoblastic leukemia. Blood 1999; 94: 1209–1217.
Den Boer ML, Harms DO, Pieters R, Kazemier KM, Gobel U, Korholz D et al. Patient stratification based on prednisolone-vincristine-asparaginase resistance profiles in children with acute lymphoblastic leukemia. J Clin Oncol 2003; 21: 3262–3268.
Kaspers GJ, Pieters R, Van Zantwijk CH, Van Wering ER, Van Der Does-Van Den Berg A, Veerman AJ . Prednisolone resistance in childhood acute lymphoblastic leukemia: vitro-vivo correlations and cross-resistance to other drugs. Blood 1998; 92: 259–266.
Stam RW, Den Boer ML, Schneider P, de Boer J, Hagelstein J, Valsecchi MG et al. Association of high-level MCL-1 expression with in vitro and in vivo prednisone resistance in MLL-rearranged infant acute lymphoblastic leukemia. Blood 2010; 115: 1018–1025.
Wei G, Twomey D, Lamb J, Schlis K, Agarwal J, Stam RW et al. Gene expression-based chemical genomics identifies rapamycin as a modulator of MCL1 and glucocorticoid resistance. Cancer Cell 2006; 10: 331–342.
Stam RW, Schneider P, Hagelstein JA, van der Linden MH, Stumpel DJ, de Menezes RX et al. Gene expression profiling-based dissection of MLL translocated and MLL germline acute lymphoblastic leukemia in infants. Blood 2010; 115: 2835–2844.
Qazi S, Uckun FM . Gene expression profiles of infant acute lymphoblastic leukaemia and its prognostically distinct subsets. Br J Haematol 2010; 149: 865–873.
Edgeworth J, Gorman M, Bennett R, Freemont P, Hogg N . Identification of p8,14 as a highly abundant heterodimeric calcium binding protein complex of myeloid cells. J Biol Chem 1991; 266: 7706–7713.
Strupat K, Rogniaux H, Van Dorsselaer A, Roth J, Vogl T . Calcium-induced noncovalently linked tetramers of MRP8 and MRP14 are confirmed by electrospray ionization-mass analysis. J Am Soc Mass Spectrom 2000; 11: 780–788.
Teigelkamp S, Bhardwaj RS, Roth J, Meinardus-Hager G, Karas M, Sorg C . Calcium-dependent complex assembly of the myeloic differentiation proteins MRP-8 and MRP-14. J Biol Chem 1991; 266: 13462–13467.
Kawasaki H, Nakayama S, Kretsinger RH . Classification and evolution of EF-hand proteins. Biometals 1998; 11: 277–295.
Carafoli E . Calcium signaling: a tale for all seasons. Proc Natl Acad Sci U S A 2002; 99: 1115–1122.
Kaiser N, Edelman IS . Calcium dependence of glucocorticoid-induced lymphocytolysis. Proc Natl Acad Sci USA 1977; 74: 638–642.
Kaspers GJ, Veerman AJ, Pieters R, Broekema GJ, Huismans DR, Kazemier KM et al. Mononuclear cells contaminating acute lymphoblastic leukaemic samples tested for cellular drug resistance using the methyl-thiazol-tetrazolium assay. Br J Cancer 1994; 70: 1047–1052.
Pieters R, Loonen AH, Huismans DR, Broekema GJ, Dirven MW, Heyenbrok MW et al. In vitro drug sensitivity of cells from children with leukemia using the MTT assay with improved culture conditions. Blood 1990; 76: 2327–2336.
Stam RW, Schneider P, de Lorenzo P, Valsecchi MG, den Boer ML, Pieters R . Prognostic significance of high-level FLT3 expression in MLL-rearranged infant acute lymphoblastic leukemia. Blood 2007; 110: 2774–2775.
Huber W, von Heydebreck A, Sultmann H, Poustka A, Vingron M . Variance stabilization applied to microarray data calibration and to the quantification of differential expression. Bioinformatics 2002; 18 (Suppl 1): S96–104.
Smyth GK . Limma: linear models for microarray data. In: Gentleman R, Carey V, Dudoit S, Irizarry R, Huber W (eds). Bioinformatics and Computational Biology Solutions Using R and Bioconductor. Springer: New York, 2005, pp397–420.
Wettenhall JM, Smyth GK . limmaGUI: a graphical user interface for linear modeling of microarray data. Bioinformatics 2004; 20: 3705–3706.
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci U S A 2005; 102: 15545–15550.
Stam RW, den Boer ML, Meijerink JP, Ebus ME, Peters GJ, Noordhuis P et al. Differential mRNA expression of Ara-C-metabolizing enzymes explains Ara-C sensitivity in MLL gene-rearranged infant acute lymphoblastic leukemia. Blood 2003; 101: 1270–1276.
Krivtsov AV, Feng Z, Lemieux ME, Faber J, Vempati S, Sinha AU et al. H3K79 methylation profiles define murine and human MLL-AF4 leukemias. Cancer Cell 2008; 14: 355–368.
Stam RW, den Boer ML, Passier MM, Janka-Schaub GE, Sallan SE, Armstrong SA et al. Silencing of the tumor suppressor gene FHIT is highly characteristic for MLL gene rearranged infant acute lymphoblastic leukemia. Leukemia 2006; 20: 264–271.
Arai K, Takano S, Teratani T, Ito Y, Yamada T, Nozawa R . S100A8 and S100A9 overexpression is associated with poor pathological parameters in invasive ductal carcinoma of the breast. Curr Cancer Drug Targets 2008; 8: 243–252.
Nicolas E, Ramus C, Berthier S, Arlotto M, Bouamrani A, Lefebvre C et al. Expression of S100A8 in leukemic cells predicts poor survival in de novo AML patients. Leukemia 2011; 25: 57–65.
Damm F, Thol F, Hollink I, Zimmermann M, Reinhardt K, van den Heuvel-Eibrink MM et al. Prevalence and prognostic value of IDH1 and IDH2 mutations in childhood AML: a study of the AML-BFM and DCOG study groups. Leukemia 2011; 25: 1704–1710.
Bootman MD, Collins TJ, Peppiatt CM, Prothero LS, MacKenzie L, De Smet P et al. Calcium signalling--an overview. Semin Cell Dev Biol 2001; 12: 3–10.
Distelhorst CW, Dubyak G . Role of calcium in glucocorticosteroid-induced apoptosis of thymocytes and lymphoma cells: resurrection of old theories by new findings. Blood 1998; 91: 731–734.
Rizzuto R, Pinton P, Ferrari D, Chami M, Szabadkai G, Magalhaes PJ et al. Calcium and apoptosis: facts and hypotheses. Oncogene 2003; 22: 8619–8627.
Susin SA, Zamzami N, Castedo M, Hirsch T, Marchetti P, Macho A et al. Bcl-2 inhibits the mitochondrial release of an apoptogenic protease. J Exp Med 1996; 184: 1331–1341.
Duchen MR . Mitochondria and calcium: from cell signalling to cell death. J Physiol 2000; 529 (Pt 1): 57–68.
Smaili SS, Hsu YT, Youle RJ, Russell JT . Mitochondria in Ca2+ signaling and apoptosis. J Bioenerg Biomembr 2000; 32: 35–46.
Holleman A, den Boer ML, Kazemier KM, Janka-Schaub GE, Pieters R . Resistance to different classes of drugs is associated with impaired apoptosis in childhood acute lymphoblastic leukemia. Blood 2003; 102: 4541–4546.
Ryckman C, Gilbert C, de Medicis R, Lussier A, Vandal K, Tessier PA . Monosodium urate monohydrate crystals induce the release of the proinflammatory protein S100A8/A9 from neutrophils. J Leukoc Biol 2004; 76: 433–440.
McConkey DJ, Nicotera P, Hartzell P, Bellomo G, Wyllie AH, Orrenius S . Glucocorticoids activate a suicide process in thymocytes through an elevation of cytosolic Ca2+ concentration. Arch Biochem Biophys 1989; 269: 365–370.
Minagawa N, Kruglov EA, Dranoff JA, Robert ME, Gores GJ, Nathanson MH . The anti-apoptotic protein Mcl-1 inhibits mitochondrial Ca2+ signals. J Biol Chem 2005; 280: 33637–33644.
Vrana JA, Bieszczad CK, Cleaveland ES, Ma Y, Park JP, Mohandas TK et al. An MCL1-overexpressing Burkitt lymphoma subline exhibits enhanced survival on exposure to serum deprivation, topoisomerase inhibitors, or staurosporine but remains sensitive to 1–beta-D-arabinofuranosylcytosine. Cancer Res 2002; 62: 892–900.
Srikrishna G . S100A8 and S100A9: New Insights into Their Roles in Malignancy. J Innate Immun 2011; 4: 31–40.
Kim LC, Song, L, Haura EB . Src kinases as therapeutic targets for cancer. Nat Rev Clin Oncol 2009; 6: 587–595.
Chen M, Sastry SK, O'Connor KL . Src kinase pathway is involved in NFAT5-mediated S100A4 induction by hyperosmotic stress in colon cancer cells. Am J Physiol Cell Physiol 2011; 300: C1155–C1163.
Acknowledgements
The authors wish to express gratitude to the members and participating hospitals of the INTERFANT-99 for supporting this study by providing leukemic samples. Members of INTERFANT-99 are: Campbell, M (PINDA), Felice, M (Argentina), Ferster, A (CLCG), Hann, I and Vora, A (UKCCSG), Hovi, L (NOPHO), Janka-Schaub, G (COALL), Li, CK (Hong Kong), Mann, G (BFM-A), Mechinaud, F (Fralle), Pieters, R (DCOG), de Rossi, G and Biondi, A (AIEOP), Rubnitz J (SJCRH), Schrappe, M (BFM-G), Silverman, L (DFCI), Stary, J (CPH), Suppiah, R (ANZCHOG), Szczepanski, T (PPLLSG), Valscecchi, M and de Lorzenzo, P (CORS). This study was financially supported by KIKA (Stichting Kinderen Kankervrij). The authors have no conflict of interest to disclose. Further financial support was provided by the Dutch Cancer Society (grant EMCR 2005–3662; RWS).
AUTHOR CONTRIBUTIONS
JAP Spijkers-Hagelstein designed and performed research, wrote manuscript; P Schneider, E Hulleman and J de Boer, performed research; O Williams performed research, and reviewed manuscript; R Pieters designed research, wrote and reviewed manuscript; RW Stam designed research, wrote and reviewed manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Spijkers-Hagelstein, J., Schneider, P., Hulleman, E. et al. Elevated S100A8/S100A9 expression causes glucocorticoid resistance in MLL-rearranged infant acute lymphoblastic leukemia. Leukemia 26, 1255–1265 (2012). https://doi.org/10.1038/leu.2011.388
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2011.388
Keywords
This article is cited by
-
S100A8 inhibition in leukemic lymphoblasts induces sensitivity to chemotherapy and inhibition of disease relapse
Medical Oncology (2022)
-
Integrative genomic analyses reveal mechanisms of glucocorticoid resistance in acute lymphoblastic leukemia
Nature Cancer (2020)
-
Differential network analysis and protein-protein interaction study reveals active protein modules in glucocorticoid resistance for infant acute lymphoblastic leukemia
Molecular Medicine (2019)
-
Elevated expression of S100A8 and S100A9 correlates with resistance to the BCL-2 inhibitor venetoclax in AML
Leukemia (2019)
-
Chemotactic effect of S100A8 and S100A9 on human eosinophilic leukemia cells, EoL-1 through TLR4
Molecular & Cellular Toxicology (2018)