Abstract
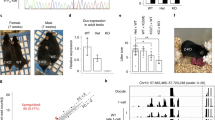
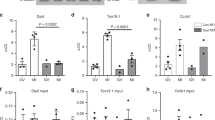
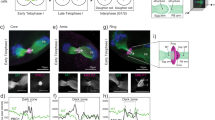
The female gamete (the oocyte) serves the distinct purpose of transmitting the maternal genome and other maternal factors that are critical for post-ovulation events1,2,3,4. Through the identification and characterization of oocyte-specific factors, we are beginning to appreciate the diverse functions of oocytes in ovarian folliculogenesis, fertilization and embryogenesis5,6. To understand these processes further, we identified genes called zygote arrest 1 (Zar1 and ZAR1 in mouse and human, respectively) as novel oocyte-specific genes. These encode proteins of 361 amino acids and 424 amino acids, respectively, which share 59% amino-acid identity and an atypical plant homeo-domain (PHD) motif7. Although Zar1-null (Zar1−/−) mice are viable and grossly normal, Zar1−/− females are infertile. Ovarian development and oogenesis through the early stages of fertilization are evidently unimpaired, but most embryos from Zar1−/− females arrest at the one-cell stage. Distinct pronuclei form and DNA replication initiates, but the maternal and paternal genomes remain separate in arrested zygotes. Fewer than 20% of the embryos derived from Zar1−/− females progress to the two-cell stage and show marked reduction in the synthesis of the transcription-requiring complex8, and no embryos develop to the four-cell stage. Thus, Zar1 is the first identified oocyte-specific maternal-effect gene that functions at the oocyte-to-embryo transition and, as such, offers new insights into the initiation of embryonic development and fertility control in mammals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Latham, K.E. Mechanisms and control of embryonic genome activation in mammalian embryos. Int. Rev. Cytol. 193, 71–124 (1999).
Latham, K.E. & Schultz, R.M. Embryonic genome activation. Front. Biosci. 6, 748–759 (2001).
Schultz, R.M. Regulation of zygotic gene activation in the mouse. Bioessays 15, 531–538 (1993).
Aoki, F., Worrad, D.M. & Schultz, R.M. Regulation of transcriptional activity during the first and second cell cycles in the pre-implantation mouse embryo. Dev. Biol. 181, 296–307 (1997).
Matzuk, M.M., Burns, K., Viveiros, M.M. & Eppig, J. Intercellular communication in the mammalian ovary: oocytes carry the conversation. Science 296, 2178–2180 (2002).
Matzuk, M.M. & Lamb, D.J. Genetic dissection of mammalian fertility pathways. Nat. Med. 8, S41–S49 (2002).
Aasland, R., Gibson, T.J. & Stewart, A.F. The PHD finger: implications for chromatin-mediated transcriptional regulation. Trends Biochem. Sci. 20, 56–59 (1995).
Conover, J.C., Temeles, G.L., Zimmermann, J.W., Burke, B. & Schultz, R.M. Stage-specific expression of a family of proteins that are major products of zygotic gene activation in the mouse embryo. Dev. Biol. 144, 392–404 (1991).
Tong, Z.B. et al. Mater, a maternal effect gene required for early embryonic development in mice. Nat. Genet. 26, 267–268 (2000).
Howell, C.Y. et al. Genomic imprinting disrupted by a maternal-effect mutation in the Dnmt1 gene. Cell 104, 829–838 (2001).
Prolla, T.A. et al. Tumour susceptibility and spontaneous mutation in mice deficient in Mlh1, Pms1 and Pms2 DNA mismatch repair. Nat. Genet. 18, 276–279 (1998).
Christians, E., Davis, A.A., Thomas, S.D. & Benjamin, I.J. Maternal effect of Hsf1 on reproductive success. Nature 407, 693–694 (2000).
Dong, J. et al. Growth differentiation factor 9 is required during early ovarian folliculogenesis. Nature 383, 531–535 (1996).
Gibbons, R.J. et al. Mutations in transcriptional regulator ATRX establish the functional significance of a PHD-like domain. Nat. Genet. 17, 146–148 (1997).
Capili, A.D., Schultz, D.C., Rauscher, I.F. & Borden, K.L. Solution structure of the PHD domain from the KAP-1 corepressor: structural determinants for PHD, RING and LIM zinc-binding domains. EMBO J. 20, 165–177 (2001).
Kumar, T.R. et al. Reproductive defects in γ-glutamyl transpeptidase-deficient mice. Endocrinology 141, 4270–4277 (2000).
Flach, G., Johnson, M.H., Braude, P.R., Taylor, R.A. & Bolton, V.N. The transition from maternal to embryonic control in the two-cell mouse embryo. EMBO J. 1, 681–686 (1982).
Tong, Z.B., Bondy, C.A., Zhou, J. & Nelson, L.M. A human homologue of mouse Mater, a maternal-effect gene essential for early embryonic development. Hum. Reprod. 17, 903–911 (2002).
Albrecht, U., Eichele, G., Helms, J.A. & Lu, H.C. Visualization of gene expression patterns by in situ hybridization. in Molecular and Cellular Methods in Developmental Toxicology (ed. Daston, G.P.) 23–48 (CRC Press, Boca Raton, Florida, 1997).
Elvin, J.A., Yan, C., Wang, P., Nishimori, K. & Matzuk, M.M. Molecular characterization of the follicle defects in the growth differentiation factor 9-deficient ovary. Mol. Endocrinol. 13, 1018–1034 (1999).
Matzuk, M.M. & Bradley, A. Structure of the mouse activin receptor type II gene. Biochem. Biophys. Res. Commun. 185, 404–413 (1992).
Matzuk, M.M., Finegold, M.J., Su, J.-G.J., Hsueh, A.J.W. & Bradley, A. α-Inhibin is a tumor-suppressor gene with gonadal specificity in mice. Nature 360, 313–319 (1992).
Bradley, A. Production and analysis of chimeric mice-teratocarcinomas and embryonic stem cells: a practical approach. in Production and Analysis of Chimeric Mice (ed. Robinson, E.J.) 113–151 (Oxford, London, 1987).
Eppig, J.J. Mouse oocyte maturation, fertilization, and pre-implantation development in vitro. in A Comparative Methods Approach to the Study of Oocytes and Embryos (ed. Richter, J.D.) 3–9 (Oxford University Press, Oxford, 1999).
Yan, W. et al. Identification of Gasz, an evolutionarily conserved gene expressed exclusively in germ cells and encoding a protein with four ankyrin repeats, a sterile-alpha motif, and a basic leucine zipper. Mol. Endocrinol. 16, 1168–1184 (2002).
Ferreira, J. & Carmo-Fonseca, M. Genome replication in early mouse embryos follows a defined temporal and spatial order. J. Cell. Sci. 110, 889–897 (1997).
Yaffe, M.B. et al. A motif-based profile scanning approach for genome-wide prediction of signaling pathways. Nat. Biotechnol. 19, 348–353 (2001).
Elvin, J.A., Clark, A.T., Wang, P., Wolfman, N.M. & Matzuk, M.M. Paracrine actions of growth differentiation factor 9 in the mammalian ovary. Mol. Endocrinol. 13, 1035–1048 (1999).
Pedersen, T. & Peters, H. Proposal for a classification of oocytes and follicles in the mouse ovary. J. Reprod. Fertil. 17, 555–557 (1968).
Acknowledgements
We thank M. Berry, R. Burgess, K. Burns, T. Gridley, G. Kopf and T. Kumar for critical review of the manuscript, S. Baker for aid in manuscript formatting and C. Brown, K. Burns, L. Chen, L. Erdos, M. O'Brien, P. Wang and C. Yan for technical assistance. These studies were supported in part by a sponsored research grant from Wyeth Research and US National Institutes of Health grants (to M.M.M. and J.J.E.). X. Wu was supported in part by a post-doctoral fellowship from the Lalor Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
This work was supported by a grant from Wyeth Research as described in the Acknowledgments.
Rights and permissions
About this article
Cite this article
Wu, X., Viveiros, M., Eppig, J. et al. Zygote arrest 1 (Zar1) is a novel maternal-effect gene critical for the oocyte-to-embryo transition. Nat Genet 33, 187–191 (2003). https://doi.org/10.1038/ng1079
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1079
This article is cited by
-
Landscape of pathogenic mutations in premature ovarian insufficiency
Nature Medicine (2023)
-
A new enrichment approach for candidate gene detection in unexplained recurrent pregnancy loss and implantation failure
Molecular Genetics and Genomics (2023)
-
Centenarian clocks: epigenetic clocks for validating claims of exceptional longevity
GeroScience (2023)
-
Sexual determination in zebrafish
Cellular and Molecular Life Sciences (2022)
-
Maternal effect factors that contribute to oocytes developmental competence: an update
Journal of Assisted Reproduction and Genetics (2022)