Abstract
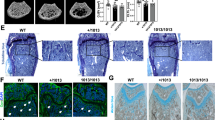
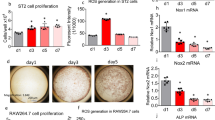
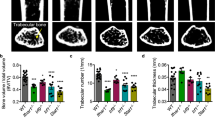
The nfkbl and nfkb2 genes encode closely related products regulating immune and inflammatory responses1–3. Their role during development and differentiation remains unclear. The generation of nfkb1 null mice (p50−/−) resulted in altered immune responses, but had no effect on development4. Similarly, nfkb2 knockout mice (p52−/−) did not show developmental defects (J.C. et al., manuscript submitted). We have investigated the potential for in vivo compensatory functions of these genes by generating double-knockout mice. The surprising result was that the animals developed osteopetrosis because of a defect in osteoclast differentiation, suggesting redundant functions of NF-κB1 and NF-κB2 proteins in the development of this cell lineage. The osteopetrotic phenotype was rescued by bone marrow transplantation, indicating that the hematopoietic component was impaired. These results define a new mouse osteopetrotic mutant and implicate NF-κB proteins in bone development, raising new directions in the treatment of bone disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Grilli, M., Chiu, J. & Lenardo, M. NF-κB and Rel: Participants in a multiform tran-scriptional regulatory system. Int. Rev. Cytol. 143, 1–62 (1993).
Kabrun, N. & Enrietto, P. The Rel family of proteins in oncogenesis and differentiation. Semin. Cancer Biol. 5, 103–112 (1994).
Kopp, E. & Ghosh, S. NF-κB and Rel proteins in innate immunity. Adv. Immunol. 58, 1–27 (1995).
Sha, W., Liou, H.-C., Tuomanen, E. & Baltimore, D. Targeted disruption of the p50 subunit of NF-κB leads to multifocal defects in immune responses. Cell 80, 321–330 (1995).
Minkin, C. Bone acid phosphatase: Tartrate-resistant acid phosphatase as a marker of osteoclast function. Calcif. Tissue Int. 34, 285–293 (1982).
Takahashi, N. et al. Postmitotic osteoclast precursors are mononuclear cells which express macrophage-associated phenotypes. Dev. Biol. 163, 212–221 (1994).
Manolagas, S.C. Role of cytokines in bone resorption. Bone 17, 63S–67S (1995).
Crigoriadis, A.E., Schellander, K., Wang, Z.-Q. & Wagner, E.F. Osteoblasts are target cells for transformation in c-fos transgenic mice. J. Cell Biol. 122, 685–701 (1993).
Pralet, B., Male, P., Neff, L. & Baron, R. Identification of a functional mononuclear precursor of the osteoclast in chicken medullary bone marrow cultures. J. Bone Miner. Res. 7, 405–414 (1992).
Johnson, R.S., Spiegelman, B.M. & Papaioannou, V. Pleiotropic effects of a null mutation in the c-fos proto-oncogene. Cell 71, 577–586 (1992).
Wang, Z.-Q. et al. Bone and haematopoietic defects in mice lacking c-fos. Nature 360, 741–745 (1992).
Stein, B. et al. Cross-coupling of the NF-kappa B p65 and Fos/Jun transcription factors produces potentiated biological function. EMBO J. 12, 3879–3891 (1993).
Grigoriadis, A.E., Wang, Z.-Q. & Wagner, E. Fos and bone cell development: Lessons from a nuclear oncogene. Trends Genet. 11, 436–442 (1995).
Soriano, P., Montgomery, C., Geske, R. & Bradley, A. Targeted disruption of the c-src proto-oncogene leads to osteopetrosis in mice. Cell 64, 693–702 (1991).
Suda, T., Udagawa, N., Nakamura, J., Miyaura, C. & Takahashi, N. Modulation of osteoclast differentiation by local factors. Bone 17, 87S–91S (1995).
Vortkamp, A. et al. Regulation of rate of cartilage differentiation by Indian Hedgehog and PTH-related protein. Science 273, 613–622 (1996).
Weir, E. et al. Targeted overexpression of parathyroid hormone-related peptide in chondrocytes causes chondrodysplasia and delayed endochondral bone formation. Proc. Natl. Acad. Sd. USA 93, 10240–10245 (1996).
Jimi, E. et al. lnterleukin-1 alfa activates an NF-kappaB-like factor in osteoclast-like cells. J. Biol. Chem. 271, 4605–4608 (1996).
Whyte, M.P. Carbonic anhydrase II deficiency. Gin. Orthop. Relat. Res. 294, 52–63 (1993).
Manolagas, S.C., Bellido, T. & Jilka, R.L. New insights into the cellular, biochemical, and molecular basis of postmenopausal and senile osteoporosis: roles of IL-6 and gpl30. Int. J. Immunopharmacol. 17, 109–116 (1995).
Stein, B. & Yang, M.X. Repression of the interleukin-6 promoter by estrogen receptor is mediated by NF-κB and C/EBPβ. Mol. Cell. Biol. 15, 4971–4979 (1995).
Coligan, J.E., Kruisbeek, A.M., Margulies, D.H., Shevach, E.M. & Strober, W. Isolation and Fractionation of Mononuclear Cell Populations. (Wiley, New York, 1992).
Grigoriadis, A.E. et al. c-Fos: A key regulator of osteoclast-macrophage lineage determination and bone remodeling. Science 266, 443–448 (1994).
Tondravi, M.M. et al. Osteopetrosis in mice lacking haematopoietic transcription factor PU.1. Nature 386, 81–84 (1997).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Iotsova, V., Caamaño, J., Loy, J. et al. Osteopetrosis in mice lacking NF-κB1 and NF-κB2. Nat Med 3, 1285–1289 (1997). https://doi.org/10.1038/nm1197-1285
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm1197-1285
This article is cited by
-
Recent advances of NFATc1 in rheumatoid arthritis-related bone destruction: mechanisms and potential therapeutic targets
Molecular Medicine (2024)
-
Temporal gene expression profiling during early-stage traumatic temporomandibular joint bony ankylosis in a sheep model
BMC Oral Health (2024)
-
Relationship between diet-related inflammation and bone health under different levels of body mass index
Journal of Orthopaedic Surgery and Research (2023)
-
Osteoarthritis: pathogenic signaling pathways and therapeutic targets
Signal Transduction and Targeted Therapy (2023)
-
Effects of Co-exposure to Fluoride and Arsenic on TRAF-6 Signaling and NF-κB Pathway of Bone Metabolism
Biological Trace Element Research (2023)