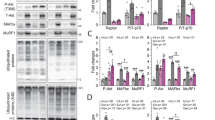
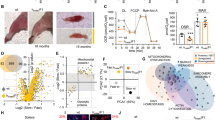
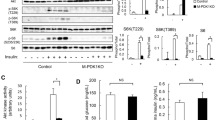
Abstract
Skeletal muscle atrophy occurs in aging and pathological conditions, including cancer, diabetes and AIDS1. Treatment of atrophy is based on either preventing protein-degradation pathways, which are activated during atrophy, or activating protein-synthesis pathways, which induce muscle hypertrophy2. Here we show that neuronal nitric oxide synthase (nNOS) regulates load-induced hypertrophy by activating transient receptor potential cation channel, subfamily V, member 1 (TRPV1). The overload-induced hypertrophy was prevented in nNOS-null mice. nNOS was transiently activated within 3 min after overload. This activation promoted formation of peroxynitrite, a reaction product of nitric oxide with superoxide3, which was derived from NADPH oxidase 4 (Nox4). Nitric oxide and peroxynitrite then activated Trpv1, resulting in an increase of intracellular Ca2+ concentration ([Ca2+]i) that subsequently triggered activation of mammalian target of rapamycin (mTOR). Notably, administration of the TRPV1 agonist capsaicin induced hypertrophy without overload and alleviated unloading- or denervation-induced atrophy. These findings identify nitric oxide, peroxynitrite and [Ca2+]i as the crucial mediators that convert a mechanical load into an intracellular signaling pathway and lead us to suggest that TRPV1 could be a new therapeutic target for treating muscle atrophy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lecker, S.H., Goldberg, A.L. & Mitch, W.E. Protein degradation by the ubiquitin-proteasome pathway in normal and disease states. J. Am. Soc. Nephrol. 17, 1807–1819 (2006).
Sandri, M. Signaling in muscle atrophy and hypertrophy. Physiology (Bethesda) 23, 160–170 (2008).
Heck, D.E. *NO, RSNO, ONOO-, NO+, *NOO, NOx–dynamic regulation of oxidant scavenging, nitric oxide stores, and cyclic GMP-independent cell signaling. Antioxid. Redox Signal. 3, 249–260 (2001).
Hoffman, E.P. & Nader, G.A. Balancing muscle hypertrophy and atrophy. Nat. Med. 10, 584–585 (2004).
Bodine, S.C. et al. Akt/mTOR pathway is a crucial regulator of skeletal muscle hypertrophy and can prevent muscle atrophy in vivo. Nat. Cell Biol. 3, 1014–1019 (2001).
Goodman, C.A. et al. A phosphatidylinositol 3-kinase/protein kinase B–independent activation of mammalian target of rapamycin signaling is sufficient to induce skeletal muscle hypertrophy. Mol. Biol. Cell 21, 3258–3268 (2010).
Hornberger, T.A. et al. Mechanical stimuli regulate rapamycin-sensitive signalling by a phosphoinositide 3-kinase–, protein kinase B– and growth factor–independent mechanism. Biochem. J. 380, 795–804 (2004).
Zoncu, R., Efeyan, A. & Sabatini, D.M. mTOR: from growth signal integration to cancer, diabetes and ageing. Nat. Rev. Mol. Cell Biol. 12, 21–35 (2011).
Ma, X.M. & Blenis, J. Molecular mechanisms of mTOR-mediated translational control. Nat. Rev. Mol. Cell Biol. 10, 307–318 (2009).
Brenman, J.E. et al. Interaction of nitric oxide synthase with the postsynaptic density protein PSD-95 and α1-syntrophin mediated by PDZ domains. Cell 84, 757–767 (1996).
Stamler, J.S. & Meissner, G. Physiology of nitric oxide in skeletal muscle. Physiol. Rev. 81, 209–237 (2001).
Asai, A. et al. Primary role of functional ischemia, quantitative evidence for the two-hit mechanism, and phosphodiesterase-5 inhibitor therapy in mouse muscular dystrophy. PLoS ONE 2, e806 (2007).
Wozniak, A.C. & Anderson, J.E. Nitric oxide–dependence of satellite stem cell activation and quiescence on normal skeletal muscle fibers. Dev. Dyn. 236, 240–250 (2007).
Yoshida, T. et al. Nitric oxide activates TRP channels by cysteine S-nitrosylation. Nat. Chem. Biol. 2, 596–607 (2006).
Adachi, T. et al. S-Glutathiolation by peroxynitrite activates SERCA during arterial relaxation by nitric oxide. Nat. Med. 10, 1200–1207 (2004).
Trebak, M., Ginnan, R., Singer, H.A. & Jourd′heuil, D. Interplay between calcium and reactive oxygen/nitrogen species: an essential paradigm for vascular smooth muscle signaling. Antioxid. Redox Signal. 12, 657–674 (2010).
Sellman, J.E. et al. In vivo inhibition of nitric oxide synthase impairs upregulation of contractile protein mRNA in overloaded plantaris muscle. J. Appl. Physiol. 100, 258–265 (2006).
Soltow, Q.A. et al. Ibuprofen inhibits skeletal muscle hypertrophy in rats. Med. Sci. Sports Exerc. 38, 840–846 (2006).
Kemp-Harper, B. & Feil, R. Meeting report: cGMP matters. Sci. Signal. 1, pe12 (2008).
Carnesecchi, S. et al. A key role for NOX4 in epithelial cell death during development of lung fibrosis. Antioxid. Redox Signal. 15, 607–619 (2011).
Bedard, K. & Krause, K.H. The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol. Rev. 87, 245–313 (2007).
Laleu, B. et al. First in class, potent, and orally bioavailable NADPH oxidase isoform 4 (Nox4) inhibitors for the treatment of idiopathic pulmonary fibrosis. J. Med. Chem. 53, 7715–7730 (2010).
Tallini, Y.N. et al. Imaging cellular signals in the heart in vivo: cardiac expression of the high-signal Ca2+ indicator GCaMP2. Proc. Natl. Acad. Sci. USA 103, 4753–4758 (2006).
Piétri-Rouxel, F. et al. DHPR α1S subunit controls skeletal muscle mass and morphogenesis. EMBO J. 29, 643–654 (2010).
Luo, Z. et al. TRPV1 activation improves exercise endurance and energy metabolism through PGC-1α upregulation in mice. Cell Res. 22, 551–564 (2012).
Xin, H. et al. Vanilloid receptor expressed in the sarcoplasmic reticulum of rat skeletal muscle. Biochem. Biophys. Res. Commun. 332, 756–762 (2005).
Suzuki, N. et al. NO production results in suspension-induced muscle atrophy through dislocation of neuronal NOS. J. Clin. Invest. 117, 2468–2476 (2007).
Gulati, P. et al. Amino acids activate mTOR complex 1 via Ca2+/CaM signaling to hVps34. Cell Metab. 7, 456–465 (2008).
Cunningham, J.T. et al. mTOR controls mitochondrial oxidative function through a YY1–PGC-1α transcriptional complex. Nature 450, 736–740 (2007).
Sandri, M. et al. PGC-1α protects skeletal muscle from atrophy by suppressing FoxO3 action and atrophy-specific gene transcription. Proc. Natl. Acad. Sci. USA 103, 16260–16265 (2006).
Aoki, Y. et al. In-frame dystrophin following exon 51–skipping improves muscle pathology and function in the exon 52–deficient mdx mouse. Mol. Ther. 18, 1995–2005 (2010).
Kuroda, J. et al. NADPH oxidase 4 (Nox4) is a major source of oxidative stress in the failing heart. Proc. Natl. Acad. Sci. USA 107, 15565–15570 (2010).
Whitehead, N.P., Yeung, E.W., Froehner, S.C. & Allen, D.G. Skeletal muscle NADPH oxidase is increased and triggers stretch-induced damage in the mdx mouse. PLoS ONE 5, e15354 (2010).
Ono, Y., Boldrin, L., Knopp, P., Morgan, J.E. & Zammit, P.S. Muscle satellite cells are a functionally heterogeneous population in both somite-derived and branchiomeric muscles. Dev. Biol. 337, 29–41 (2010).
Bakondi, E. et al. Role of intracellular calcium mobilization and cell-density–dependent signaling in oxidative-stress–induced cytotoxicity in HaCaT keratinocytes. J. Invest. Dermatol. 121, 88–95 (2003).
Acknowledgements
We thank M. Kotlikoff (Department of Biomedical Science, College of Veterinary Medicine, Cornell University, Ithaca, New York, USA) for providing pCAGGS-GCaMP2 mice, K.-H. Krause (Department of Pathology and Immunology, University of Geneva, Geneva, Switzerland) for Nox4-null mice, Y. Ono for the technical support to isolate satellite cells and GenKyoTex (Geneva, Switzerland) for supplying GKT136901. We also thank D. Glass, I. Kii, V. Ullrich, B. Gähwiler and Y. Aoki for reading the manuscript and contributing to valuable discussions. This work was supported by a Grant-in-Aid for Scientific Research (B), a Grant-in-Aid for Japan Society for the Promotion of Science Fellows and a grant from the Association Française contre les Myopathies (AFM, France).
Author information
Authors and Affiliations
Contributions
N.I. conceived and performed the experiments. A.K., Y.M.-S., U.T.R. and S.T. conducted and supervised the experiments. N.I. and U.T.R. designed the experiments and wrote the manuscript, and all authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 2441 kb)
Rights and permissions
About this article
Cite this article
Ito, N., Ruegg, U., Kudo, A. et al. Activation of calcium signaling through Trpv1 by nNOS and peroxynitrite as a key trigger of skeletal muscle hypertrophy. Nat Med 19, 101–106 (2013). https://doi.org/10.1038/nm.3019
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3019
This article is cited by
-
The emerging role of Piezo1 channels in skeletal muscle physiology
Biophysical Reviews (2023)
-
The role of periostin in cardiac fibrosis
Heart Failure Reviews (2023)
-
Myokine Response to Blood-Flow Restricted Resistance Exercise in Younger and Older Males in an Untrained and Resistance-Trained State: A Pilot Study
Journal of Science in Sport and Exercise (2023)
-
Differences in muscle satellite cell dynamics during muscle hypertrophy and regeneration
Skeletal Muscle (2022)
-
Disturbance of calcium homeostasis and myogenesis caused by TET2 deletion in muscle stem cells
Cell Death Discovery (2022)