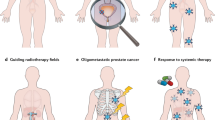
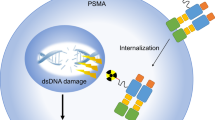
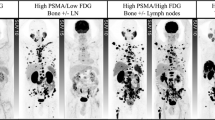
Abstract
Imaging is a critical component of diagnosis, staging and monitoring, all of which factor heavily in treatment decision-making for cancer patients. Agents, such as antibodies, can target molecules that are relatively unique to cancer cells. Prostate-specific membrane antigen (PSMA) is the most well-established, highly restricted prostate-cancer-related cell membrane antigen known. Ten years ago, the FDA approved 111In-capromab pendetide for use in imaging soft-tissue, but not bone, sites of metastatic prostate cancer for presurgical staging or the evaluation of PSA relapse after local therapy. For presurgical patients with high-risk disease but negative bone, CT and MRI scans, capromab demonstrated the ability to identify some patients with positive nodes, thereby sparing them an unnecessary surgical procedure. But there have been no follow-up studies to indicate that high-risk patients with a negative capromab scan have a lower failure rate after surgery. In the setting of PSA relapse, capromab is compromised by its inability to sensitively image bone metastases; bone is the first site of metastatic prostate cancer in 72% of patients. The problem with imaging bone metastases is that capromab detects an antigenic site on the intracellular portion of PSMA—a site not accessible to circulating antibodies. Early results indicate that second-generation antibodies that target the extracellular domain of PSMA might provide significant benefits in the imaging of prostate cancer.
Key Points
-
Prostate-specific membrane antigen (PSMA) is the most well-established, highly restricted prostate cancer-related cell membrane antigen known
-
In 1996, the FDA approved 111In-capromab pendetide (capromab) for imaging soft-tissue, but not bone, sites of metastatic prostate cancer
-
Capromab detects an antigenic site on the intracellular portion of PSMA, a site not accessible to circulating antibody, which thus explains its inability to bind to well-vascularized metastases in the bone marrow
-
The utility of capromab is compromised by its inability to sensitively image bone metastases, as bone is the first site of metastatic prostate cancer spread in approximately 75% of patients
-
The inability to image bone metastases probably explains the inconsistent results reported on the use of capromab for staging patients with PSA relapse
-
Early results indicate that second-generation antibodies that target the extracellular domain of PSMA are able to target and image bone metastases, and, therefore, might provide significant benefits when imaging prostate cancer
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Rifkin MD et al. (1990) Comparison of magnetic resonance imaging and ultrasonography in staging early prostate cancer. N Eng J Med 323: 621–626
Gasser TC et al. (1991) MRI and ultrasonography in staging prostate cancer. N Eng J Med 324: 494–495
Tempany CM and McNeil BJ (2001) Advances in biomedical imaging. JAMA 285: 562–567
Harisinghani MG et al. (2003) Non-invasive detection of clinically occult lymph-node metastases in prostate cancer. N Eng J Med 348: 2491–2499
Babaian RJ et al. (1987) Radioimmunological imaging of metastatic prostate cancer with 111Indium-labeled monoclonal antibody PAY 276. J Urol 137: 439–443
Leroy M et al. (1989) Radioimmunodetection of lymph node invasion in prostate cancer. The use of iodine 123 (1231)-labeled monoclonal anti-prostatic acid phosphatase (PAP) 227 A F(ab')2 antibody fragments in vivo. Cancer 64: 1–5
Horoszewicz JS et al. (1987) Monoclonal antibodies to a new antigenic marker in epithelial cells and serum of prostatic cancer patients. Anticancer Res 7: 927–936
Israeli RS et al. (1993) Molecular cloning of a complementary DNA encoding a prostate-specific membrane antigen. Cancer Res 53: 227–230
Israeli RS et al. (1994) Expression of the prostate-specific membrane antigen. Cancer Res 54: 1807–1811
Wright GL Jr et al. (1995) Expression of prostate-specific membrane antigen in normal, benign, and malignant prostate tissues. Urol Oncol 1: 18–28
Troyer JK et al. (1995) Detection and characterization of the prostate-specific membrane antigen (PSMA) in tissue extracts and body fluids. Int J Cancer 62: 552–558
Sokoloff RL et al. (2000) A dual-monoclonal sandwich assay for prostate-specific membrane antigen: levels in tissues, seminal fluid and urine. Prostate 43: 150–157
Davis MI et al. (2005) Crystal structure of prostate-specific membrane antigen, a tumor marker and peptidase. PNAS 102: 5981–5986
Wright GL Jr et al. (1996) Upregulation of prostate-specific membrane antigen after androgen-deprivation therapy. Urology 48: 326–334
Bostwick DG et al. (1998) Prostate specific membrane antigen expression in prostatic intraepithelial neoplasia and adenocarcinoma. Cancer 82: 2256–2261
Sweat SD et al. (1998) Prostate-specific membrane antigen expression is greatest in prostate adenocarcinoma and lymph node metastases. Urology 52: 637–640
Wynant GE et al. (1991) Immunoscintigraphy of prostatic cancer: preliminary results with 111In-labeled monoclonal antibody 7E11-C5.3 (CYT-356). Prostate 18: 229–241
Deb N et al. (1996) Treatment of hormone-refractory prostate cancer with 90Y-CYT-356 monoclonal antibody. Clin Can Res 2: 1289–1297
Manyak MJ et al. (1999) Immunoscintigraphy with indium-111-capromab pendetide: evaluation before definitive therapy in patients with prostate cancer. Urology 54: 1058–1063
Babaian RJ et al. (1994) Radioimmunoscintigraphy of pelvic lymph nodes with 111indium-labeled monoclonal antibody CYT-356. J Urol 152: 1952–1955
Hinkle GH et al. (1998) Multicenter radioimmunoscintigraphic evaluation of patients with prostate carcinoma using indium-111 capromab pendetide. Cancer 83: 739–747
Ponsky LE et al. (2002) Evaluation of preoperative ProstaScint scans in the prediction of nodal disease. Prostate Cancer Prostatic Dis 5: 132–135
Polascik TJ et al. (1999) Comparison of clinical staging algorithms and 111 indium Capromab pendetide immunoscintigraphy in the prediction of lymph node involvement in high risk prostate carcinoma patients. Cancer 85: 1586–1592
Kahn D et al. (1998) Radioimmunoscintigraphy with In-111-labeled capromab pendetide predicts prostate cancer response to salvage radiotherapy after failed radical prostatectomy. J Clin Oncol 16: 284–289
Kahn D et al. (1999) 111In-capromab pendetide scan predicts response to radiotherapy to the prostate fossa in men with tumor recurrence following radical prostatectomy: 3 year follow-up [abstract]. J Urol 161 (Suppl 4): a239
Levesque PE et al. (1998) radiolabelled monoclonal antibody 111-indium labeled CYT-356 localizes extraprostatic recurrent carcinoma after prostatectomy. Urology 51: 978–984
Wilkenson S et al. (2004) Role of 111-indium capromab pendetide imaging for assessing biochemical failure after radical prostatectomy. J Urol 172: 133–136
Thomas CT et al. (2003) Indium-111 capromab pendetide radioimmunoscintigraphy and prognosis for durable biochemical response to salvage radiation therapy in men after failed prostatectomy. JCO 21: 1715–1721
DeWyngaert JK (2004) et al. Procedure for unmasking localization information from ProstaScint scans for prostate radiation therapy treatment planning. Int J Radiat Oncl Biol Phys 60: 654–662
Schettino CJ et al. (2004) Impact of fusion of indium-111 capromab pendetide volume data sets with those from MRI or CT in patients with recurrent prostate cancer. Am J Roentgenol 183: 519–524
Sodee DB et al. (1998) Prostate cancer and prostate bed SPECT imaging with Prostascint: semi-quantitative correlation with prostatic biopsy results. Prostate 37: 140–148
Ellis RJ et al. (2001) Radioimmunoguided imaging of prostatee cancer foci with histopathological correlation. Int J Radiat Oncol Biol Phys 49: 1281–1286
Ellis RJ et al. (2003) Four year biochemical outcome after radioimmunoguided transperineal brachytherapy for patients with prostate adenocarcinoma. Int J Radiat Oncol Biol Phys 57: 362–370
Saitoh H et al. (1990) Two different lymph node metastatic patterns of a prostatic cancer. Cancer 65: 1843–1846
Troyer JK et al. (1995) Biochemical characterization and mapping of the 7E11-C5.3 epitope of the prostate-specific membrane antigen. Urol Oncol 1: 29–37
Troyer JK et al. (1997) Location of prostate-specific membrane antigen in the LNCaP prostate carcinoma cell line. Prostate 30: 232–242
Liu H et al. (1997) Monoclonal antibodies to the extracellular domain of prostate specific membrane antigen also react with tumor endothelium. Cancer Res 57: 3629–3634
Smith-Jones PM et al. (2003) Radiolabeled monoclonal antibodies specific to the extracellular domain of prostate-specific membrane antigen: preclinical studies in nude mice bearing LNCaP human prostate tumor. J Nuc Med 44: 610–617
Smith-Jones PM et al. (2000) In vitro characterization of radiolabelled monoclonal antibodies specific for the extracellular domain of prostate-specific membrane antigen. Cancer Res 60: 5237–5243
Liu H et al. (1998) Constitutive and antibody-induced internalization of prostate-specific membrane antigen. Cancer Res 58: 4055–4060
Milowsky MI et al. (2004) Phase I trial of yttrium-90-labeled anti-prostate-specific membrane antigen monoclonal antibody J591 for androgen-independent prostate cancer. J Clin Oncol 22: 2522–2531
Bander NH (2005) Phase I trial of 177Lutetium-labeled J591, a monoclonal antibody to prostate-specific membrane antigen, in patients with androgen-independent prostate cancer. J Clin Onc 23: 4591–4601
Bander NH et al. (2003) Targeted systemic therapy of prostate cancer with a monoclonal antibody to prostate-specific membrane antigen. Semin Oncol 30: 667–676
Bander NH et al. (2003) Targeting metastatic prostate cancer with radiolabeled monoclonal antibody J591 to the extracellular domain of prostate specific membrane antigen. J Urol 170: 1717–1721
Hamilton A et al. (1998) A novel humanized antibody against prostate specific membrane antigen (PSMA) for in vivo targeting and therapy. Proc Am Assoc Cancer Res 39: 440
Michalet X et al. (2005) Quantum dots for live cells, in vivo imaging and diagnostics. Science 307: 538–544
Xiaohu G et al. (2004) In vivo cancer targeting and imaging with semiconductor quantum dots. Nat Biotechnol 22: 969–976
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Neil H Bander developed monoclonal antibodies to the extracellular domain of prostate-specific membrane antigen and assigned patent rights to Cornell Research Foundation. These patents were licensed to BZL Biologics Inc. Neil H Bander is a paid consultant to BZL Biologics Inc.
Rights and permissions
About this article
Cite this article
Bander, N. Technology Insight: monoclonal antibody imaging of prostate cancer. Nat Rev Urol 3, 216–225 (2006). https://doi.org/10.1038/ncpuro0452
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpuro0452
This article is cited by
-
The Need to Pair Molecular Monitoring Devices with Molecular Imaging to Personalize Health
Molecular Imaging and Biology (2022)
-
The diagnostic value of PET/CT imaging with the 68Ga-labeled PSMA-ligand in the follow up assessment of prostate cancer after therapy
Egyptian Journal of Radiology and Nuclear Medicine (2020)
-
Inconsistent Detection of Sites of Metastatic Non-Clear Cell Renal Cell Carcinoma with PSMA-Targeted [18F]DCFPyL PET/CT
Molecular Imaging and Biology (2019)
-
Combined Early and Late [68Ga]PSMA-HBED-CC PET Scans Improve Lesion Detectability in Biochemical Recurrence of Prostate Cancer with Low PSA Levels
Molecular Imaging and Biology (2019)
-
68Ga-PSMA and 11C-Choline comparison using a tri-modality PET/CT-MRI (3.0 T) system with a dedicated shuttle
European Journal of Hybrid Imaging (2018)