Abstract
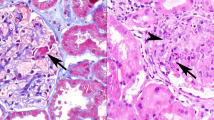
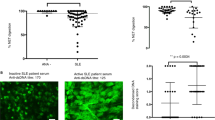
Pauci-immune focal necrotizing glomerulonephritis (FNGN) is a severe inflammatory disease associated with autoantibodies to neutrophil cytoplasmic antigens (ANCA). Here we characterize autoantibodies to lysosomal membrane protein-2 (LAMP-2) and show that they are a new ANCA subtype present in almost all individuals with FNGN. Consequently, its prevalence is nearly twice that of the classical ANCAs that recognize myeloperoxidase or proteinase-3. Furthermore, antibodies to LAMP-2 cause pauci-immune FNGN when injected into rats, and a monoclonal antibody to human LAMP-2 (H4B4) induces apoptosis of human microvascular endothelium in vitro. The autoantibodies in individuals with pauci-immune FNGN commonly recognize a human LAMP-2 epitope (designated P41–49) with 100% homology to the bacterial adhesin FimH, with which they cross-react. Rats immunized with FimH develop pauci-immune FNGN and also develop antibodies to rat and human LAMP-2. Finally, we show that infections with fimbriated pathogens are common before the onset of FNGN. Thus, FimH-triggered autoimmunity to LAMP-2 provides a previously undescribed clinically relevant molecular mechanism for the development of pauci-immune FNGN.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Jennette, J.C. & Falk, R.J. Small-vessel vasculitis. N. Engl. J. Med. 337, 1512–1523 (1997).
Morgan, M.D., Harper, L., Williams, J. & Savage, C. Anti-neutrophil cytoplasm associated glomerulonephritis. J. Am. Soc. Nephrol. 17, 1224–1234 (2006).
Davies, D.J., Moran, J.E., Niall, J.F. & Ryan, G.B. Segmental necrotizing glomerulonephritis with antineutrophil antibody: possible arbovirus aetiology? Br. Med. J. (Clin. Res. Ed.) 285, 606 (1982).
van der Woude, F.J. et al. Autoantibodies against neutrophils and monocytes: tool for diagnosis and marker of disease activity in Wegener's granulomatosis. Lancet 1, 425–429 (1985).
Jennette, J.C., Xiao, H. & Falk, R.J. Pathogenesis of vascular inflammation by anti-neutrophil cytoplasmic antibodies. J. Am. Soc. Nephrol. 17, 1235–1242 (2006).
Xiao, H. et al. Antineutrophil cytoplasmic autoantibodies specific for myeloperoxidase cause glomerulonephritis and vasculitis in mice. J. Clin. Invest. 110, 955–963 (2002).
Huugen, D. et al. Aggravation of anti-myeloperoxidase antibody-induced glomerulonephritis by bacterial lipopolysaccharide: role of tumor necrosis factor-α. Am. J. Pathol. 167, 47–58 (2005).
Pfister, H. et al. Antineutrophil cytoplasmic autoantibodies against the murine homolog of proteinase 3 (Wegener autoantigen) are pathogenic in vivo. Blood 104, 1411–1418 (2004).
van der Geld, Y.M. et al. Rats and mice immunised with chimeric human/mouse proteinase 3 produce autoantibodies to mouse PR3 and rat granulocytes. Ann. Rheum. Dis. 66, 1679–1682 (2007).
Kain, R. et al. A novel class of autoantigens of anti-neutrophil cytoplasmic antibodies (ANCA) in necrotizing and crescentic glomerulonephritis. J. Exp. Med. 181, 585–597 (1995).
Carlsson, S.R., Roth, J., Piller, F. & Fukuda, M. Isolation and characterization of human lysosomal membrane glycoproteins, h-LAMP-1 and h-LAMP-2. Major sialoglycoproteins carrying polylactosaminoglycan. J. Biol. Chem. 263, 18911–18919 (1988).
Eskelinen, E.L. et al. Unifying nomenclature for the isoforms of the lysosomal membrane protein LAMP-2. Traffic 6, 1058–1061 (2005).
Sawada, R., Lowe, J.B. & Fukuda, M. E-selectin–dependent adhesion efficiency of colonic carcinoma cells is increased by genetic manipulation of their cell surface lysosomal membrane glycoprotein-1 expression levels. J. Biol. Chem. 268, 12675–12681 (1993).
Dice, J.F. Chaperone-mediated autophagy. Autophagy 3, 295–299 (2007).
Zhou, D. et al. Lamp-2a facilitates MHC class II presentation of cytoplasmic antigens. Immunity 22, 571–581 (2005).
Gough, N.R. & Fambrough, D.M. Different steady state subcellular distributions of the three splice variants of lysosome-associated membrane protein LAMP-2 are determined largely by the COOH-terminal amino acid residue. J. Cell Biol. 137, 1161–1169 (1997).
Wegener, F. Über generalisierte septische Gefäßerkrankungen. Verh. Dtsch. Pathol. Ges. 29, 202–209 (1936).
Pinching, A.J. et al. Relapses in Wegener's granulomatosis: the role of infection. BMJ 281, 836–838 (1980).
Stegeman, C.A., Tervaert, J.W., de Jong, P.E. & Kallenberg, C.G. Trimethoprim sulfamethoxazole (co-trimoxazole) for the prevention of relapses of Wegener's granulomatosis. Dutch Co-Trimoxazole Wegener Study Group. N. Engl. J. Med. 335, 16–20 (1996).
Wucherpfennig, K.W. Mechanisms for the induction of autoimmunity by infectious agents. J. Clin. Invest. 108, 1097–1104 (2001).
Fourneau, J.M., Bach, J.M., van Endert, P.M. & Bach, J.F. The elusive case for a role of mimicry in autoimmune diseases. Mol. Immunol. 40, 1095–1102 (2004).
Pendergraft, W.F., III et al. Autoimmunity is triggered by cPR-3105–201, a protein complementary to human autoantigen proteinase-3. Nat. Med. 10, 72–79 (2004).
Franssen, C.F. et al. In vitro neutrophil activation by antibodies to proteinase 3 and myeloperoxidase from patients with crescentic glomerulonephritis. J. Am. Soc. Nephrol. 10, 1506–1515 (1999).
Falk, R.J., Terrell, R.S., Charles, L.A. & Jennette, J.C. Anti-neutrophil cytoplasmic autoantibodies induce neutrophils to degranulate and produce oxygen radicals in vitro. Proc. Natl. Acad. Sci. USA 87, 4115–4119 (1990).
Haslett, C., Guthrie, L.A., Kopaniak, M.M., Johnston, R.B., Jr & Henson, P.M. Modulation of multiple neutrophil functions by preparative methods or trace concentrations of bacterial lipopolysaccharide. Am. J. Pathol. 119, 101–110 (1985).
Tse, W.Y., Nash, G.B., Hewins, P., Savage, C.O. & Adu, D. ANCA-induced neutrophil F-actin polymerization: implications for microvascular inflammation. Kidney Int. 67, 130–139 (2005).
Kriehuber, E. et al. Isolation and characterization of dermal lymphatic and blood endothelial cells reveal stable and functionally specialized cell lineages. J. Exp. Med. 194, 797–808 (2001).
Kerjaschki, D., Ullrich, R., Exner, M., Orlando, R.A. & Farquhar, M.G. Induction of passive Heymann nephritis with antibodies specific for a synthetic peptide derived from the receptor-associated protein. J. Exp. Med. 183, 2007–2015 (1996).
Granger, B.L. et al. Characterization and cloning of lgp110, a lysosomal membrane glycoprotein from mouse and rat cells. J. Biol. Chem. 265, 12036–12043 (1990).
Little, M.A. et al. Antineutrophil cytoplasm antibodies directed against myeloperoxidase augment leukocyte-microvascular interactions in vivo. Blood 106, 2050–2058 (2005).
Ruth, A.J. et al. Anti-neutrophil cytoplasmic antibodies and effector CD4+ cells play nonredundant roles in anti-myeloperoxidase crescentic glomerulonephritis. J. Am. Soc. Nephrol. 17, 1940–1949 (2006).
González-Polo, R.A. et al. The apoptosis/autophagy paradox: autophagic vacuolization before apoptotic death. J. Cell Sci. 118, 3091–3102 (2005).
Massey, A.C., Kaushik, S., Sovak, G., Kiffin, R. & Cuervo, A.M. Consequences of the selective blockage of chaperone-mediated autophagy. Proc. Natl. Acad. Sci. USA 103, 5805–5810 (2006).
Dengjel, J. et al. Autophagy promotes MHC class II presentation of peptides from intracellular source proteins. Proc. Natl. Acad. Sci. USA 102, 7922–7927 (2005).
Schmid, D., Pypaert, M. & Münz, C. Antigen-loading compartments for major histocompatibility complex class II molecules continuously receive input from autophagosomes. Immunity 26, 79–92 (2007).
Yuki, N. et al. Carbohydrate mimicry between human ganglioside GM1 and Campylobacter jejuni lipooligosaccharide causes Guillain-Barre syndrome. Proc. Natl. Acad. Sci. USA 101, 11404–11409 (2004).
Levin, M.C. et al. Autoimmunity due to molecular mimicry as a cause of neurological disease. Nat. Med. 8, 509–513 (2002).
Savige, J. et al. Antineutrophil cytoplasmic antibody (ANCA)-associated systemic vasculitis after immunisation with bacterial proteins. Clin. Exp. Rheumatol. 20, 783–789 (2002).
Connell, I. et al. Type 1 fimbrial expression enhances Escherichia coli virulence for the urinary tract. Proc. Natl. Acad. Sci. USA 93, 9827–9832 (1996).
Martinez, J.J., Mulvey, M.A., Schilling, J.D., Pinkner, J.S. & Hultgren, S.J. Type 1 pilus–mediated bacterial invasion of bladder epithelial cells. EMBO J. 19, 2803–2812 (2000).
Langermann, S. et al. Prevention of mucosal Escherichia coli infection by FimH-adhesin–based systemic vaccination. Science 276, 607–611 (1997).
Langermann, S. et al. Vaccination with FimH adhesin protects cynomolgus monkeys from colonization and infection by uropathogenic Escherichia coli. J. Infect. Dis. 181, 774–778 (2000).
Jennette, J.C. et al. Nomenclature of systemic vasculitides. Proposal of an international consensus conference. Arthritis Rheum. 37, 187–192 (1994).
Neumann, I. et al. Histological and clinical predictors of early and late renal outcome in ANCA-associated vasculitis. Nephrol. Dial. Transplant. 20, 96–104 (2005).
Skrincosky, D. et al. Altered Golgi localisation of Core 2 β-1,6-N-Acetylglucosaminyltransferase leads to decreased synthesis of branched O-glycans. J. Biol. Chem. 272, 22695–22702 (1997).
Kain, R., Angata, K., Kerjaschki, D. & Fukuda, M. Molecular cloning and expression of a novel human trans-Golgi network glycoprotein, TGN51, that contains multiple tyrosine-containing motifs. J. Biol. Chem. 273, 981–988 (1998).
Kozarsky, K., Kingsley, D. & Krieger, M. Use of a mutant cell line to study the kinetics and function of O-linked glycosylation of low density lipoprotein receptors. Proc. Natl. Acad. Sci. USA 85, 4335–4339 (1988).
Savige, J. et al. International group for consensus statement on testing and reporting of antineutrophil cytoplasmic antibodies (ANCA). Am. J. Clin. Pathol. 120, 312–318 (2003).
Schembri, M.A., Hasman, H. & Klemm, P. Expression and purification of the mannose recognition domain of the FimH adhesin. FEMS Microbiol. Lett. 188, 147–151 (2000).
Horvat, R., Hovorka, A., Dekan, G., Poczewski, H. & Kerjaschki, D. Endothelial cell membranes contain podocalyxin—the major sialoprotein of visceral glomerular epithelial cells. J. Cell Biol. 102, 484–491 (1986).
Acknowledgements
This paper is dedicated to the memory of Fokko van der Woude. We thank A. Jungbauer (University of Agriculture and Forestry, Vienna) for synthetic peptides. We thank Kidney Research UK, who generously funded R.K.'s Senior Research Fellowship (KRUK SF3/2000-2005) at the University of Aberdeen and Scottish Hospitals Endowment Research Trust (RG15/02) for supporting parts of this project. A.R. is funded by an EU Marie Curie Excellence Chair (MEXC-CT-2006-042742). Parts of this work were funded by the Austrian Federal Ministry of Science and Research. M.F.'s research is supported by grant RO1CA48737. We are indebted to H. Schachner for technical assistance and to A. Jäger for his help in preparing the figures. We also would like to thank the many physicians who provided sera and subject details.
Author information
Authors and Affiliations
Contributions
R.K.: design, execution, supervision and analysis of experiments, and manuscript writing. M.E.: development of human LAMP-2 ELISA and usage of SPOT assays. R.B.: cloning and generation of fusion proteins, western blots, human LAMP-2 and inhibition ELISA. R.Z.: collection and analysis of subject data. D.C.: in vivo experiments. C.A.L.: human LAMP-2 and inhibition ELISA of subject sera. A.D.: glycoepitope analysis. I.R.: experimental tissue culture work. R.J.: western blot analysis of antibody binding to bacterial proteins. O.A.: in vitro experiments with polymorphonuclear and endothelial cells. S.S.: myeloperoxidase and proteinase-3 ELISA. G.S.-P.: collection and analysis of subject data. M.F.: provision of human LAMP-2 complementary DNA constructs, antibodies and advice. P.K.: provision of FimH cDNA construct, antibodies and advice. A.J.R.: design and analysis of experiments and manuscript writing. D.K.: design and analysis of experiments and manuscript writing.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–4, Supplementary Tables 1–3 and Supplementary Methods (PDF 786 kb)
Rights and permissions
About this article
Cite this article
Kain, R., Exner, M., Brandes, R. et al. Molecular mimicry in pauci-immune focal necrotizing glomerulonephritis. Nat Med 14, 1088–1096 (2008). https://doi.org/10.1038/nm.1874
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.1874
This article is cited by
-
Nephritis-associated plasmin receptor (NAPlr)-positive glomerulonephritis in a case of ANCA-negative small vessel vasculitis
CEN Case Reports (2022)
-
Autoantibodies in neurological disease
Nature Reviews Immunology (2021)
-
Neuroimmunologie von COVID‑19
Der Nervenarzt (2021)
-
De novo pauci-immune glomerulonephritis in renal allografts
Modern Pathology (2020)
-
ANCA-Diagnostik bei Vaskulitiden
Zeitschrift für Rheumatologie (2020)