Abstract
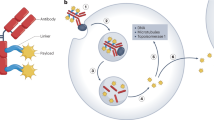
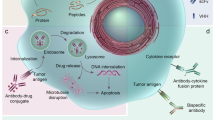
Antibody-drug conjugates enhance the antitumor effects of antibodies and reduce adverse systemic effects of potent cytotoxic drugs. However, conventional drug conjugation strategies yield heterogenous conjugates with relatively narrow therapeutic index (maximum tolerated dose/curative dose). Using leads from our previously described phage display–based method to predict suitable conjugation sites, we engineered cysteine substitutions at positions on light and heavy chains that provide reactive thiol groups and do not perturb immunoglobulin folding and assembly, or alter antigen binding. When conjugated to monomethyl auristatin E, an antibody against the ovarian cancer antigen MUC16 is as efficacious as a conventional conjugate in mouse xenograft models. Moreover, it is tolerated at higher doses in rats and cynomolgus monkeys than the same conjugate prepared by conventional approaches. The favorable in vivo properties of the near-homogenous composition of this conjugate suggest that our strategy offers a general approach to retaining the antitumor efficacy of antibody-drug conjugates, while minimizing their systemic toxicity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Adams, G.P. & Weiner, L.M. Monoclonal antibody therapy of cancer. Nat. Biotechnol. 23, 1147–1157 (2005).
Baker, M. Upping the ante on antibodies. Nat. Biotechnol. 23, 1065–1072 (2005).
Polakis, P. Arming antibodies for cancer therapy. Curr. Opin. Pharmacol. 5, 382–387 (2005).
Wu, A.M. & Senter, P.D. Arming antibodies: prospects and challenges for immunoconjugates. Nat. Biotechnol. 23, 1137–1146 (2005).
Lambert, J.M. Drug-conjugated monoclonal antibodies for the treatment of cancer. Curr. Opin. Pharmacol. 5, 543–549 (2005).
Chen, Y. et al. Armed antibodies targeting the mucin repeats of the ovarian cancer antigen, MUC16, are highly efficacious in animal tumor models. Cancer Res. 67, 4924–4932 (2007).
Doronina, S.O. et al. Development of potent monoclonal antibody auristatin conjugates for cancer therapy. Nat. Biotechnol. 21, 778–784 (2003).
Hamblett, K.J. et al. Effects of drug loading on the antitumor activity of a monoclonal antibody drug conjugate. Clin. Cancer Res. 10, 7063–7070 (2004).
Henry, M.D. et al. A prostate-specific membrane antigen-targeted monoclonal antibody-chemotherapeutic conjugate designed for the treatment of prostate cancer. Cancer Res. 64, 7995–8001 (2004).
Kovtun, Y.V. et al. Antibody-drug conjugates designed to eradicate tumors with homogeneous and heterogeneous expression of the target antigen. Cancer Res. 66, 3214–3221 (2006).
Liu, C. & Chari, R.V. The development of antibody delivery systems to target cancer with highly potent maytansinoids. Expert Opin. Investig. Drugs 6, 169–172 (1997).
Ross, S. et al. Prostate stem cell antigen as therapy target: tissue expression and in vivo efficacy of an immunoconjugate. Cancer Res. 62, 2546–2553 (2002).
Sanderson, R.J. et al. In vivo drug-linker stability of an anti-CD30 dipeptide-linked auristatin immunoconjugate. Clin. Cancer Res. 11, 843–852 (2005).
Sun, M.M. et al. Reduction-alkylation strategies for the modification of specific monoclonal antibody disulfides. Bioconjug. Chem. 16, 1282–1290 (2005).
Tassone, P. et al. In vitro and in vivo activity of the maytansinoid immunoconjugate huN901–N2′-deacetyl-N2′-(3-mercapto-1-oxopropyl)-maytansine against CD56+ multiple myeloma cells. Cancer Res. 64, 4629–4636 (2004).
Tolcher, A.W. et al. Cantuzumab mertansine, a maytansinoid immunoconjugate directed to the CanAg antigen: a phase I, pharmacokinetic, and biologic correlative study. J. Clin. Oncol. 21, 211–222 (2003).
Doronina, S.O. et al. Enhanced activity of monomethylauristatin F through monoclonal antibody delivery: effects of linker technology on efficacy and toxicity. Bioconjug. Chem. 17, 114–124 (2006).
Ma, D. et al. Potent antitumor activity of an auristatin-conjugated, fully human monoclonal antibody to prostate-specific membrane antigen. Clin. Cancer Res. 12, 2591–2596 (2006).
Tse, K.F. et al. CR011, a fully human monoclonal antibody-auristatin E conjugate, for the treatment of melanoma. Clin. Cancer Res. 12, 1373–1382 (2006).
Wang, L., Amphlett, G., Blattler, W.A., Lambert, J.M. & Zhang, W. Structural characterization of the maytansinoid-monoclonal antibody immunoconjugate, huN901–DM1, by mass spectrometry. Protein Sci. 14, 2436–2446 (2005).
Jefferis, R. Structure-function relationships of the IgG subclasses. in The Human IgG Subclasses: Molecular Analysis of Structure, Function and Regulation (ed. Shakib, F.) 93–108 (Pergamon Press, Oxford, 1990).
Junutula, J.R. et al. Rapid identification of reactive cysteine residues for site-specific labeling of antibody-Fabs. J. Immunol. Methods 332, 41–52 (2008).
Dubowchik, G.M. et al. Cathepsin B-labile dipeptide linkers for lysosomal release of doxorubicin from internalizing immunoconjugates: model studies of enzymatic drug release and antigen-specific in vitro anticancer activity. Bioconjug. Chem. 13, 855–869 (2002).
Francisco, J.A. et al. cAC10-vcMMAE, an anti-CD30-monomethyl auristatin E conjugate with potent and selective antitumor activity. Blood 102, 1458–1465 (2003).
Mirsalis, J.C. et al. Toxicity of dolastatin 10 in mice, rats and dogs and its clinical relevance. Cancer Chemother. Pharmacol. 44, 395–402 (1999).
McDonagh, C.F. et al. Engineered antibody-drug conjugates with defined sites and stoichiometries of drug attachment. Protein Eng. Des. Sel. 19, 299–307 (2006).
Michaelsen, T.E. et al. One disulfide bond in front of the second heavy chain constant region is necessary and sufficient for effector functions of human IgG3 without a genetic hinge. Proc. Natl. Acad. Sci. USA 91, 9243–9247 (1994).
Romans, D.G., Tilley, C.A., Crookston, M.C., Falk, R.E. & Dorrington, K.J. Conversion of incomplete antibodies to direct agglutinins by mild reduction: evidence for segmental flexibility within the Fc fragment of immunoglobulin G. Proc. Natl. Acad. Sci. USA 74, 2531–2535 (1977).
Seegan, G.W., Smith, C.A. & Schumaker, V.N. Changes in quaternary structure of IgG upon reduction of the interheavy-chain disulfide bond. Proc. Natl. Acad. Sci. USA 76, 907–911 (1979).
Lyons, A. et al. Site-specific attachment to recombinant antibodies via introduced surface cysteine residues. Protein Eng. 3, 703–708 (1990).
Stimmel, J.B. et al. Site-specific conjugation on serine right-arrow cysteine variant monoclonal antibodies. J. Biol. Chem. 275, 30445–30450 (2000).
Chapman, A.P. et al. Therapeutic antibody fragments with prolonged in vivo half-lives. Nat. Biotechnol. 17, 780–783 (1999).
Xiang, T., Lundell, E., Sun, Z. & Liu, H. Structural effect of a recombinant monoclonal antibody on hinge region peptide bond hydrolysis. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 858, 254–262 (2007).
Acknowledgements
The authors wish to thank our Genentech colleagues: Jennifer Speer for preparing trastuzumab THIOMAB DNA constructs; Mary Cole for insights into the OVCAR-3 intraperitoneal efficacy model; Elmer Wu, Darshana Patel, Mark Rowen and Anthony Delucchi for providing critical reagents; Natalia Gomez and George Dutina for large-scale transient transfection/fermentation; Fred Jacobson and Charity Bechtel for their help with analytical characterization of TDCs; and Allen Ebens for critical review of the manuscript. We thank Damon Meyer and his colleagues at Seattle Genetics for preparation of the early lots of anti-MUC16 ADC and TDC and for many helpful comments and suggestions. We thank employees of NPIL Pharma UK for their assistance with large-scale conjugations. We also thank the staff of Oncotest for conducting some of the efficacy studies described in this manuscript. Anti-MMAE mouse monoclonal antibody SG2.15 was generously provided by Seattle Genetics, Inc.
Author information
Authors and Affiliations
Contributions
J.R.J. and W.M. led the overall program, designed experiments, performed in vitro studies, analyzed the data and wrote the manuscript. Y.C. and M.S.D. humanized the anti-MUC16 antibody. S.B. generated the anti-MUC16 THIOMAB DNA constructs and performed pilot expression studies. M.S., E.D. and J.G. performed larger-scale antibody production. H.R. established procedures for TDC conjugation and analytical characterization. C.C.L. carried out analytical characterization of TDCs. S.W., S.P.T., Y.L., Y.G.M., C.N. and J.Y. performed in vitro binding studies. S.C., R. Venook and S.R. performed in vivo efficacy studies. D.D.L. designed and analyzed pharmacokinetic studies. A.K., K.M. and K.F. designed and executed safety assessment studies. V.K., S.D.S., W.L.W., H.B.L., R. Vandlen, M.X.S., R.H.S. and P.P. provided direction and guidance for the various functional areas and assisted in writing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
All authors are full-time employees of Genentech. Inc.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–17, Tables 1–5, Methods (PDF 3037 kb)
Rights and permissions
About this article
Cite this article
Junutula, J., Raab, H., Clark, S. et al. Site-specific conjugation of a cytotoxic drug to an antibody improves the therapeutic index. Nat Biotechnol 26, 925–932 (2008). https://doi.org/10.1038/nbt.1480
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.1480
This article is cited by
-
The target atlas for antibody-drug conjugates across solid cancers
Cancer Gene Therapy (2024)
-
Exploring the next generation of antibody–drug conjugates
Nature Reviews Clinical Oncology (2024)
-
Antibody-Drug Conjugate Overview: a State-of-the-art Manufacturing Process and Control Strategy
Pharmaceutical Research (2024)
-
Design of high avidity and low affinity antibodies for in situ control of antibody drug conjugate targeting
Scientific Reports (2022)
-
Cysteine metabolic engineering and selective disulfide reduction produce superior antibody-drug-conjugates
Scientific Reports (2022)