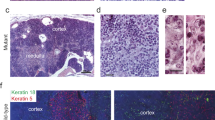
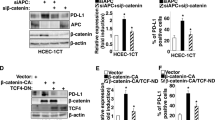
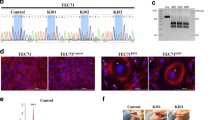
Abstract
Loss of the adenomatous polyposis coli (APC) protein is a common initiating event in colon cancer. Here we show that thymocyte-specific loss of APC deregulated β-catenin signaling and suppressed Notch-dependent transcription. These events promoted the proliferation of cells of the double-negative 3 and 4 stages and reduced rearrangements between the variable, diversity and joining regions of the gene encoding T cell receptor (TCR) β, encouraging developmental progression of aberrant thymocytes lacking pre-TCR and αβ TCR. Simultaneously, the loss of APC prolonged the mitotic metaphase-to-anaphase checkpoint and impaired chromosome segregation, blocking development beyond the double-negative 4 stage. The result was extensive thymic atrophy and increased frequencies of thymocytes with chromosomal abnormalities. Thus, loss of APC in immature thymocytes has consequences distinct from those of deregulation of β-catenin signaling and is essential for T cell differentiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
van de Wetering, M., de Lau, W. & Clevers, H. WNT signaling and lymphocyte development. Cell 109, S13–S19 (2002).
Morin, P.J. et al. Activation of β-catenin-Tcf signaling in colon cancer by mutations in β-catenin or Apc. Science 275, 1787–1790 (1997).
Smits, R. et al. Apc1638N: a mouse model for familial adenomatous polyposis-associated desmoid tumors and cutaneous cysts. Gastroenterology 114, 275–283 (1998).
Sparks, A.B., Morin, P.J., Vogelstein, B. & Kinzler, K.W. Mutational analysis of the APC/β-catenin/Tcf pathway in colorectal cancer. Cancer Res. 58, 1130–1134 (1998).
Miyoshi, Y. et al. Germ-line mutations of the Apc gene in 53 familial adenomatous polyposis patients. Proc. Natl. Acad. Sci. USA 89, 4452–4456 (1992).
Powell, S.M. et al. Molecular diagnosis of familial adenomatous polyposis. N. Engl. J. Med. 329, 1982–1987 (1993).
Miyaki, M. et al. Coexistence of somatic and germ-line mutations of APC gene in desmoid tumors from patients with familial adenomatous polyposis. Cancer Res. 53, 5079–5082 (1993).
Korinek, V. et al. Constitutive transcriptional activation by a β-catenin-Tcf complex in APC−/− colon carcinoma. Science 275, 1784–1787 (1997).
Shih, I.M. et al. Top-down morphogenesis of colorectal tumors. Proc. Natl. Acad. Sci. USA 98, 2640–2645 (2001).
Kaplan, K.B. et al. A role for the Adenomatous Polyposis Coli protein in chromosome segregation. Nat. Cell Biol. 3, 429–432 (2001).
Fodde, R. et al. Mutations in the APC tumour suppressor gene cause chromosomal instability. Nat. Cell Biol. 3, 433–438 (2001).
Martin, C.H. et al. Efficient thymic immigration of B220+ lymphoid-restricted bone marrow cells with T precursor potential. Nat. Immunol. 4, 866–873 (2003).
Shortman, K. & Wu, L. Early T lymphocyte progenitors. Annu. Rev. Immunol. 14, 29–47 (1996).
Allman, D. et al. Thymopoiesis independent of common lymphoid progenitors. Nat. Immunol. 4, 168–174 (2003).
Schwarz, B.A. & Bhandoola, A. Circulating hematopoietic progenitors with T lineage potential. Nat. Immunol. 5, 953–960 (2004).
Kondo, M., Weissman, I.L. & Akashi, K. Identification of clonogenic common lymphoid progenitors in mouse bone marrow. Cell 91, 661–672 (1997).
Porritt, H.E. et al. Heterogeneity among DN1 prothymocytes reveals multiple progenitors with different capacities to generate T cell and non-T cell lineages. Immunity 20, 735–745 (2004).
Radtke, F. et al. Deficient T cell fate specification in mice with an induced inactivation of Notch1. Immunity 10, 547–558 (1999).
Schmitt, T.M. & Zuniga-Pflucker, J.C. Induction of T cell development from hematopoietic progenitor cells by delta-like-1 in vitro. Immunity 17, 749–756 (2002).
von Boehmer, H. Aspects of lymphocyte developmental biology. Immunol. Today 18, 260–262 (1997).
Wolfer, A., Wilson, A., Nemir, M., MacDonald, H.R. & Radtke, F. Inactivation of Notch1 impairs VDJβ rearrangement and allows pre-TCR-independent survival of early αβ Lineage Thymocytes. Immunity 16, 869–879 (2002).
Schilham, M.W. et al. Critical involvement of Tcf-1 in expansion of thymocytes. J. Immunol. 161, 3984–3991 (1998).
Ioannidis, V., Beermann, F., Clevers, H. & Held, W. The β-catenin–TCF-1 pathway ensures CD4+CD8+ thymocyte survival. Nat. Immunol. 2, 691–697 (2001).
Okamura, R.M. et al. Redundant regulation of T cell differentiation and TCRα gene expression by the transcription factors LEF-1 and TCF-1. Immunity 8, 11–20 (1998).
Xu, Y., Banerjee, D., Huelsken, J., Birchmeier, W. & Sen, J.M. Deletion of β-catenin impairs T cell development. Nat. Immunol. 4, 1177–1182 (2003).
Gounari, F. et al. Somatic activation of β-catenin bypasses pre-TCR signaling and TCR selection in thymocyte development. Nat. Immunol. 2, 863–869 (2001).
Fodde, R. et al. A targeted chain-termination mutation in the mouse APC gene results in multiple intestinal tumors. Proc. Natl. Acad. Sci. USA 91, 8969–8973 (1994).
Moser, A.R., Pitot, H.C. & Dove, W.F. A dominant mutation that predisposes to multiple intestinal neoplasia in the mouse. Science 247, 322–324 (1990).
Oshima, M. et al. Loss of Apc heterozygosity and abnormal tissue building in nascent intestinal polyps in mice carrying a truncated Apc gene. Proc. Natl. Acad. Sci. USA 92, 4482–4486 (1995).
Harada, N. et al. Intestinal polyposis in mice with a dominant stable mutation of the β-catenin gene. EMBO J. 18, 5931–5942 (1999).
Lee, P.P. et al. A critical role for Dnmt1 and DNA methylation in T cell development, function, and survival. Immunity 15, 763–774 (2001).
Moore, N.C., Anderson, G., Williams, G.T., Owen, J.J. & Jenkinson, E.J. Developmental regulation of bcl-2 expression in the thymus. Immunology 81, 115–119 (1994).
Anderson, S.J., Abraham, K.M., Nakayama, T., Singer, A. & Perlmutter, R.M. Inhibition of T-cell receptor β-chain gene rearrangement by overexpression of the non-receptor protein tyrosine kinase p56lck. EMBO J. 11, 4877–4886 (1992).
Duncan, A.W. et al. Integration of Notch and Wnt signaling in hematopoietic stem cell maintenance. Nat. Immunol. 6, 314–322 (2005).
Galceran, J., Sustmann, C., Hsu, S.C., Folberth, S. & Grosschedl, R. LEF1-mediated regulation of Delta-like1 links Wnt and Notch signaling in somitogenesis. Genes Dev. 18, 2718–2723 (2004).
Nicolas, M. et al. Notch1 functions as a tumor suppressor in mouse skin. Nat. Genet. 33, 416–421 (2003).
Martinez Arias, A. Interactions between Wingless and Notch during the assignment of cell fates in Drosophila. Int. J. Dev. Biol. 42, 325–333 (1998).
Sicinska, E. et al. Requirement for cyclin D3 in lymphocyte development and T cell leukemias. Cancer Cell 4, 451–461 (2003).
Aoki, K., Tamai, Y., Horiike, S., Oshima, M. & Taketo, M.M. Colonic polyposis caused by mTOR-mediated chromosomal instability in Apc+/Δ716Cdx2+/− compound mutant mice. Nat. Genet. 35, 323–330 (2003).
McDougall, S., Peterson, C.L. & Calame, K. A transcriptional enhancer 3′ of C β 2 in the T cell receptor β locus. Science 241, 205–208 (1988).
Su, L.K. et al. APC binds to the novel protein EB1. Cancer Res. 55, 2972–2977 (1995).
Zumbrunn, J., Kinoshita, K., Hyman, A.A. & Nathke, I.S. Binding of the adenomatous polyposis coli protein to microtubules increases microtubule stability and is regulated by GSK3 β phosphorylation. Curr. Biol. 11, 44–49 (2001).
Cleveland, D.W., Mao, Y. & Sullivan, K.F. Centromeres and kinetochores: from epigenetics to mitotic checkpoint signaling. Cell 112, 407–421 (2003).
Rieder, C.L., Schultz, A., Cole, R. & Sluder, G. Anaphase onset in vertebrate somatic cells is controlled by a checkpoint that monitors sister kinetochore attachment to the spindle. J. Cell Biol. 127, 1301–1310 (1994).
Li, X. & Nicklas, R.B. Mitotic forces control a cell-cycle checkpoint. Nature 373, 630–632 (1995).
Fehling, H.J., Krotkova, A., Saint-Ruf, C. & von Boehmer, H. Crucial role of the pre-T-cell receptor α gene in development of αβ but not γδ T cells. Nature 375, 795–798 (1995).
Hoffman, E.S. et al. Productive T-cell receptor β-chain gene rearrangement: coincident regulation of cell cycle and clonality during development in vivo. Genes Dev. 10, 948–962 (1996).
Ciofani, M. et al. Obligatory role for cooperative signaling by pre-TCR and Notch during thymocyte differentiation. J. Immunol. 172, 5230–5239 (2004).
Gounari, F. et al. Tracing lymphopoiesis with the aid of a pTα-controlled reporter gene. Nat. Immunol. 3, 489–496 (2002).
Hodgkin, P.D. et al. The importance of efficacy and partial agonism in evaluating models of B lymphocyte activation. Int. Rev. Immunol. 15, 101–127 (1997).
Acknowledgements
The authors thank J.C. Zúñiga-Pflücker (University of Toronto, Toronto, Canada) for advice, support and the OP9-DL1 cells; I. Khan for help with the OP9-DL1 cultures; K. Georgopoulos (Massachusetts General Hospital), P. Sicinski and R. Van Etten for critical comments on the manuscript; I. Aifantis (University of Chicago), X. Li and A. Campese for the NotchIC retrovirus; and H. von Boehmer and R.M. Meyer for their interest and support. Supported by the National Institutes of Health (R01 AI059676-01 to F.G. and R01 CA104547-01A1 to K.K.), the Medical Foundation (Smith Family New Investigator Award to F.G.), the Dana Farber Cancer Institute (National Colorectal Cancer Research Alliance Award to K.K.) and the Claudia Adams Barr Program (K.K.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Detection of mitotic abnormalities in DN LckCre-APClox/loxD468 thymocytes. (PDF 570 kb)
Rights and permissions
About this article
Cite this article
Gounari, F., Chang, R., Cowan, J. et al. Loss of adenomatous polyposis coli gene function disrupts thymic development. Nat Immunol 6, 800–809 (2005). https://doi.org/10.1038/ni1228
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1228
This article is cited by
-
Wnt–β-catenin activation epigenetically reprograms Treg cells in inflammatory bowel disease and dysplastic progression
Nature Immunology (2021)
-
Delayed maturation of thymic epithelium in mice with specific deletion of β-catenin gene in FoxN1 positive cells
Histochemistry and Cell Biology (2021)
-
A new method for discovering EMAST sequences in animal models of cancer
Scientific Reports (2018)
-
Depletion of p21-activated kinase 1 up-regulates the immune system of APC∆14/+ mice and inhibits intestinal tumorigenesis
BMC Cancer (2017)
-
Inflammation and Colorectal Cancer
Current Colorectal Cancer Reports (2017)