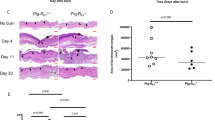
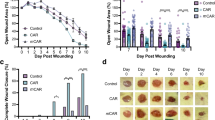
Abstract
Annually, 1.25 million individuals suffer burns in the United States and 6.5 million experience chronic skin ulcers, often from diabetes, pressure or venous stasis1. Growth factors are essential mediators of wound repair1,2, but their success as therapeutics in wound treatment has, so far, been limited1,2. Therefore, there is a need to identify new wound-response regulatory factors, but few have appeared in recent years1,2. Progranulin3 (also called granulin or epithelin precursor4, acrogranin5 or PC-derived growth factor6) is a growth factor involved in tumorigenesis6,7,8,9,10,11 and development12,13. Peptides derived from progranulin have been isolated from inflammatory cells14, which led to suggestions that progranulin gene products are involved in the wound response10,14, but this remains undemonstrated. We report that in murine transcutaneous puncture wounds, progranulin mRNA is expressed in the inflammatory infiltrate and is highly induced in dermal fibroblasts and endothelia following injury. When applied to a cutaneous wound, progranulin increased the accumulation of neutrophils, macrophages, blood vessels and fibroblasts in the wound. It acts directly on isolated dermal fibroblasts and endothelial cells to promote division, migration and the formation of capillary-like tubule structures. Progranulin is, therefore, a probable wound-related growth factor.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Singer, A.J. & Clark, R.A. Cutaneous wound healing. N. Engl. J. Med. 341, 738–746 (1999).
Martin, P. Wound healing—aiming for perfect skin regeneration. Science 276, 75–81 (1997).
Bhandari, V., Palfree, R.G. & Bateman, A. Isolation and sequence of the granulin precursor cDNA from human bone marrow reveals tandem cysteine-rich granulin domains. Proc. Natl. Acad. Sci. USA 89, 1715–1719 (1992).
Plowman, G.D. et al. The epithelin precursor encodes two proteins with opposing activities on epithelial cell growth. J. Biol. Chem. 267, 13073–13078 (1992).
Baba, T. et al. Acrogranin, an acrosomal cysteine-rich glycoprotein, is the precursor of the growth-modulating peptides, granulins, and epithelins, and is expressed in somatic as well as male germ cells, Mol. Reprod. Dev. 34, 233–243 (1993).
Zhou, J., Gao, G., Crabb, J.W. & Serrero, G. Purification of an autocrine growth factor homologous with mouse epithelin precursor from a highly tumorigenic cell line. J. Biol. Chem. 268, 10863–10869 (1993).
Xu, S.Q. et al. The granulin/epithelin precursor abrogates the requirement for the insulin-like growth factor 1 receptor for growth in vitro. J. Biol. Chem. 273, 20078–20083 (1998).
Lu, R. & Serrero, G. Inhibition of PC cell-derived growth factor (PCDGF, epithelin/granulin precursor) expression by antisense PCDGF cDNA transfection inhibits tumorigenicity of the human breast carcinoma cell line MDA-MB-468. Proc. Natl. Acad. Sci. USA 97, 3993–3998 (1998).
He, Z. & Bateman, A. Progranulin gene expression regulates epithelial cell growth and promotes tumor growth in vivo. Cancer Res. 59, 3222–3229 (1999).
Zanocco-Marani, T. et al. Biological activities and signaling pathways of the granulin/epithelin precursor. Cancer Res. 59, 5331–5340 (1999).
Liau, L.M. et al. Identification of a human glioma-associated growth factor gene, granulin, using differential immuno-absorption. Cancer Res. 60, 1353–1360 (2000).
Diaz-Cueto, L., Stein, P., Jacobs, A., Schultz, R.M. & Gerton, G.L. Modulation of mouse preimplantation embryo development by acrogranin (epithelin/granulin precursor). Dev. Biol. 217, 406–418 (2000).
Suzuki, M., Yoshida, S., Nishihara, M. & Takahashi, M. Identification of a sex steroid-inducible gene in the neonatal rat hypothalamus. Neurosci. Lett. 242, 127–130 (1998).
Bateman, A., Belcourt, D., Bennett, H., Lazure, C. & Solomon, S. Granulins, a novel class of peptide from leukocytes. Biochem. Biophys. Res. Commun. 173, 1161–1168 (1990).
Shoyab, M., McDonald, V.L., Byles, C., Todaro, G.J. & Plowman, G.D. Epithelins 1 and 2: isolation and characterization of two cysteine-rich growth-modulating proteins. Proc. Natl. Acad. Sci. USA 87, 7912–7916 (1990).
Fiddes, J.C. et al. Preclinical wound-healing studies with recombinant human basic fibroblast growth factor. Ann. NY Acad. Sci. 638, 316–328 (1991).
He, Z., Ismail, A., Kriazhev, L., Saddvakassova, G. & Bateman, A. Progranulin (PC-cell-derived growth factor/Acrogranin) regulates invasion and cell survival. Cancer Res. 62, 5590–5596 (2002).
Nicosia, R.F. & Ottinetti, A. Modulation of microvascular growth and morphogenesis by reconstituted basement membrane gel in three-dimensional cultures of rat aorta: a comparative study of angiogenesis in matrigel, collagen, fibrin, and plasma clot. In Vitro Cell Dev. Biol. 26, 119–128 (1990).
Kubota, Y., Kleinman, H.K., Martin, G.R. & Lawley, T.J. Role of laminin and basement membrane in the morphological differentiation of human endothelial cells into capillary-like structures. J. Cell Biol. 107, 1589–1598 (1988).
Kitajima, I., Unoki, K. & Maruyama, I. Phosphorothioate oligodeoxynucleotides inhibit basic fibroblast growth factor-induced angiogenesis in vitro and in vivo. Antisense Nucleic Acid Drug Dev. 9, 233–239 (1999).
Yamagishi, S. et al. Advanced glycation end products-driven angiogenesis in vitro. Induction of the growth and tube formation of human microvascular endothelial cells through autocrine vascular endothelial growth factor. J. Biol. Chem. 272, 8723–8730 (1997).
Carter, B. & Halper, J. Transforming growth factor e is a novel mediator of wound repair. Wound Repair Regen. 4, 259–268 (1996).
Daniel, R., He, Z., Carmichael, K.P., Halper, J. & Bateman, A. Cellular localization of gene expression for progranulin. J. Histochem. Cytochem. 48, 999–1009 (2000).
Chipman, S.D. et al. Defective pro α 2(I) collagen synthesis in a recessive mutation in mice: a model of human osteogenesis imperfecta. Proc. Natl. Acad. Sci. USA 90, 1701–1705 (1993).
Nishida, M. et al. Isolation and characterization of human and rat cardiac microvascular endothelial cells. Am. J. Physiol. 264, H639–652 (1993).
Acknowledgements
We thank A. Mah for assistance with the in situ hybridization and W. Powell for performing neutrophil chemoattractant assays. This work was supported by a grant from the Canadian Institutes of Health Research to A.B. (MT-011397), by studentships from the Research Institute of the Royal Victoria Hospital to Z.H. and C.H.P.O., by NIH grant to J.H. (CA71023) and by a grant from The University of Georgia Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
He, Z., Ong, C., Halper, J. et al. Progranulin is a mediator of the wound response. Nat Med 9, 225–229 (2003). https://doi.org/10.1038/nm816
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm816
This article is cited by
-
Identification of potential biological processes and key genes in diabetes-related stroke through weighted gene co-expression network analysis
BMC Medical Genomics (2024)
-
Progranulin (PGRN) as a regulator of inflammation and a critical factor in the immunopathogenesis of cardiovascular diseases
Journal of Inflammation (2023)
-
The Role of Progranulin (PGRN) in the Pathogenesis of Ischemic Stroke
Cellular and Molecular Neurobiology (2023)
-
Granulin as an important immune molecule involved in lamprey tissue repair and regeneration by promoting cell proliferation and migration
Cellular & Molecular Biology Letters (2022)
-
Progranulin from different gliocytes in the nucleus accumbens exerts distinct roles in FTD- and neuroinflammation-induced depression-like behaviors
Journal of Neuroinflammation (2022)