Abstract
The adult mammalian heart is incapable of regeneration following cardiomyocyte loss, which underpins the lasting and severe effects of cardiomyopathy. Recently, it has become clear that the mammalian heart is not a post-mitotic organ. For example, the neonatal heart is capable of regenerating lost myocardium1, and the adult heart is capable of modest self-renewal2,3. In both of these scenarios, cardiomyocyte renewal occurs via the proliferation of pre-existing cardiomyocytes, and is regulated by aerobic-respiration-mediated oxidative DNA damage4,5. Therefore, we reasoned that inhibiting aerobic respiration by inducing systemic hypoxaemia would alleviate oxidative DNA damage, thereby inducing cardiomyocyte proliferation in adult mammals. Here we report that, in mice, gradual exposure to severe systemic hypoxaemia, in which inspired oxygen is gradually decreased by 1% and maintained at 7% for 2 weeks, results in inhibition of oxidative metabolism, decreased reactive oxygen species production and oxidative DNA damage, and reactivation of cardiomyocyte mitosis. Notably, we find that exposure to hypoxaemia 1 week after induction of myocardial infarction induces a robust regenerative response with decreased myocardial fibrosis and improvement of left ventricular systolic function. Genetic fate-mapping analysis confirms that the newly formed myocardium is derived from pre-existing cardiomyocytes. These results demonstrate that the endogenous regenerative properties of the adult mammalian heart can be reactivated by exposure to gradual systemic hypoxaemia, and highlight the potential therapeutic role of hypoxia in regenerative medicine.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Porrello, E. R. et al. Transient regenerative potential of the neonatal mouse heart. Science 331, 1078–1080 (2011)
Bergmann, O. et al. Dynamics of cell generation and turnover in the human heart. Cell 161, 1566–1575 (2015)
Senyo, S. E. et al. Mammalian heart renewal by pre-existing cardiomyocytes. Nature 493, 433–436 (2013)
Puente, B. N. et al. The oxygen-rich postnatal environment induces cardiomyocyte cell-cycle arrest through DNA damage response. Cell 157, 565–579 (2014)
Kimura, W. et al. Hypoxia fate mapping identifies cycling cardiomyocytes in the adult heart. Nature 523, 226–230 (2015)
Kikuchi, K. et al. Primary contribution to zebrafish heart regeneration by gata4+ cardiomyocytes. Nature 464, 601–605 (2010)
Poss, K. D., Wilson, L. G. & Keating, M. T. Heart regeneration in zebrafish. Science 298, 2188–2190 (2002)
Jopling, C. et al. Zebrafish heart regeneration occurs by cardiomyocyte dedifferentiation and proliferation. Nature 464, 606–609 (2010)
Adam-Vizi, V. & Chinopoulos, C. Bioenergetics and the formation of mitochondrial reactive oxygen species. Trends Pharmacol. Sci. 27, 639–645 (2006)
Roach, R. C. & Hackett, P. H. Frontiers of hypoxia research: acute mountain sickness. J. Exp. Biol. 204, 3161–3170 (2001)
Subramanian, R. K. et al. Normative data for arterial blood gas and electrolytes in anesthetized rats. Indian J. Pharmacol. 45, 103–104 (2013)
Wise, D. R. et al. Hypoxia promotes isocitrate dehydrogenase-dependent carboxylation of α-ketoglutarate to citrate to support cell growth and viability. Proc. Natl Acad. Sci. USA 108, 19611–19616 (2011)
Poleshko, A. G., Lobanok, E. S. & Volotovskii, I. D. Effect of hypoxia on porphyrin metabolism in bone marrow mesenchymal stem cells. Bull. Exp. Biol. Med. 157, 167–171 (2014)
Vicario, C., Juanes, M. C., Martín-Barrientos, J. & Medina, J. M. Effect of postnatal hypoxia on ammonia metabolism during the early neonatal period in the rat. Biol. Neonate 57, 119–125 (1990)
Löffler, M. The “anti-pyrimidine effect” of hypoxia and brequinar sodium (NSC 368390) is of consequence for tumor cell growth. Biochem. Pharmacol. 43, 2281–2287 (1992)
Skappak, C., Regush, S., Cheung, P. Y. & Adamko, D. J. Identifying hypoxia in a newborn piglet model using urinary NMR metabolomic profiling. PLoS One 8, e65035 (2013)
Mullen, A. R. et al. Oxidation of alpha-ketoglutarate is required for reductive carboxylation in cancer cells with mitochondrial defects. Cell Reports 7, 1679–1690 (2014)
Kasai, H. & Nishimura, S. Hydroxylation of the C-8 position of deoxyguanosine by reducing agents in the presence of oxygen. Nucleic Acids Symp. Ser. 12, 165–167 (1983)
Kobs, R. W., Muvarak, N. E., Eickhoff, J. C. & Chesler, N. C. Linked mechanical and biological aspects of remodeling in mouse pulmonary arteries with hypoxia-induced hypertension. Am. J. Physiol. Heart Circ. Physiol. 288, H1209–H1217 (2005)
Djordjevic, B. & Szybalski, W. Genetics of human cell lines. III. Incorporation of 5-bromo- and 5-iododeoxyuridine into the deoxyribonucleic acid of human cells and its effect on radiation sensitivity. J. Exp. Med. 112, 509–531 (1960)
Saffhill, R. & Ockey, C. H. Strand breaks arising from the repair of the 5-bromodeoxyuridine-substituted template and methyl methanesulphonate-induced lesions can explain the formation of sister chromatid exchanges. Chromosoma 92, 218–224 (1985)
Morris, S. M. The genetic toxicology of 5-bromodeoxyuridine in mammalian cells. Mutat. Res. 258, 161–188 (1991)
Malliaras, K. et al. Cardiomyocyte proliferation and progenitor cell recruitment underlie therapeutic regeneration after myocardial infarction in the adult mouse heart. EMBO Mol. Med. 5, 191–209 (2013)
Murray, T. V. et al. Redox regulation of cardiomyocyte cell cycling via an ERK1/2 and c-Myc-dependent activation of cyclin D2 transcription. J. Mol. Cell. Cardiol. 79, 54–68 (2015)
Le Belle, J. E. et al. Proliferative neural stem cells have high endogenous ROS levels that regulate self-renewal and neurogenesis in a PI3K/Akt-dependant manner. Cell Stem Cell 8, 59–71 (2011)
Takahashi, A. et al. Mitogenic signalling and the p16INK4a–Rb pathway cooperate to enforce irreversible cellular senescence. Nat. Cell Biol. 8, 1291–1297 (2006)
Perales-Clemente, E., Folmes, C. D. & Terzic, A. Metabolic regulation of redox status in stem cells. Antioxid. Redox Signal. 21, 1648–1659 (2014)
Mahmoud, A. I. et al. Meis1 regulates postnatal cardiomyocyte cell cycle arrest. Nature 497, 249–253 (2013)
Acknowledgements
We thank the McDermott Center Sequencing and Bioinformatics Cores for sequencing and analysis, J. Richardson and J. Shelton for assistance with histology, G. Jay for assistance with the hypoxia protocol, K. Nham for help with coronary imaging and A. Darehshouri for assistance with electron microscopy. This work is supported by the NIH (grant 1R01HL115275-01) (H.A.S.), and Center for Regenerative Science and Medicine. C.X.S. was partially supported by NIH grant UL1TR001105. A.M.S. is supported by the British Heart Foundation (CH/1999001/11735).
Author information
Authors and Affiliations
Contributions
Y.N., D.C.C. and W.K. designed and performed the experiments and wrote the manuscript. Y.N., D.C.C., W.K. and H.A.S. analysed the data. S.T. performed experiments related to animal husbandry as well as various immunohistochemical studies. S.A. and A.A. performed confocal imaging quantification and studies. C.X.S. and A.M.S. performed mass-spectrometry-based analysis and quantification. H.Z. and J.E.F. performed coronary casting and the data analysis. M.T.K. and L.I.S. performed mitochondrial function and mass spectrometry analysis of metabolic enzymes. C.X. supervised the analysis of RNA-seq studies. R.J.D. and Z.H. performed metabolite measurements and data analysis. O.O. contributed coronary imaging. Z.L. performed RNA sample preparation for RNA-seq. C.C.Z. designed and supervised RNA-seq studies. J.A.H. and G.S. acquired and analysed pulmonary artery acceleration time data. H.A.S. conceived the project, contributed to experimental design and manuscript preparation. All authors approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks R. Lee and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
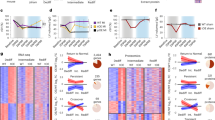
Extended Data Figure 1 Metabolic effects of hypoxia.
a, Food consumption over the course of hypoxia exposure demonstrating a gradual decline (n = 9). b, Blood gas and pH analysis (n = 14 for normoxia control group, and n = 9 for hypoxia group). c, Two days of exposure to 7% O2 showed a significant increase in number of tdTomato+ cardiomyocytes in αMHC-CreERT2-ODD;R26/tdTomato double reporter mice, indicating an enhancement of Hif1α stabilization (n = 4 each). Scale bars, 50 μm. d, Comprehensive quantitative mass spectrometry analysis of enzymes involved in Krebs cycle and fatty acid β oxidation (n = 3 each). e, Dihydrorhodamine 123 staining on fresh cryosections revealed a significant decrease in ROS levels in hypoxia-treated hearts (n = 3 each). Scale bars, 100 μm. Data are presented as mean ± s.e.m. *P < 0.05, **P < 0.01.
Extended Data Figure 3 Hypoxia metabolome profile.
a, Principal component analysis (PCA) plot for the 10 extracts shown in Extended Data Fig. 2. b, The list of differentially regulated metabolic pathways in hypoxic heart compared with control normoxic hearts.
Extended Data Figure 4 Effect of hypoxia on cardiac growth.
a, Heart weight, body weight (n = 6 each), right ventricular weight to body weight ratio, and left ventricular and septum weight to body weight ratio (n = 4 for normoxia, and 5 for hypoxia) after 2 weeks of exposure to 7% oxygen. b, Wall dimension of right ventricular, intraventricular septum and left ventricular wall were all increased after 2 weeks of exposure to 7% oxygen (n = 3 each). Cardiomyocyte size in the right ventricle showed a significant increase after 2 weeks exposure to 7% oxygen (n = 3 each). c, Complete dissociation of cardiomyocytes yields a larger pellet of digested cardiomyocyte. d, Two weeks of exposure to hypoxia results in an increased mononucleated cardiomyocytes and reduced binucleated cardiomyocytes. Cardiomyocytes were isolated from whole ventricle (n = 3 each).
Extended Data Figure 5 RNA-seq analysis of hypoxic and normoxic left ventricles.
a–h, Ontology analysis was performed using DAVID Functional Annotation Tool. The heat maps show a number of dysregulated pathways including metabolism, cell cycle, angiogenesis and hypertrophy. Red and green colours represent upregulated and downregulated genes, respectively (n = 2 each duplicate).
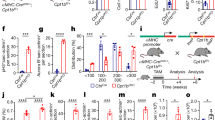
Extended Data Figure 6 Differential effect of hypoxia on cardiomyocyte proliferation.
a, Expansion of tdTomato+ cardiomyocytes in αMHC-CreERT2-ODD;R26/tdTomato indicates enhancement of cell division in ODD+ cardiomyocytes (n = 4 for normoxia, and 3 for hypoxia). Scale bars, 100 μm. b, The number of pH3S10/tdTomato double-positive cardiomyocytes showed an increase in hypoxic αMHC-CreERT2-ODD;R26/tdTomato transgenic mice (rate of tdTomato+/pH3S10+ double-positive cardiomyocytes to tdTomato+ single-positive cardiomyocytes, n = 3 each; rate of tdTomato+/pH3S10+ double-positive cardiomyocytes to all pH3S10+, n = 6 for all pH3S10+, n = 3 for tdTomato+/pH3S10+ double-positive). Scale bars, 10 μm. c, Distribution of proliferating cardiomyocytes in the border and remote zones. The majority of pH3S10+ and BrdU+ cardiomyocytes were localized to the remote zone rather than the MI border zone (n = 3 each).
Extended Data Figure 7 Effect of 3 weeks of hypoxia on heart regeneration.
a, Schematic of 3 week hypoxia protocol. Mice were exposed to gradual FiO2 reduction to 7% (one week following MI), which was then maintained for 3 weeks. Mice were then either euthanized, or subjected to gradual FiO2 increase back to normoxia. b, Cumulative survival curve (Kaplan–Meier survival plot) of MI and sham-operated mice after reaching 7% of oxygen (n = 8 for sham-operated mice, and n = 14 for MI mice). c, Left ventricular function for sham and MI groups exposed to 3 weeks of 7% FiO2 (longer duration hypoxia) (n = 8 for normoxia sham-operated mice, and n = 7 for hypoxia sham-operated mice; n = 9 for normoxia MI mice, and n = 6 for hypoxia MI mice). d, PVacceleration time after return to normoxia showed no evidence of pulmonary hypertension (manifested by shortened PV acceleration time) (n = 3 each).
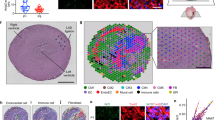
Extended Data Figure 9 Exposure of mice to mild hypoxia (10% O2) with or without MitoTEMPO.
a, Schematic of gradual induction of mild hypoxaemia. b, Heart weight to body weight ratio showed a slight increase in the mild hypoxia group (n = 5 each). c, d, Cell size (c) and pH3S10 quantification (d) showed no significant difference between mild hypoxia and normoxia groups (n = 3 each). e, Schematic of MI followed by 10% hypoxia protocol. f, Echocardiography did not show significant improvement in left ventricular ejection fraction following exposure to 10% hypoxia (n = 7 for sham-operated mice, and n = 13 for hypoxia MI mice).
Source data
Rights and permissions
About this article
Cite this article
Nakada, Y., Canseco, D., Thet, S. et al. Hypoxia induces heart regeneration in adult mice. Nature 541, 222–227 (2017). https://doi.org/10.1038/nature20173
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature20173
This article is cited by
-
Animal models to study cardiac regeneration
Nature Reviews Cardiology (2024)
-
N-Acetyltransferase 10 represses Uqcr11 and Uqcrb independently of ac4C modification to promote heart regeneration
Nature Communications (2024)
-
Telomere-based treatment strategy of cardiovascular diseases: imagination comes to reality
Genome Instability & Disease (2024)
-
Regeneration of the heart: from molecular mechanisms to clinical therapeutics
Military Medical Research (2023)
-
Redifferentiated cardiomyocytes retain residual dedifferentiation signatures and are protected against ischemic injury
Nature Cardiovascular Research (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.