Abstract
The biliary system, pancreas and liver all develop from the nearby foregut at almost the same time in mammals. The molecular mechanisms that determine the identity of each organ in this complex area are unknown. Hes1 encodes the basic helix-loop-helix protein Hes1 (ref. 1), which represses positive basic helix-loop-helix genes2 such as Neurog3 (ref. 3). Expression of Hes1 is controlled by the evolutionarily conserved Notch pathway4. Hes1 operates as a general negative regulator of endodermal endocrine differentiation5,6, and defects in Notch signaling lead to accelerated pancreatic endocrine differentiation7,8. Mutations in JAG1, encoding a Notch ligand, cause the Alagille syndrome in humans9,10, characterized by poor development of the biliary system11, suggesting that the Notch pathway is also involved in normal biliary development. Here we show that Hes1 is expressed in the extrahepatic biliary epithelium throughout development and that Hes1-deficient mice2 have gallbladder agenesis and severe hypoplasia of extrahepatic bile ducts. Biliary epithelium in Hes1−/− mice ectopically expresses the proendocrine gene Neurog3 (refs. 12,13), differentiates into endocrine and exocrine cells and forms acini and islet-like structures in the mutant bile ducts. Thus, biliary epithelium has the potential for pancreatic differentiation and Hes1 determines biliary organogenesis by preventing the pancreatic differentiation program, probably by directly repressing transcription of Neurog3.
Similar content being viewed by others
Main
We first examined the expression of the Notch signaling components in the biliary system and other foregut-derived organs during mouse embryogenesis. We carried out semiquantitative RT-PCR using gallbladder, liver and pancreas isolated from normal CD1 mouse embryos (Fig. 1). We detected Jag1 and Jag2, encoding Notch ligands, in the foregut, gallbladder and pancreas. Of the four Notch genes, Notch1 and Notch2 were expressed in the gallbladder and pancreas after embryonic day (E) 12.5. We observed high levels of Hes1 mRNA in the foregut, gallbladder and pancreas but not in the liver throughout development. On the other hand, we did not detect Hes5, encoding another transcriptional repressor of the Hes family14, in any endodermal organ. These findings suggest that the Notch pathway may contribute to biliary and pancreatic development.
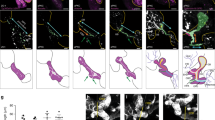
To further clarify the role of the Notch pathway in biliary development, we next analyzed biliary morphogenesis in Hes1-deficient mice2. At E9.5, Hes1−/− embryos had no apparent abnormalities: both wild-type and Hes1−/− mice had the bile duct budding from the foregut (Fig. 2a,d). At E11.5, normal elongation of the extrahepatic biliary duct and branching of the ventral pancreas from the common bile duct were clearly observed in Hes1+/+ mice (Fig. 2b,c). Hes1−/− mice, however, had a short, truncated remnant of the common bile duct and proliferation of aggregated epithelial cells that looked like budding vesicles with a central cavity, resembling the future pancreatic acini (Fig. 2e). Staining with a bile duct–specific lectin15, Dolichos biflorus agglutinin (DBA), showed that the aggregated epithelial cells had lost DBA binding (Fig. 2f), like the branching pancreas in wild-type mice (Fig. 2c). These results imply a functional role of Hes1 in maintaining the biliary properties, repressing the vesicular formation of epithelial cells and promoting the growth of the ductal structure of the prospective biliary epithelium.
(a,b,d,e) Staining of developing extrahepatic bile ducts with hematoxylin and eosin. Arrows in a,d indicate biliary budding from the hepatic diverticulum (HD). In the mutant (e), a remnant of the common bile duct and proliferation of aggregated epithelial cells are observed. (c,f) Adjacent sections stained with biliary-specific DBA. The ventral pancreas (c, red arrow) and aggregated epithelial cells (f, red asterisk) are negative for DBA binding, whereas the normal extrahepatic biliary system (c) and the remnant of the bile duct (f, arrowhead) are positive. CBD, common bile duct; Du, duodenum; GB, gallbladder; Li, liver; Pa, ventral pancreas; PV, main portal vein. Scale bars, 100 μm.
Because most homozygous Hes1 mutant mice die by E18.5, we examined E17.5 mice in detail to see the ultimate form of biliary morphogenesis. All Hes1−/− embryos that we examined had agenesis of the gallbladder and cystic duct (Fig. 3g), whereas wild-type littermates had these organs (Fig. 3a). We examined serial sagittal sections from the duodenal papilla to intrahepatic bile ducts by DBA staining. In wild-type mice, we observed well-formed extrahepatic bile ducts (Fig. 3b–e). In Hes1−/− embryos, the duodenal portion of the common bile duct was small, truncated halfway along and connected to a pancreas-like structure (Fig. 3h,i), and we observed pancreatic tissue at the missing part of the common bile duct. On the other hand, intrahepatic bile ducts or primordia of hepatic ducts, probably originating from the hepatoblast, developed at the porta hepatis in Hes1−/− mice, although these twisted and fragmented lumen structures were never connected with the common bile duct (Fig. 3i–k). The common bile duct, which was observed in horizontal sections at the level of the first part of the duodenum (Fig. 3f), was totally replaced by pancreatic tissue in Hes1−/− embryos (Fig. 3l).
Hes1-deficient mice. Macroscopic view, showing a normal gallbladder and cystic duct (a) and the absence of these organs in the mutant (g). (b–e and h–k) Serial sagittal sections of the biliary system stained with DBA. In the wild-type mice, the extrahepatic biliary system from the duodenal papilla (b, arrow) to the common bile duct (CBD; d) and cystic duct (CD; e) are completely formed. In the mutant, the small remnant of the common bile duct disappears halfway along (h,i, arrows). The fragmented lumen structures (i–k, arrowheads) are never connected with the common bile duct. (f,l) Horizontal sections stained with hematoxylin and eosin and antibody to amylase (dark purple), showing that the common bile duct in Hes1+/+ mouse (f) is totally replaced by pancreatic tissue (Pa) in Hes1−/− littermates (l). Du, duodenum; Li, liver; PV, main portal vein. Scale bars, 100 μm.
To examine whether apoptotic cell death contributes to the failure of biliary development in Hes1−/− mice, we carried out TUNEL staining of the hypoplastic bile ducts. We found no more apoptotic cells in biliary epithelium of mutant mice than in that of wild-type mice (data not shown).
In Hes1-deficient mice, the hypoplastic biliary system seemed to be converted to pancreatic tissue. To test this possibility, we examined the expression of various pancreatic markers during cholangiogenesis. We found four types of endocrine cells in biliary epithelium of Hes1−/− mice: cells expressing glucagon (α cells), insulin (β cells), somatostatin (δ cells) and pancreatic peptide (PP cells; Fig. 4h–m). None of the insulin-positive cells in the mutant bile ducts coexpressed glucagon (Fig. 4o), indicating that these were not early embryonic multihormonal cells, but rather cells along the normal differentiation pathway to mature β cells16,17. These ectopic insulin-positive cells also coexpressed Ipf1, Isl1 and Glut2 (Fig. 4p–r), like mature pancreatic β cells. The hormone-expressing cells formed clusters, leaving the epithelial layer around the biliary lumen (Fig. 4o), like the pancreatic islet cells migrating out of the pancreatic ducts. Mature islets did not appear in either the biliary system or pancreas in Hes1-deficient mice throughout development. Electron microscopic studies showed that the biliary epithelial cells possessed numerous secretory granules (Fig. 4s,t), confirming at the single-cell level that they had differentiated into endocrine cells. Cells positive for postmitotic endocrine marker Isl1 (ref. 18) or exocrine product carboxypeptidase A were abundantly distributed in the mutant bile ducts (Fig. 4j), suggesting that widespread pancreatic differentiation had replaced the biliary epithelium. In contrast to pancreatic development in the mutant5, which showed accelerated differentiation of only glucagon-expressing endocrine cells, biliary epithelium in Hes1-deficient mice showed exocrine differentiation also. Amylase immunostaining clearly showed acinar development from the common bile ducts of Hes1−/− mice (Fig. 4n). These biliary cells of pancreatic nature were never detected in the normal cholangiogenesis of wild-type littermates (Fig. 4a–f), although small numbers of exocrine cells expressing amylase did appear in bile ducts of wild-type mice in the late embryonic stage (Fig. 4g). These findings indicate that inactivation of Hes1 causes the biliary epithelium to adopt the pancreatic fate and to fully differentiate into endocrine and exocrine cells that are components of premature islets and acini.
Pancreatic cell-type differentiation occurs in Hes1- deficient bile ducts, as indicated by the presence (arrowheads in i–n) of the endocrine markers glucagon (h,i), insulin (k), pancreatic peptide (PP; l), somatostatin (m) and Isl1 (j, red) and of the exocrine markers carboxypeptidase A (CA; j, green) and amylase (n). Cytokeratin immunostaining (a,b,d–i and k–n) is observed in the epithelial cells of the bile duct, duodenum and pancreas. b–g, i–n and the outlined areas in a and h show the common bile duct. Pancreatic acini (n, arrows) are budding from the mutant bile duct. (o) Insulin (red) and glucagon (green) are detected in separate sets of biliary epithelial cells located in the outlined area in k. (p–r) Mature β cells are generated in E16 Hes1-deficient bile duct. Confocal micrographs showing a small cluster of the ectopic insulin-positive β cells coexpressing nuclear Ipf1 (p, green), nuclear Isl1 (q, red) and cell membranous Glut2 (r, green). (s,t) Electron micrographs (EM) of biliary epithelial cells in E12.5 Hes1−/− mice. These cells containing tight junctions (TJ) and microvilli (MV) had numerous secretory granules (t, arrowheads). The outlined area in s is magnified in t. CBD, common bile duct; Du, duodenum; Li, liver; Lu, lumen of the bile duct; N, nucleus; PV, main portal vein. Scale bars: a–n,100 μm; p–r, 10 μm; s, 3 μm.
The phenotypic conversion from biliary to pancreatic development in the Hes1 mutant mice prompted us to examine the expression of Hes1 and Neurog3 (encoding neurogenin 3) in the developing biliary system to see whether the pancreatic differentiation program5,6,7,8 operates ectopically in the mutant. In situ hybridization showed that Hes1 was expressed in the epithelial cells of the extrahepatic biliary system throughout normal cholangiogenesis (Fig. 5a–c). In addition, in Hes1−/− embryos, Neurog3 was highly expressed ectopically in the developing biliary epithelium (Fig. 5f,g), with a pattern similar to that seen in the normal pancreas. In wild-type littermates, the Neurog3 signal was confined to the pancreas and not detected in the biliary epithelium (Fig. 5d,e). Taken together, these findings suggest that the pancreatic endocrine differentiation program of the biliary epithelium in Hes1−/− mice is triggered by the ectopic expression of Neurog3, which acts as a proendocrine gene to specify cell fate12,13.
(a–c) Hes1 is continuously expressed from the budding primordium of the common bile duct (a, arrow) to the epithelia of the extrahepatic biliary system. (d–g) Ectopic Neurog3 expression of Hes1−/− embryos in the primordium of the bile duct (f, arrowhead), the abnormally aggregated biliary epithelial cells (f, red asterisk; see also Fig. 2e) and the remnant of the bile duct (g, red arrowhead). In Hes1+/+ littermates, Neurog3 is never detected in the biliary system, despite strong positive signals in the budding (d, arrow) and the mature ventral pancreas (Pa; e). CBD, common bile duct; CD, cystic duct; Du, duodenum; GB, gallbladder; HD, hepatic diverticulum; Li, liver; PV, main portal vein. Scale bars, 100 μm.
Our results show that endodermal Hes1 controls not only endodermal endocrine differentiation5,6 but also biliary organogenesis. Furthermore, they also indicate that the prospective biliary epithelium maintains the potential for pancreatic differentiation in the absence of Hes1. The ontogenetic relation between the common bile duct and the pancreas in sea lampreys supports the common evolutionary origin of these two organs. In this primitive vertebrate, adult endocrine pancreas tissue develops from the larval extrahepatic common bile duct through the process of transdifferentiation19. At a more general level, multipotency or plasticity, such as that seen in the biliary cells, may be a common characteristic of endoderm-derived organogenesis, similar to the conversion from pancreatic progenitors to duodenal cells that has recently been reported in Ptf1a-deficient mice20.
The development of endoderm-derived organs, including the pancreas and liver, is regulated by mesenchymal-epithelial cell interactions8,21,22,23. Expression of Ipf1 (ref. 24) in endodermal epithelial cells renders those cells responsive to mesenchyme-derived differentiation signals that induce pancreatic morphogenesis25. During mouse embryogenesis, Ipf1 was strongly expressed in the biliary system and the pancreas (Fig. 6). In addition, Hes1 represses Neurog3 (refs. 5,26), and we observed upregulation of expression of Neurog3 in bile ducts of Hes1-deficient mice (Fig. 5f,g). The facts suggest that Hes1 inhibits the pancreatic endocrine differentiation program in the biliary epithelium by repressing expression of Neurog3, probably by keeping the biliary epithelial cells unresponsive to mesenchyme-derived differentiation signals for the adjacent ventral pancreas. Some differentiation factors that are normally repressed by Hes1 may inhibit the development of pancreatic exocrine differentiation in the biliary system. Our finding that inactivation of Hes1 induces the conversion of biliary epithelium to pancreatic tissue may provide a substantial basis for future research concerning reconstitutional therapy of various endodermal cell types, including pancreatic β cells, using the abundant biliary cells.
Nuclear Ipf1 is detected in the gallbladder (GB), common bile duct (CBD) and the branching ventral pancreas (Pa) at E11.5 (see also Fig. 2b). Du, duodenum; Li, liver; PV, main portal vein. Scale bar, 100 μm.
Methods
Mice.
We used Hes1-deficient mice established on a CD1 genetic background2 and control CD1 mice in this study. We bred the mice on site and analyzed embryos at E9.5 to E17.5. We carried out experiments using protocols approved by the Laboratory Animal Resource Center, University of Tsukuba.
RT-PCR.
We used primers and reaction conditions as described for amplification27. We isolated the gallbladder, liver, pancreas and brain from CD1 embryos and neonatal mice with forceps under a microscope. We used brain cDNA and water as positive and negative controls, respectively. We carried out semiquantitative PCR for Notch-related molecules using quantitatively normalized samples as described27.
Histological analyses.
We carried out all histological analyses of the embryonic biliary system using serial sagittal sections, except for those shown in Figure 3f,l. To select a central longitudinal section of the biliary system, we completely sectioned whole embryos and stained every 3rd (E9.5) or every 20th (E17.5) section with hematoxylin and eosin. We then stained all adjacent sections with DBA or by immunohistochemistry or in situ hybridization.
We carried out DBA staining using DBA–horseradish peroxidase (EY Laboratories) as described15. For immunostaining, we used rabbit antibody to glucagon (DAKO), guinea pig antibody to insulin (DAKO), mouse antibody to insulin (sigma), rabbit antibody to somatostatin (NICHIREI), rabbit antibody to pancreatic peptide (DAKO), rabbit antibody to cytokeratin (DAKO), rabbit antibody to carboxypeptidase A (ANAWA), rabbit antibody to amylase (Biomeda), rabbit antibody to Ipf1 (CHEMICON), rabbit antibody to Glut2 (Alpha Diagnostic) and mouse antibody to Isl1 (clones 39.4D5 and 40.2D6, Development Studies Hybridoma Bank, University of Iowa). Immunohistochemical signals were detected with peroxidase-labeled Envision+ antibody to rabbit IgG (DAKO), diaminobenzidine and ammonium nickel sulfate. The immunofluorescent secondary antibodies used were Cy3-conjugated antibody to rabbit IgG (Jackson ImmunoResearch Laboratories) and Alexa488-conjugated antibody to mouse IgG (Molecular Probes). For TUNEL assays, we used an In situ Cell Death Detection Kit (Roche Molecular Biochemicals). We carried out in situ hybridization of Hes1 and Neurog3 using digoxigenin-labeled cRNA probes according to the reported protocol28.
References
Sasai, Y., Kageyama, R., Tagawa, Y., Shigemoto, R. & Nakanishi, S. Two mammalian helix-loop-helix factors structurally related to Drosophila hairy and Enhancer of split. Genes Dev. 6, 2620–2634 (1992).
Ishibashi, M. et al. Targeted disruption of mammalian hairy and Enhancer of split homolog-1 (HES-1) leads to up-regulation of neural helix-loop-helix factors, premature neurogenesis, and severe neural tube defects. Genes Dev. 9, 3136–3148 (1995).
Sommer, L., Ma, Q. & Anderson, D.J. Neurogenins, a novel family of atonal-related bHLH transcription factors, are putative mammalian neuronal determination genes that reveal progenitor cell heterogeneity in the developing CNS and PNS. Mol. Cell Neurosci. 8, 221–241 (1996).
Jarriault, S. et al. Signalling downstream of activated mammalian Notch. Nature 377, 355–358 (1995).
Jensen, J. et al. Control of endodermal endocrine development by Hes-1. Nat. Genet. 24, 36–44 (2000).
Skipper, M. & Lewis, J. Getting to the guts of enteroendocrine differentiation. Nat. Genet. 24, 3–4 (2000).
Apelqvist, A. et al. Notch signalling controls pancreatic cell differentiation. Nature 400, 877–881 (1999).
Edlund, H. Pancreatic organogenesis—developmental mechanisms and implications for therapy. Nat. Rev. Genet. 3, 524–532 (2002).
Li, L. et al. Alagille syndrome is caused by mutations in human Jagged1, which encodes a ligand for Notch1. Nat. Genet. 16, 243–251 (1997).
Oda, T. et al. Mutations in the human Jagged1 gene are responsible for Alagille syndrome. Nat. Genet. 16, 235–242 (1997).
Alagille, D., Odievre, M., Gautier, M. & Dommergues, J.P. Hepatic ductular hypoplasia associated with characteristic facies, vertebral malformations, retarded physical, mental, and sexual development, and cardiac murmur. J. Pediatr. 86, 63–71 (1975).
Gradwohl, G., Dierich, A., LeMeur, M. & Guillemot, F. Neurogenin3 is required for the development of the four endocrine cell lineages of the pancreas. Proc. Natl. Acad. Sci. USA 97, 1607–1611 (2000).
Schwitzgebel, V.M. et al. Expression of neurogenin3 reveals an islet cell precursor population in the pancreas. Development 127, 3533–3542 (2000).
Ohtsuka, T. et al. Hes1 and Hes5 as notch effectors in mammalian neuronal differentiation. EMBO J. 18, 2196–2207 (1999).
Shiojiri, N. & Katayama, H. Development of Dolichos biflorus agglutinin (DBA) binding sites in the bile duct of the embryonic mouse liver. Anat. Embryol. (Berl.) 178, 15–20 (1988).
Pang, K., Mukonoweshuro, C. & Wong, G.G. Beta cells arise from glucose transporter type 2 (Glut2)-expressing epithelial cells of the developing rat pancreas. Proc. Natl. Acad. Sci. USA 91, 9559–9563 (1994).
Herrera, P.L. Adult insulin- and glucagon-producing cells differentiate from two independent cell lineages. Development 127, 2317–2322 (2000).
Ahlgren, U., Pfaff, S.L., Jessell, T.M., Edlund, T. & Edlund, H. Independent requirement for ISL1 in formation of pancreatic mesenchyme and islet cells. Nature 385, 257–260 (1997).
Elliott, W.M. & Youson, J.H. Development of the adult endocrine pancreas during metamorphosis in the sea lamprey, Petromyzon marinus L. II. Electron microscopy and immunocytochemistry. Anat. Rec. 237, 271–290 (1993).
Kawaguchi, Y. et al. The role of the transcriptional regulator Ptf1a in converting intestinal to pancreatic progenitors. Nat. Genet. 32, 128–134 (2002).
Golosow, N. & Grobstein, C. Epitheliomesenchymal interaction in pancreatic morphogenesis. Dev. Biol. 4, 242–255 (1962).
Kim, S.K. & Hebrok, M. Intercellular signals regulating pancreas development and function. Genes Dev. 15, 111–127 (2001).
Zaret, K.S. Hepatocyte differentiation: from the endoderm and beyond. Curr. Opin. Genet. Dev. 11, 568–574 (2001).
Ohlsson, H., Karlsson, K. & Edlund, T. IPF1, a homeodomain-containing transactivator of the insulin gene. EMBO J. 12, 4251–4259 (1993).
Ahlgren, U., Jonsson, J. & Edlund, H. The morphogenesis of the pancreatic mesenchyme is uncoupled from that of the pancreatic epithelium in IPF1/PDX1-deficient mice. Development 122, 1409–1416 (1996).
Lee, J.C. et al. Regulation of the pancreatic pro-endocrine gene Neurogenin3. Diabetes 50, 928–936 (2001).
Kaneta, M. et al. A role for Pref-1 and HES-1 in thymocyte development. J. Immunol. 164, 256–264 (2000).
Tomita, K., Moriyoshi, K., Nakanishi, S., Guillemot, F. & Kageyama, R. Mammalian achaete-scute and atonal homologs regulate neuronal versus glial fate determination in the central nervous system. EMBO J. 19, 5460–5472 (2000).
Acknowledgements
We thank M. Nagata, M. Kobayashi, S. Takahashi, Y. Kawachi, T. Yamaoka and M. Itakura for critical discussions and T. Ohto, S. Itoh, N. Kajiwara and R. Hirochika for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Sumazaki, R., Shiojiri, N., Isoyama, S. et al. Conversion of biliary system to pancreatic tissue in Hes1-deficient mice. Nat Genet 36, 83–87 (2004). https://doi.org/10.1038/ng1273
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1273
This article is cited by
-
Biliary atresia: the development, pathological features, and classification of the bile duct
Pediatric Surgery International (2024)
-
Comparative study on a novel lobule structure of the zebrafish liver and that of the mammalian liver
Cell and Tissue Research (2022)
-
Engineering human hepato-biliary-pancreatic organoids from pluripotent stem cells
Nature Protocols (2021)
-
Porcine models for studying complications and organ crosstalk in diabetes mellitus
Cell and Tissue Research (2020)
-
A morphogenetic EphB/EphrinB code controls hepatopancreatic duct formation
Nature Communications (2019)