Abstract
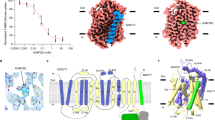
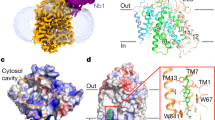
Glucose transporters are essential for metabolism of glucose in cells of diverse organisms from microbes to humans, exemplified by the disease-related human proteins GLUT1, 2, 3 and 4. Despite rigorous efforts, the structural information for GLUT1–4 or their homologues remains largely unknown. Here we report three related crystal structures of XylE, an Escherichia coli homologue of GLUT1–4, in complex with d-xylose, d-glucose and 6-bromo-6-deoxy-d-glucose, at resolutions of 2.8, 2.9 and 2.6 Å, respectively. The structure consists of a typical major facilitator superfamily fold of 12 transmembrane segments and a unique intracellular four-helix domain. XylE was captured in an outward-facing, partly occluded conformation. Most of the important amino acids responsible for recognition of d-xylose or d-glucose are invariant in GLUT1–4, suggesting functional and mechanistic conservations. Structure-based modelling of GLUT1–4 allows mapping and interpretation of disease-related mutations. The structural and biochemical information reported here constitutes an important framework for mechanistic understanding of glucose transporters and sugar porters in general.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Thorens, B. & Mueckler, M. Glucose transporters in the 21st Century. Am. J. Physiol. Endocrinol. Metab. 298, E141–E145 (2010)
Pascual, J. M. et al. GLUT1 deficiency and other glucose transporter diseases. Eur. J. Endocrinol. 150, 627–633 (2004)
Scheepers, A., Joost, H. G. & Schurmann, A. The glucose transporter families SGLT and GLUT: molecular basis of normal and aberrant function. JPEN J. Parenter. Enteral Nutr. 28, 364–371 (2004)
Faham, S. et al. The crystal structure of a sodium galactose transporter reveals mechanistic insights into Na+/sugar symport. Science 321, 810–814 (2008)
Mueckler, M. Facilitative glucose transporters. Eur. J. Biochem. 219, 713–725 (1994)
Mueckler, M. et al. Sequence and structure of a human glucose transporter. Science 229, 941–945 (1985)
Klepper, J. Glucose transporter deficiency syndrome (GLUT1DS) and the ketogenic diet. Epilepsia 49 (Suppl 8). 46–49 (2008)
Brockmann, K. The expanding phenotype of GLUT1-deficiency syndrome. Brain Dev. 31, 545–552 (2009)
Scheffer, I. E. GLUT1 deficiency: a glut of epilepsy phenotypes. Neurology 78, 524–525 (2012)
Santer, R. et al. Mutations in GLUT2, the gene for the liver-type glucose transporter, in patients with Fanconi–Bickel syndrome. Nature Genet. 17, 324–326 (1997)
Leturque, A., Brot-Laroche, E. & Le Gall, M. GLUT2 mutations, translocation, and receptor function in diet sugar managing. Am. J. Physiol. Endocrinol. Metab. 296, E985–E992 (2009)
Simpson, I. A. et al. The facilitative glucose transporter GLUT3: 20 years of distinction. Am. J. Physiol. Endocrinol. Metab. 295, E242–E253 (2008)
Amann, T. & Hellerbrand, C. GLUT1 as a therapeutic target in hepatocellular carcinoma. Expert Opin. Ther. Targets 13, 1411–1427 (2009)
Macheda, M. L., Rogers, S. & Best, J. D. Molecular and cellular regulation of glucose transporter (GLUT) proteins in cancer. J. Cell. Physiol. 202, 654–662 (2005)
Mueckler, M. The molecular biology of glucose transport: relevance to insulin resistance and non-insulin-dependent diabetes mellitus. J. Diabetes Complications 7, 130–141 (1993)
Leney, S. E. & Tavare, J. M. The molecular basis of insulin-stimulated glucose uptake: signalling, trafficking and potential drug targets. J. Endocrinol. 203, 1–18 (2009)
Nelson, D. L. & Cox, M. M. Lehninger Principles of Biochemistry 1158 (W. H. Freeman, 2008)
Pao, S. S., Paulsen, I. T. & Saier, M. H., Jr Major facilitator superfamily. Microbiol. Mol. Biol. Rev. 62, 1–34 (1998)
Henderson, P. J. & Maiden, M. C. Homologous sugar transport proteins in Escherichia coli and their relatives in both prokaryotes and eukaryotes. Phil. Trans. R. Soc. Lond. B 326, 391–410 (1990)
Ozcan, S. & Johnston, M. Function and regulation of yeast hexose transporters. Microbiol. Mol. Biol. Rev. 63, 554–569 (1999)
Buttner, M. The monosaccharide transporter(-like) gene family in Arabidopsis. FEBS Lett. 581, 2318–2324 (2007)
Li, F. et al. Characterization of sucrose transporter alleles and their association with seed yield-related traits in Brassica napus L. BMC Plant Biol. 11, 168 (2011)
Mueckler, M. & Makepeace, C. Model of the exofacial substrate-binding site and helical folding of the human Glut1 glucose transporter based on scanning mutagenesis. Biochemistry 48, 5934–5942 (2009)
Abramson, J. et al. Structure and mechanism of the lactose permease of Escherichia coli. Science 301, 610–615 (2003)
Huang, Y., Lemieux, M. J., Song, J., Auer, M. & Wang, D. N. Structure and mechanism of the glycerol-3-phosphate transporter from Escherichia coli. Science 301, 616–620 (2003)
Yin, Y., He, X., Szewczyk, P., Nguyen, T. & Chang, G. Structure of the multidrug transporter EmrD from Escherichia coli. Science 312, 741–744 (2006)
Dang, S. et al. Structure of a fucose transporter in an outward-open conformation. Nature 467, 734–738 (2010)
Newstead, S. et al. Crystal structure of a prokaryotic homologue of the mammalian oligopeptide–proton symporters, PepT1 and PepT2. EMBO J. 30, 417–426 (2011)
Lam, V. M., Daruwalla, K. R., Henderson, P. J. & Jones-Mortimer, M. C. Proton-linked d-xylose transport in Escherichia coli. J. Bacteriol. 143, 396–402 (1980)
Davis, E. O., Jones-Mortimer, M. C. & Henderson, P. J. Location of a structural gene for xylose–H+ symport at 91 min on the linkage map of Escherichia coli K12. J. Biol. Chem. 259, 1520–1525 (1984)
Davis, E. O. & Henderson, P. J. The cloning and DNA sequence of the gene xylE for xylose–proton symport in Escherichia coli K12. J. Biol. Chem. 262, 13928–13932 (1987)
Maiden, M. C., Davis, E. O., Baldwin, S. A., Moore, D. C. & Henderson, P. J. Mammalian and bacterial sugar transport proteins are homologous. Nature 325, 641–643 (1987)
Henderson, P. J. Proton-linked sugar transport systems in bacteria. J. Bioenerg. Biomembr. 22, 525–569 (1990)
Screpanti, E. & Hunte, C. Discontinuous membrane helices in transport proteins and their correlation with function. J. Struct. Biol. 159, 261–267 (2007)
Snider, C. & White, S. Membrane Proteins of Known 3D Structure (Stephen H. White Lab., Univ. of California Irvine, 2011)
Lu, F. et al. Structure and mechanism of the uracil transporter UraA. Nature 472, 243–246 (2011)
Ressl, S., Terwisscha van Scheltinga, A. C., Vonrhein, C., Ott, V. & Ziegler, C. Molecular basis of transport and regulation in the Na+/betaine symporter BetP. Nature 458, 47–52 (2009)
Gao, X. et al. Structure and mechanism of an amino acid antiporter. Science 324, 1565–1568 (2009)
Fang, Y. et al. Structure of a prokaryotic virtual proton pump at 3.2 Å resolution. Nature 460, 1040–1043 (2009)
Gao, X. et al. Mechanism of substrate recognition and transport by an amino acid antiporter. Nature 463, 828–832 (2010)
Desai, T. A. & Rao, C. V. Regulation of arabinose and xylose metabolism in Escherichia coli. Appl. Environ. Microbiol. 76, 1524–1532 (2010)
Xiang, Z., Steinbach, P. J., Jacobson, M. P., Friesner, R. A. & Honig, B. Prediction of side-chain conformations on protein surfaces. Proteins 66, 814–823 (2007)
Wang, D. et al. Glut-1 deficiency syndrome: clinical, genetic, and therapeutic aspects. Ann. Neurol. 57, 111–118 (2005)
Cunningham, P., Afzal-Ahmed, I. & Naftalin, R. J. Docking studies show that d-glucose and quercetin slide through the transporter GLUT1. J. Biol. Chem. 281, 5797–5803 (2006)
Brockmann, K. et al. Autosomal dominant Glut-1 deficiency syndrome and familial epilepsy. Ann. Neurol. 50, 476–485 (2001)
Ho, Y. Y. et al. Glucose transporter type 1 deficiency syndrome (Glut1DS): methylxanthines potentiate GLUT1 haploinsufficiency in vitro. Pediatr. Res. 50, 254–260 (2001)
Franco, P. J. & Brooker, R. J. Functional roles of Glu-269 and Glu-325 within the lactose permease of Escherichia coli. J. Biol. Chem. 269, 7379–7386 (1994)
Guan, L. & Kaback, H. R. Lessons from lactose permease. Annu. Rev. Biophys. Biomol. Struct. 35, 67–91 (2006)
Schneider, T. R. & Sheldrick, G. M. Substructure solution with SHELXD. Acta Crystallogr. D 58, 1772–1779 (2002)
DeLano, W. L. The PyMOL Molecular Graphics System. http://www.pymol.org (2002)
Saier, M. H., Jr, Yen, M. R., Noto, K., Tamang, D. G. & Elkan, C. The Transporter Classification Database: recent advances. Nucleic Acids Res. 37, D274–D278 (2009)
Saier, M. H., Jr, Tran, C. V. & Barabote, R. D. TCDB: the Transporter Classification Database for membrane transport protein analyses and information. Nucleic Acids Res. 34, D181–D186 (2006)
Thompson, J. D., Higgins, D. G. & Gibson, T. J. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22, 4673–4680 (1994)
Felsenstein, J. PHYLIP—Phylogeny Inference Package (version 3.2). Cladistics 5, 164–166 (1989)
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997)
Collaborative Computational Project Number 4. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Cryst. 40, 658–674 (2007)
Cowtan, K. dm: an automated procedure for phase improvement by density modification. Joint CCP4 ESF-EACBM Newsl. Prot. Crystallogr. 31, 34–38 (1994)
Adams, P. D. et al. PHENIX: building new software for automated crystallographic structure determination. Acta Crystallogr. D 58, 1948–1954 (2002)
Veenhoff, L. M. & Poolman, B. Substrate recognition at the cytoplasmic and extracellular binding site of the lactose transport protein of Streptococcus thermophilus. J. Biol. Chem. 274, 33244–33250 (1999)
Scheich, C., Kummel, D., Soumailakakis, D., Heinemann, U. & Bussow, K. Vectors for co-expression of an unrestricted number of proteins. Nucleic Acids Res. 35, e43 (2007)
Acknowledgements
We thank J. He, L. Tang, F. Yu and S. Huang at Shanghai Synchrotron Radiation Facility, and K. Hasegawa and T. Kumasaka at the SPring-8 beamline BL41XU, for on-site assistance. This work was supported by funds from the Ministry of Science and Technology (grant numbers 2009CB918802 and 2011CB910501), projects 31125009 and 91017011 of the National Natural Science Foundation of China, and funds from Tsinghua University.
Author information
Authors and Affiliations
Contributions
L.S., X.Z. and N.Y. designed all experiments. L.S., X.Z., C.Y., X.G. and N.Y. performed the experiments for structural determination, homology-based structure modelling and biochemical analysis. X.S. and Y.R. synthesized 6-BrGlc. All authors analysed the data and contributed to manuscript preparation. N.Y. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains a Supplementary Discussion, Supplementary Figures 1-10 and Supplementary Table 1. (PDF 2913 kb)
Rights and permissions
About this article
Cite this article
Sun, L., Zeng, X., Yan, C. et al. Crystal structure of a bacterial homologue of glucose transporters GLUT1–4. Nature 490, 361–366 (2012). https://doi.org/10.1038/nature11524
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11524
This article is cited by
-
Multiple roles for the cytoplasmic C-terminal domains of the yeast cell surface receptors Rgt2 and Snf3 in glucose sensing and signaling
Scientific Reports (2024)
-
Ion and lipid orchestration of secondary active transport
Nature (2024)
-
Establishing mammalian GLUT kinetics and lipid composition influences in a reconstituted-liposome system
Nature Communications (2023)
-
GLUT4 gene rs5418 polymorphism is associated with increased coronary heart disease risk in a Uygur Chinese population
BMC Cardiovascular Disorders (2022)
-
Machine learning and comparative genomics approaches for the discovery of xylose transporters in yeast
Biotechnology for Biofuels and Bioproducts (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.