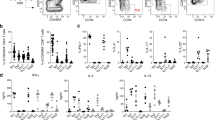
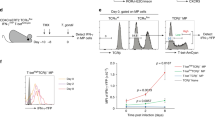
Abstract
Dendritic cells can prime naive CD4+ T cells; however, here we demonstrate that dendritic cell–mediated priming was insufficient for the development of T helper type 2 cell–dependent immunity. We identify basophils as a dominant cell population that coexpressed major histocompatibility complex class II and interleukin 4 message after helminth infection. Basophilia was promoted by thymic stromal lymphopoietin, and depletion of basophils impaired immunity to helminth infection. Basophils promoted antigen-specific CD4+ T cell proliferation and interleukin 4 production in vitro, and transfer of basophils augmented the population expansion of helminth-responsive CD4+ T cells in vivo. Collectively, our studies suggest that major histocompatibility complex class II–dependent interactions between basophils and CD4+ T cells promote T helper type 2 cytokine responses and immunity to helminth infection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Mosmann, T.R., Cherwinski, H., Bond, M.W., Giedlin, M.A. & Coffman, R.L. Two types of murine helper T cell clone. I. Definition according to profiles of lymphokine activities and secreted proteins. J. Immunol. 136, 2348–2357 (1986).
Reiner, S.L. Development in motion: helper T cells at work. Cell 129, 33–36 (2007).
Mowen, K.A. & Glimcher, L.H. Signaling pathways in Th2 development. Immunol. Rev. 202, 203–222 (2004).
Anthony, R.M., Rutitzky, L.I., Urban, J.F., Jr., Stadecker, M.J. & Gause, W.C. Protective immune mechanisms in helminth infection. Nat. Rev. Immunol. 7, 975–987 (2007).
Cohn, L., Elias, J.A. & Chupp, G.L. Asthma: mechanisms of disease persistence and progression. Annu. Rev. Immunol. 22, 789–815 (2004).
Perrigoue, J.G., Marshall, F.A. & Artis, D. On the hunt for helminths: innate immune cells in the recognition and response to helminth parasites. Cell. Microbiol. 10, 1757–1764 (2008).
Medzhitov, R. Recognition of microorganisms and activation of the immune response. Nature 449, 819–826 (2007).
Kapsenberg, M.L. Dendritic-cell control of pathogen-driven T-cell polarization. Nat. Rev. Immunol. 3, 984–993 (2003).
MacDonald, A.S. & Maizels, R.M. Alarming dendritic cells for Th2 induction. J. Exp. Med. 205, 13–17 (2008).
Amsen, D. et al. Instruction of distinct CD4 T helper cell fates by different notch ligands on antigen-presenting cells. Cell 117, 515–526 (2004).
MacDonald, A.S., Straw, A.D., Dalton, N.M. & Pearce, E.J. Cutting edge: Th2 response induction by dendritic cells: a role for CD40. J. Immunol. 168, 537–540 (2002).
Ekkens, M.J. et al. The role of OX40 ligand interactions in the development of the Th2 response to the gastrointestinal nematode parasite Heligmosomoides polygyrus. J. Immunol. 170, 384–393 (2003).
Gessner, A., Mohrs, K. & Mohrs, M. Mast cells, basophils, and eosinophils acquire constitutive IL-4 and IL-13 transcripts during lineage differentiation that are sufficient for rapid cytokine production. J. Immunol. 174, 1063–1072 (2005).
Mohrs, K., Wakil, A.E., Killeen, N., Locksley, R.M. & Mohrs, M. A two-step process for cytokine production revealed by IL-4 dual-reporter mice. Immunity 23, 419–429 (2005).
Min, B. et al. Basophils produce IL-4 and accumulate in tissues after infection with a Th2-inducing parasite. J. Exp. Med. 200, 507–517 (2004).
Skokos, D. et al. Mast cell-derived exosomes induce phenotypic and functional maturation of dendritic cells and elicit specific immune responses in vivo. J. Immunol. 170, 3037–3045 (2003).
MacKenzie, J.R., Mattes, J., Dent, L.A. & Foster, P.S. Eosinophils promote allergic disease of the lung by regulating CD4+ Th2 lymphocyte function. J. Immunol. 167, 3146–3155 (2001).
Padigel, U.M. et al. Eosinophils act as antigen-presenting cells to induce immunity to Strongyloides stercoralis in mice. J. Infect. Dis. 196, 1844–1851 (2007).
Voehringer, D., Reese, T.A., Huang, X., Shinkai, K. & Locksley, R.M. Type 2 immunity is controlled by IL-4/IL-13 expression in hematopoietic non-eosinophil cells of the innate immune system. J. Exp. Med. 203, 1435–1446 (2006).
Ohnmacht, C. & Voehringer, D. Basophil effector function and homeostasis during helminth infection. Blood 113, 2816–2825 (2008).
Voehringer, D., Shinkai, K. & Locksley, R.M. Type 2 immunity reflects orchestrated recruitment of cells committed to IL-4 production. Immunity 20, 267–277 (2004).
Sokol, C.L., Barton, G.M., Farr, A.G. & Medzhitov, R. A mechanism for the initiation of allergen-induced T helper type 2 responses. Nat. Immunol. 9, 310–318 (2008).
Else, K.J. & Finkelman, F.D. Intestinal nematode parasites, cytokines and effector mechanisms. Int. J. Parasitol. 28, 1145–1158 (1998).
Cliffe, L.J. & Grencis, R.K. The Trichuris muris system: a paradigm of resistance and susceptibility to intestinal nematode infection. Adv. Parasitol. 57, 255–307 (2004).
Else, K.J., Finkelman, F.D., Maliszewski, C.R. & Grencis, R.K. Cytokine-mediated regulation of chronic intestinal helminth infection. J. Exp. Med. 179, 347–351 (1994).
Lemos, M.P., Fan, L., Lo, D. & Laufer, T.M. CD8α+ and CD11b+ dendritic cell-restricted MHC class II controls Th1 CD4+ T cell immunity. J. Immunol. 171, 5077–5084 (2003).
Zaph, C. et al. Epithelial-cell-intrinsic IKK-β expression regulates intestinal immune homeostasis. Nature 446, 552–556 (2007).
Artis, D. et al. RELMβ/FIZZ2 is a goblet cell-specific immune-effector molecule in the gastrointestinal tract. Proc. Natl. Acad. Sci. USA 101, 13596–13600 (2004).
Artis, D. Epithelial-cell recognition of commensal bacteria and maintenance of immune homeostasis in the gut. Nat. Rev. Immunol. 8, 411–420 (2008).
Else, K.J. & Grencis, R.K. Antibody-independent effector mechanisms in resistance to the intestinal nematode parasite Trichuris muris. Infect. Immun. 64, 2950–2954 (1996).
Zaph, C. et al. Persistence and function of central and effector memory CD4+ T cells following infection with a gastrointestinal helminth. J. Immunol. 177, 511–518 (2006).
Mohrs, M., Shinkai, K., Mohrs, K. & Locksley, R.M. Analysis of type 2 immunity in vivo with a bicistronic IL-4 reporter. Immunity 15, 303–311 (2001).
Denzel, A. et al. Basophils enhance immunological memory responses. Nat. Immunol. 9, 733–742 (2008).
Owyang, A.M. et al. Interleukin 25 regulates type 2 cytokine-dependent immunity and limits chronic inflammation in the gastrointestinal tract. J. Exp. Med. 203, 843–849 (2006).
Humphreys, N.E., Xu, D., Hepworth, M.R., Liew, F.Y. & Grencis, R.K. IL-33, a potent inducer of adaptive immunity to intestinal nematodes. J. Immunol. 180, 2443–2449 (2008).
Suzukawa, M. et al. An IL-1 cytokine member, IL-33, induces human basophil activation via its ST2 receptor. J. Immunol. 181, 5981–5989 (2008).
Pecaric-Petkovic, T., Didichenko, S.A., Kaempfer, S., Spiegl, N. & Dahinden, C.A. Human basophils and eosinophils are the direct target leukocytes of the novel IL-1 family member IL-33. Blood 113, 1526–1534 (2009).
Smithgall, M.D. et al. IL-33 amplifies both Th1- and Th2-type responses through its activity on human basophils, allergen-reactive Th2 cells, iNKT and NK cells. Int. Immunol. 20, 1019–1030 (2008).
Fallon, P.G. et al. Identification of an interleukin (IL)-25-dependent cell population that provides IL-4, IL-5, and IL-13 at the onset of helminth expulsion. J. Exp. Med. 203, 1105–1116 (2006).
Taylor, J.J., Mohrs, M. & Pearce, E.J. Regulatory T cell responses develop in parallel to Th responses and control the magnitude and phenotype of the Th effector population. J. Immunol. 176, 5839–5847 (2006).
Allenspach, E.J., Lemos, M.P., Porrett, P.M., Turka, L.A. & Laufer, T.M. Migratory and lymphoid-resident dendritic cells cooperate to efficiently prime naive CD4 T cells. Immunity 29, 795–806 (2008).
Ehrlich, P. Beitrage zur Kenntins der granulierten Bindegewebs zellen und der eosinophilen Leukocythen. Arch Anat Physiol Lpz 3, 166–169 (1879).
Oh, K., Shen, T., Le Gros, G. & Min, B. Induction of Th2 type immunity in a mouse system reveals a novel immunoregulatory role of basophils. Blood 109, 2921–2927 (2007).
Gauchat, J.F. et al. Induction of human IgE synthesis in B cells by mast cells and basophils. Nature 365, 340–343 (1993).
Obata, K. et al. Basophils are essential initiators of a novel type of chronic allergic inflammation. Blood 110, 913–920 (2007).
Min, B., Le Gros, G. & Paul, W.E. Basophils: a potential liaison between innate and adaptive immunity. Allergol. Int. 55, 99–104 (2006).
Scheu, S. et al. Activation of the integrated stress response during T helper cell differentiation. Nat. Immunol. 7, 644–651 (2006).
Brown, S.J., Galli, S.J., Gleich, G.J. & Askenase, P.W. Ablation of immunity to Amblyomma americanum by anti-basophil serum: cooperation between basophils and eosinophils in expression of immunity to ectoparasites (ticks) in guinea pigs. J. Immunol. 129, 790–796 (1982).
Shen, T. et al. T cell-derived IL-3 plays key role in parasite infection-induced basophil production but is dispensable for in vivo basophil survival. Int. Immunol. 20, 1201–1209 (2008).
Taylor, B.C. et al. TSLP regulates intestinal immunity and inflammation in mouse models of helminth infection and colitis. J. Exp. Med. 206, 655–667 (2009).
Yoo, J. et al. Spontaneous atopic dermatitis in mice expressing an inducible thymic stromal lymphopoietin transgene specifically in the skin. J. Exp. Med. 202, 541–549 (2005).
Zhou, B. et al. Thymic stromal lymphopoietin as a key initiator of allergic airway inflammation in mice. Nat. Immunol. 6, 1047–1053 (2005).
Schmitz, J. et al. IL-33, an interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity 23, 479–490 (2005).
Angkasekwinai, P. et al. Interleukin 25 promotes the initiation of proallergic type 2 responses. J. Exp. Med. 204, 1509–1517 (2007).
Allakhverdi, Z., Smith, D.E., Comeau, M.R. & Delespesse, G. Cutting edge: the ST2 ligand IL-33 potently activates and drives maturation of human mast cells. J. Immunol. 179, 2051–2054 (2007).
Saenz, S.A., Taylor, B.C. & Artis, D. Welcome to the neighborhood: epithelial cell-derived cytokines license innate and adaptive immune responses at mucosal sites. Immunol. Rev. 226, 172–190 (2008).
Falcone, F.H., Pritchard, D.I. & Gibbs, B.F. Do basophils play a role in immunity against parasites? Trends Parasitol. 17, 126–129 (2001).
Schramm, G. et al. Cutting edge: IPSE/α-1, a glycoprotein from Schistosoma mansoni eggs, induces IgE-dependent, antigen-independent IL-4 production by murine basophils in vivo. J. Immunol. 178, 6023–6027 (2007).
Van Rooijen, N. The liposome-mediated macrophage 'suicide' technique. J. Immunol. Methods 124, 1–6 (1989).
Acknowledgements
We thank M. Mohrs (Trudeau Institute) for 4get mice; A. Troy for critical reading of this manuscript; E. Tait for generating the bone marrow chimeras; and the University of Pennsylvania Flow Cytometry Core and Center for Molecular Studies in Digestive and Liver Diseases Morphology Core for assistance with sorting and immunofluorescence staining. Supported by the National Institutes of Health (Artis lab: AI61570 and AI074878 to D.A., T32 training grant AI007532-08 to J.G.P., F31 training grant GM082187 to S.A.S. and T32 CA09140-30 to B.C.T.; Pearce lab: AI32573 and AI53825; and Laufer lab), the Burroughs Wellcome Fund (Artis lab: D.A.), the National Institute of Diabetes and Digestive Kidney Diseases (Artis lab: DK50306), the Crohn's and Colitis Foundation of America (Artis lab: D.A. and M.G.N.) and the University of Pennsylvania (Artis lab: D.A. and T.M.L.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
M.R.C. works for Amgen.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 (PDF 2443 kb)
Rights and permissions
About this article
Cite this article
Perrigoue, J., Saenz, S., Siracusa, M. et al. MHC class II–dependent basophil–CD4+ T cell interactions promote TH2 cytokine–dependent immunity. Nat Immunol 10, 697–705 (2009). https://doi.org/10.1038/ni.1740
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1740
This article is cited by
-
The association between eosinophils (CD16+ eosinophils), basophils (CD203+ basophils), and CD23 B lymphocytes in patients with atopic dermatitis on dupilumab therapy: pilot study
Dermatology and Therapy (2023)
-
Balamuthia mandrillaris trophozoites ingest human neuronal cells via a trogocytosis-independent mechanism
Parasites & Vectors (2022)
-
Whipworm and roundworm infections
Nature Reviews Disease Primers (2020)
-
Thymus-specific serine protease, a protease that shapes the CD4 T cell repertoire
Immunogenetics (2019)
-
Shuang-Huang-Lian prevents basophilic granulocyte activation to suppress Th2 immunity
BMC Complementary and Alternative Medicine (2018)