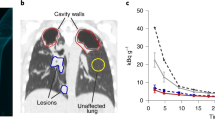
Abstract
The absence of a gold standard to determine when antibiotics induce a sterilizing cure has confounded the development of new approaches to treat pulmonary tuberculosis (PTB). We detected positron emission tomography and computerized tomography (PET–CT) imaging response patterns consistent with active disease, along with the presence of Mycobacterium tuberculosis (MTB) mRNA in sputum and bronchoalveolar lavage samples, in a substantial proportion of adult, HIV-negative patients with PTB after a standard 6-month treatment plus 1 year follow-up, including patients with a durable cure and others who later developed recurrent disease. The presence of MTB mRNA in the context of nonresolving and intensifying lesions on PET–CT images might indicate ongoing transcription, suggesting that even apparently curative treatment for PTB may not eradicate all of the MTB bacteria in most patients. This suggests an important complementary role for the immune response in maintaining a disease-free state. Sterilizing drugs or host-directed therapies, and better treatment response markers, are probably needed for the successful development of improved and shortened PTB-treatment strategies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
10 November 2017
In the version of this article initially published, two authors in the Catalysis TB–Biomarker Consortium were incorrectly identified as Lani Theart and Coenie Kogelenberg. The authors’ names are Lani Thiart and Coenie Koegelenberg. Also, in Figure 3c, the three patients who initiated TB retreatment after EOT + 1y had ‘mixed’ response patterns at EOT + 1y, as is written in the text, and not ‘resolved’ response patterns, as is shown in the original version of the figure. The errors have been corrected in the HTML and PDF versions of the article.
19 October 2016
In the version of this article initially published, one of the authors of the Catalysis TB–Biomarker Consortium was incorrectly identified as Nelita Burger. The author's name is Nelita Du Plessis. The error has been corrected in the HTML and PDF versions of the article.
References
Cox, H.S., Morrow, M. & Deutschmann, P.W. Long term efficacy of DOTS regimens for tuberculosis: systematic review. Br. Med. J. 336, 484–487 (2008).
World Health Organization. Global tuberculosis report 2015 (WHO Press, Geneva, 2015).
Gillespie, S.H. et al. Four-month moxifloxacin-based regimens for drug-sensitive tuberculosis. N. Engl. J. Med. 371, 1577–1587 (2014).
Jindani, A. et al. High-dose rifapentine with moxifloxacin for pulmonary tuberculosis. N. Engl. J. Med. 371, 1599–1608 (2014).
Luzze, H. et al. Relapse more common than re-infection in recurrent tuberculosis 1–2 years post treatment in urban Uganda. Int. J. Tuberc. Lung Dis. 17, 361–367 (2013).
Merle, C.S. et al. A 4-month gatifloxacin-containing regimen for treating tuberculosis. N. Engl. J. Med. 371, 1588–1598 (2014).
Sonnenberg, P. et al. HIV-1 and recurrence, relapse and reinfection of tuberculosis after cure: a cohort study in South African mineworkers. Lancet 358, 1687–1693 (2001).
Stop TB Partnership. The global plan to stop TB 2016–2020. http://www.stoptb.org/global/plan/plan2/# (2016).
Johnson, J.L. et al. Shortening treatment in adults with non-cavitary tuberculosis and 2-month culture conversion. Am. J. Respir. Crit. Care Med. 180, 558–563 (2009).
Warner, D.F. & Mizrahi, V. Shortening treatment for tuberculosis—to basics. N. Engl. J. Med. 371, 1642–1643 (2014).
Seon, H.J., Kim, Y.I., Lim, S.C., Kim, Y.H. & Kwon, Y.S. Clinical significance of residual lesions in chest computed tomography after anti-tuberculosis treatment. Int. J. Tuberc. Lung Dis. 18, 341–346 (2014).
Ralph, A.P. et al. A simple, valid numerical score for grading chest X-ray severity in adult smear-positive pulmonary tuberculosis. Thorax 65, 863–869 (2010).
Pasipanodya, J.G. et al. Pulmonary impairment after tuberculosis and its contribution to TB burden. BMC Public Health 10, 259 (2010).
Wejse, C. et al. TBscore: signs and symptoms from tuberculosis patients in a low-resource setting have predictive value and may be used to assess clinical course. Scand. J. Infect. Dis. 40, 111–120 (2008).
Pasipanodya, J.G. et al. Pulmonary impairment after tuberculosis. Chest 131, 1817–1824 (2007).
Pasipanodya, J.G. et al. Using the St. George respiratory questionnaire to ascertain health quality in persons with treated pulmonary tuberculosis. Chest 132, 1591–1598 (2007).
Hnizdo, E., Singh, T. & Churchyard, G. Chronic pulmonary function impairment caused by initial and recurrent pulmonary tuberculosis following treatment. Thorax 55, 32–38 (2000).
Ramos, L.M.M., Sulmonett, N., Ferreira, C.S., Henriques, J.F. & de Miranda, S.S. Functional profile of patients with tuberculosis sequelae in a university hospital. J. Bras. Pneumol. 32, 43–47 (2006).
Nihues, S., de S.E. et al. Chronic symptoms and pulmonary dysfunction in post-tuberculosis Brazilian patients. Braz. J. Infect. Dis. 19, 492–497 (2015).
Baig, I.M., Saeed, W. & Khalil, K.F. Post-tuberculous chronic obstructive pulmonary disease. J. Coll. Physicians Surg. Pak. 20, 542–544 (2010).
Mitchison, D.A. Basic mechanisms of chemotherapy. Chest 76 (Suppl. 6), 771–781 (1979).
Hu, Y. et al. Detection of mRNA transcripts and active transcription in persistent Mycobacterium tuberculosis induced by exposure to rifampin or pyrazinamide. J. Bacteriol. 182, 6358–6365 (2000).
Rustad, T.R. et al. Global analysis of mRNA stability in Mycobacterium tuberculosis. Nucleic Acids Res. 41, 509–517 (2013).
Ignatov, D.V. et al. Dormant nonculturable Mycobacterium tuberculosis retains stable low-abundant mRNA. BMC Genomics 16, 954 (2015).
Cheebsumon, P. et al. Effects of image characteristics on performance of tumor delineation methods: a test–retest assessment. J. Nucl. Med. 52, 1550–1558 (2011).
Huang, W. et al. Standard uptake value and metabolic tumor volume of [18F]FDG PET–CT predict short-term outcome early in the course of chemoradiotherapy in advanced non-small-cell lung cancer. Eur. J. Nucl. Med. Mol. Imaging 38, 1628–1635 (2011).
Dibble, E.H. et al. [18F]FDG metabolic tumor volume and total glycolytic activity of oral cavity and oropharyngeal squamous cell cancer: adding value to clinical staging. J. Nucl. Med. 53, 709–715 (2012).
Lin, P.L. et al. Sterilization of granulomas is common in active and latent tuberculosis despite within-host variability in bacterial killing. Nat. Med. 20, 75–79 (2014).
Via, L.E. et al. Infection dynamics and response to chemotherapy in a rabbit model of tuberculosis using 2-[18F]fluoro-deoxy-D-glucose positron emission tomography and computed tomography. Antimicrob. Agents Chemother. 56, 4391–4402 (2012).
Lin, P.L. et al. Radiologic responses in cynomolgous macaques for assessing tuberculosis chemotherapy regimens. Antimicrob. Agents Chemother. 57, 4237–4244 (2013).
Coleman, M.T. et al. Early changes by [18F]fluorodeoxyglucose positron emission tomography co-registered with computed tomography predict outcome after Mycobacterium tuberculosis infection in cynomolgus macaques. Infect. Immun. 82, 2400–2404 (2014).
Davis, S.L. et al. Non-invasive pulmonary 2-[18F]fluoro-deoxy-D-glucose positron emission tomography correlates with bactericidal activity of tuberculosis drug treatment. Antimicrob. Agents Chemother. 53, 4879–4884 (2009).
Martinez, V. et al. [18F]FDG PET–CT in tuberculosis: an early non-invasive marker of therapeutic response. Int. J. Tuberc. Lung Dis. 16, 1180–1185 (2012).
Dureja, S., Sen, I.B. & Acharya, S. Potential role of [18F]FDG PET–CT as an imaging biomarker for the non-invasive evaluation in uncomplicated skeletal tuberculosis: a prospective clinical observational study. Eur. Spine J. 23, 2449–2454 (2014).
Demura, Y. et al. Usefulness of [18F]fluorodeoxyglucose positron emission tomography for diagnosing disease activity and monitoring therapeutic response in patients with pulmonary mycobacteriosis. Eur. J. Nucl. Med. Mol. Imaging 36, 632–639 (2009).
Chen, R.Y. et al. PET–CT imaging correlates with treatment outcome in patients with multidrug-resistant tuberculosis. Sci. Transl. Med. 6, 265ra166 (2014).
Stelzmueller, I. et al. [18F]FDG PET–CT in the initial assessment and for follow-up in patients with tuberculosis. Clin. Nucl. Med. 41, e187–e194 (2016).
Sathekge, M., Maes, A., Kgomo, M., Stoltz, A. & Van de Wiele, C. Use of [18F]FDG PET to predict response to first-line tuberculostatics in HIV-associated tuberculosis. J. Nucl. Med. 52, 880–885 (2011).
World Health Organization. Treatment of Tuberculosis Guidelines 4th edn. (WHO Press, Geneva, 2010).
Walter, N.D. et al. Transcriptional adaptation of drug-tolerant Mycobacterium tuberculosis during treatment of human tuberculosis. J. Infect. Dis. 212, 990–998 (2015).
Lew, J.M., Kapopoulou, A. & Jones, L.M.C.S. TubercuList—TB gene database. http://tuberculist.epfl.ch/quicksearch.php?gene+name=Rv2031c.
Yuan, Y., Crane, D.D. & Barry, C.E. III. Stationary-phase-associated protein expression in Mycobacterium tuberculosis: function of the mycobacterial α-crystallin homolog. J. Bacteriol. 178, 4484–4492 (1996).
Mittl, R.L. Jr. et al. Radiographic resolution of community-acquired pneumonia. Am. J. Respir. Crit. Care Med. 149, 630–635 (1994).
Friedrich, S.O. et al. Assessment of the sensitivity and specificity of Xpert MTB–RIF assay as an early sputum biomarker of response to tuberculosis treatment. Lancet Respir. Med. 1, 462–470 (2013).
Lahtinen, S.J. et al. Degradation of 16S rRNA, and attributes of viability of viable but nonculturable probiotic bacteria. Lett. Appl. Microbiol. 46, 693–698 (2008).
Marx, F.M. et al. The temporal dynamics of relapse and re-infection tuberculosis after successful treatment: a retrospective cohort study. Clin. Infect. Dis. 58, 1676–1683 (2014).
Middelkoop, K., Bekker, L.-G., Shashkina, E., Kreiswirth, B. & Wood, R. Retreatment tuberculosis in a South African community: the role of re-infection, HIV and antiretroviral treatment. Int. J. Tuberc. Lung Dis. 16, 1510–1516 (2012).
Breen, R.A.M. et al. How good are systemic symptoms and blood inflammatory markers at detecting individuals with tuberculosis? Int. J. Tuberc. Lung Dis. 12, 44–49 (2008).
Marais, B. et al. Tuberculosis case definition: time for critical reassessment? Int. J. Tuberc. Lung Dis. 12, 1217–1218, author reply 1218 (2008).
Dooley, K.E. et al. Risk factors for tuberculosis treatment failure, default or relapse, and outcomes of retreatment, in Morocco. BMC Public Health 11, 140 (2011).
Panjabi, R., Comstock, G.W. & Golub, J.E. Recurrent tuberculosis and its risk factors: adequately treated patients are still at high risk. Int. J. Tuberc. Lung Dis. 11, 828–837 (2007).
Hesseling, A.C. et al. Baseline sputum time to detection predicts month two culture conversion and relapse in non-HIV-infected patients. Int. J. Tuberc. Lung Dis. 14, 560–570 (2010).
Warren, R.M. et al. Patients with active tuberculosis often have different strains in the same sputum specimen. Am. J. Respir. Crit. Care Med. 169, 610–614 (2004).
Kritzinger, F.E. et al. No decrease in annual risk of tuberculosis infection in an endemic area in Cape Town, South Africa. Trop. Med. Int. Health 14, 136–142 (2009).
Shanaube, K. et al. Annual risk of tuberculous infection, using different methods, in communities with a high prevalence of TB and HIV in Zambia and South Africa. PLoS One 4, e7749 (2009).
Chien, D., Lodge, M. & Wahl, R. Reproducibility of liver and mediastinal blood pool F-18 activity as normal reference tissues. J. Nucl. Med. 52, 536 (2011).
Kinahan, P.E. & Fletcher, J.W. Positron emission tomography–computed tomography standardized uptake values in clinical practice and assessing response to therapy. Semin. Ultrasound CT MR 31, 496–505 (2010).
Fogh, S.E. et al. Pathological correlation of PET–CT-based auto-contouring for radiation planning of lung cancer. Int. J. Radiat. Oncol. Biol. Phys. 78, S202–S203 (2010).
Delbeke, D. et al. Expert opinions on positron emission tomography and computed tomography imaging in lymphoma. Oncologist 14 (Suppl. 2), 30–40 (2009).
Lowe, V.J., Hoffman, J.M., DeLong, D.M., Patz, E.F. & Coleman, R.E. Semiquantitative and visual analysis of FDG–PET images in pulmonary abnormalities. J. Nucl. Med. 35, 1771–1776 (1994).
Hatt, M. et al. Reproducibility of [18F]FDG and 3′-deoxy-3′-[18F]fluorothymidine PET tumor volume measurements. J. Nucl. Med. 51, 1368–1376 (2010).
Dann, E.J. et al. A functional dynamic scoring model to elucidate the significance of post-induction interim [18F]fluorodeoxyglucose positron emission tomography findings in patients with Hodgkin's lymphoma. Haematologica 95, 1198–1206 (2010).
Higashi, K. et al. [18F]FDG uptake by primary tumor as a predictor of intratumoral lymphatic vessel invasion and lymph node involvement in non-small-cell lung cancer: analysis of a multicenter study. J. Nucl. Med. 46, 267–273 (2005).
Barrington, S.F. et al. Concordance between four European centers of PET reporting criteria designed for use in multicenter trials in Hodgkin lymphoma. Eur. J. Nucl. Med. Mol. Imaging 37, 1824–1833 (2010).
Jacomelli, M. et al. Bronchoscopy for the diagnosis of pulmonary tuberculosis in patients with negative sputum-smear microscopy results. J. Bras. Pneumol. 38, 167–173 (2012).
Conde, M.B. et al. Comparison of sputum induction with fiberoptic bronchoscopy in the diagnosis of tuberculosis: experience at an acquired immune deficiency syndrome reference center in Rio de Janeiro, Brazil. Am. J. Respir. Crit. Care Med. 162, 2238–2240 (2000).
George, P.M. et al. Post-bronchoscopy sputum: improving the diagnostic yield in smear-negative pulmonary TB. Respir. Med. 105, 1726–1731 (2011).
Galagan, J.E. et al. The Mycobacterium tuberculosis regulatory network and hypoxia. Nature 499, 178–183 (2013).
Commandeur, S. et al. An unbiased genome-wide Mycobacterium tuberculosis gene expression approach to discover antigens targeted by human T cells expressed during pulmonary infection. J. Immunol. 190, 1659–1671 (2013).
Acknowledgements
Funding was provided by grants from the Catalysis Foundation for Health (CFH) (G.W., C.E.B. and D.A.) the Division of Intramural Research, National Institute of Allergy and Infectious Diseases (C.E.B.) and the National Institute of Allergy and Infectious Diseases, International Collaborations in Infectious Disease Research (G.W.), as well as by a bursary from the South African National Research Fund and the Medical Research Council's Clinician Scholarship Program (S.T.M.). We thank our participants for their willingness to take part in this study. We acknowledge R. Thayer and M. Urdea of CFH, the staff at the Stellenbosch University Immunology Research Group, the International TB Research Center (Seoul), the Western Cape Academic PET–CT Centre, Ithemba LABS, Tygerberg Academic Hospital's Nuclear Medicine Department and Pulmonology Unit, as well as the managers and healthcare providers from the City of Cape Town Health Department for assistance with the recruitment of participants and sample collection.
Author information
Authors and Affiliations
Consortia
Contributions
S.T.M. managed patient recruitment, follow-up and sample collection, analyzed PET–CT scans and drafted the manuscript; R.Y.C., J. Warwick, L.E.V., D.A., C.E.B., J. Winter and G.W. designed the study, analyzed results and edited the manuscript; G.T. conducted statistical analysis and edited the manuscript; S.S., K.R., A.G.L., G.D., T.V., T.S. and G.S. designed and conducted experiments, analyzed results and edited the manuscript; M.K. and M.L. managed patient recruitment, patient follow-up and sample collection, and edited the manuscript; Catalysis TB–Biomarker Consortium members L.L., L.T., B.S., N.D.P. and C.G.G.B. processed samples and conducted experiments; G.v.d.S. and K.S. managed data collection, and database and sample storage; E.M. and L.E.D. analyzed results; A.E. and S.G.-R. analyzed PET–CT scans; H.C. and J.J. managed patient recruitment and follow-up; and B.A., M.V. and C.K. recruited patients and conducted bronchoscopies.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Notes 1–4 (PDF 1711 kb)
Supplementary Dataset 1
Time to diagnosis and supporting evidence for diagnosis of recurrences (XLSX 12 kb)
Supplementary Dataset 2
Demographical data and PET/CT scan responses per patient for South Korean cohort. (XLSX 13 kb)
Supplementary Dataset 3
Sputum MTB mRNA transcript values. (XLSX 26 kb)
Supplementary Dataset 4
Bronchoalveolar lavage MTB mRNA transcript values. (XLSX 21 kb)
Supplementary Dataset 5
Summary of lesion intensities for scan response patterns. (XLSX 16 kb)
Supplementary Dataset 6
Demographical data and PET/CT scan responses per patient in South African cohort. (XLSX 30 kb)
Supplementary Dataset 7
One year after the end of treatment scan responses per patient. (XLSX 16 kb)
Rights and permissions
About this article
Cite this article
Malherbe, S., Shenai, S., Ronacher, K. et al. Persisting positron emission tomography lesion activity and Mycobacterium tuberculosis mRNA after tuberculosis cure. Nat Med 22, 1094–1100 (2016). https://doi.org/10.1038/nm.4177
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4177
This article is cited by
-
Helper T cell bias following tuberculosis chemotherapy identifies opportunities for therapeutic vaccination to prevent relapse
npj Vaccines (2023)
-
A case report of persistent drug-sensitive pulmonary tuberculosis after treatment completion
BMC Infectious Diseases (2022)
-
Anti-tuberculosis treatment strategies and drug development: challenges and priorities
Nature Reviews Microbiology (2022)
-
An optimized method for purifying, detecting and quantifying Mycobacterium tuberculosis RNA from sputum for monitoring treatment response in TB patients
Scientific Reports (2022)
-
State of the art of 18F-FDG PET/CT application in inflammation and infection: a guide for image acquisition and interpretation
Clinical and Translational Imaging (2021)