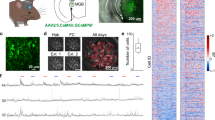
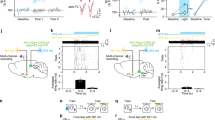
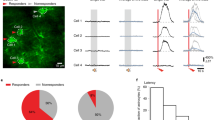
Abstract
Neural circuits underlying auditory fear conditioning have been extensively studied. Here we identified a previously unexplored pathway from the lateral amygdala (LA) to the auditory cortex (ACx) and found that selective silencing of this pathway using chemo- and optogenetic approaches impaired fear memory retrieval. Dual-color in vivo two-photon imaging of mouse ACx showed pathway-specific increases in the formation of LA axon boutons, dendritic spines of ACx layer 5 pyramidal cells, and putative LA–ACx synaptic pairs after auditory fear conditioning. Furthermore, joint imaging of pre- and postsynaptic structures showed that essentially all new synaptic contacts were made by adding new partners to existing synaptic elements. Together, these findings identify an amygdalocortical projection that is important to fear memory expression and is selectively modified by associative fear learning, and unravel a distinct architectural rule for synapse formation in the adult brain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
02 July 2018
In the version of this article initially published, Fig. 7f purported to show an example of a multi-synapse spine. However, the structure in question included a mitochondrion and microtubules, meaning that it was actually a segment of dendritic shaft. A new image showing an example of a bona fide spine has been substituted. The legend has been changed to state that the image shows two boutons rather than three. The error has been corrected in the HTML and PDF versions of the article. The original and corrected figures are shown in the accompanying Author Correction.
References
Goosens, K.A. & Maren, S. Contextual and auditory fear conditioning are mediated by the lateral, basal, and central amygdaloid nuclei in rats. Learn. Mem. 8, 148–155 (2001).
Letzkus, J.J. et al. A disinhibitory microcircuit for associative fear learning in the auditory cortex. Nature 480, 331–335 (2011).
Peter, M. et al. Induction of immediate early genes in the mouse auditory cortex after auditory cued fear conditioning to complex sounds. Genes Brain Behav. 11, 314–324 (2012).
Boatman, J.A. & Kim, J.J. A thalamo-cortico-amygdala pathway mediates auditory fear conditioning in the intact brain. Eur. J. Neurosci. 24, 894–900 (2006).
Weinberger, N.M. Specific long-term memory traces in primary auditory cortex. Nat. Rev. Neurosci. 5, 279–290 (2004).
Moczulska, K.E. et al. Dynamics of dendritic spines in the mouse auditory cortex during memory formation and memory recall. Proc. Natl. Acad. Sci. USA 110, 18315–18320 (2013).
Denk, W. & Svoboda, K. Photon upmanship: why multiphoton imaging is more than a gimmick. Neuron 18, 351–357 (1997).
Harris, K.M. & Kater, S.B. Dendritic spines: cellular specializations imparting both stability and flexibility to synaptic function. Annu. Rev. Neurosci. 17, 341–371 (1994).
Grillo, F.W. et al. Increased axonal bouton dynamics in the aging mouse cortex. Proc. Natl. Acad. Sci. USA 110, E1514–E1523 (2013).
Holtmaat, A., Wilbrecht, L., Knott, G.W., Welker, E. & Svoboda, K. Experience-dependent and cell-type-specific spine growth in the neocortex. Nature 441, 979–983 (2006).
Knott, G.W., Holtmaat, A., Wilbrecht, L., Welker, E. & Svoboda, K. Spine growth precedes synapse formation in the adult neocortex in vivo. Nat. Neurosci. 9, 1117–1124 (2006).
Lai, C.S., Franke, T.F. & Gan, W.B. Opposite effects of fear conditioning and extinction on dendritic spine remodelling. Nature 483, 87–91 (2012).
Xu, T. et al. Rapid formation and selective stabilization of synapses for enduring motor memories. Nature 462, 915–919 (2009).
Hofer, S.B., Mrsic-Flogel, T.D., Bonhoeffer, T. & Hübener, M. Experience leaves a lasting structural trace in cortical circuits. Nature 457, 313–317 (2009).
Zuo, Y., Yang, G., Kwon, E. & Gan, W.B. Long-term sensory deprivation prevents dendritic spine loss in primary somatosensory cortex. Nature 436, 261–265 (2005).
Becker, N., Wierenga, C.J., Fonseca, R., Bonhoeffer, T. & Nägerl, U.V. LTD induction causes morphological changes of presynaptic boutons and reduces their contacts with spines. Neuron 60, 590–597 (2008).
Romanski, L.M. & LeDoux, J.E. Equipotentiality of thalamo-amygdala and thalamo-cortico-amygdala circuits in auditory fear conditioning. J. Neurosci. 12, 4501–4509 (1992).
Malenka, R.C. & Bear, M.F. LTP and LTD: an embarrassment of riches. Neuron 44, 5–21 (2004).
Cauller, L. Layer I of primary sensory neocortex: where top-down converges upon bottom-up. Behav. Brain Res. 71, 163–170 (1995).
Zhang, S. et al. Selective attention. Long-range and local circuits for top-down modulation of visual cortex processing. Science 345, 660–665 (2014).
Weinberger, N.M. Physiological memory in primary auditory cortex: characteristics and mechanisms. Neurobiol. Learn. Mem. 70, 226–251 (1998).
LeDoux, J.E. Emotion circuits in the brain. Annu. Rev. Neurosci. 23, 155–184 (2000).
Armbruster, B.N., Li, X., Pausch, M.H., Herlitze, S. & Roth, B.L. Evolving the lock to fit the key to create a family of G protein-coupled receptors potently activated by an inert ligand. Proc. Natl. Acad. Sci. USA 104, 5163–5168 (2007).
Stachniak, T.J., Ghosh, A. & Sternson, S.M. Chemogenetic synaptic silencing of neural circuits localizes a hypothalamus→midbrain pathway for feeding behavior. Neuron 82, 797–808 (2014).
Zhu, Y., Wienecke, C.F., Nachtrab, G. & Chen, X. A thalamic input to the nucleus accumbens mediates opiate dependence. Nature 530, 219–222 (2016).
De Paola, V. et al. Cell type-specific structural plasticity of axonal branches and boutons in the adult neocortex. Neuron 49, 861–875 (2006).
Rubio-Garrido, P., Pérez-de-Manzo, F., Porrero, C., Galazo, M.J. & Clascá, F. Thalamic input to distal apical dendrites in neocortical layer 1 is massive and highly convergent. Cereb. Cortex 19, 2380–2395 (2009).
Schneider, D.M., Nelson, A. & Mooney, R. A synaptic and circuit basis for corollary discharge in the auditory cortex. Nature 513, 189–194 (2014).
Gilmartin, M.R., Balderston, N.L. & Helmstetter, F.J. Prefrontal cortical regulation of fear learning. Trends Neurosci. 37, 455–464 (2014).
Senn, V. et al. Long-range connectivity defines behavioral specificity of amygdala neurons. Neuron 81, 428–437 (2014).
Xiong, Q., Znamenskiy, P. & Zador, A.M. Selective corticostriatal plasticity during acquisition of an auditory discrimination task. Nature 521, 348–351 (2015).
Harris, K.D. & Mrsic-Flogel, T.D. Cortical connectivity and sensory coding. Nature 503, 51–58 (2013).
Douglas, R.J. & Martin, K.A. Neuronal circuits of the neocortex. Annu. Rev. Neurosci. 27, 419–451 (2004).
Feng, G. et al. Imaging neuronal subsets in transgenic mice expressing multiple spectral variants of GFP. Neuron 28, 41–51 (2000).
Cichon, J. & Gan, W.B. Branch-specific dendritic Ca2+ spikes cause persistent synaptic plasticity. Nature 520, 180–185 (2015).
Stuart, G.J. & Spruston, N. Dendritic integration: 60 years of progress. Nat. Neurosci. 18, 1713–1721 (2015).
Otazu, G.H., Chae, H., Davis, M.B. & Albeanu, D.F. Cortical feedback decorrelates olfactory bulb output in awake mice. Neuron 86, 1461–1477 (2015).
Wilson, R.I. & Nicoll, R.A. Endogenous cannabinoids mediate retrograde signalling at hippocampal synapses. Nature 410, 588–592 (2001).
Schafe, G.E., Nader, K., Blair, H.T. & LeDoux, J.E. Memory consolidation of Pavlovian fear conditioning: a cellular and molecular perspective. Trends Neurosci. 24, 540–546 (2001).
Toni, N., Buchs, P.A., Nikonenko, I., Bron, C.R. & Muller, D. LTP promotes formation of multiple spine synapses between a single axon terminal and a dendrite. Nature 402, 421–425 (1999).
Kleindienst, T., Winnubst, J., Roth-Alpermann, C., Bonhoeffer, T. & Lohmann, C. Activity-dependent clustering of functional synaptic inputs on developing hippocampal dendrites. Neuron 72, 1012–1024 (2011).
Fu, M., Yu, X., Lu, J. & Zuo, Y. Repetitive motor learning induces coordinated formation of clustered dendritic spines in vivo. Nature 483, 92–95 (2012).
Park, H. & Poo, M.M. Neurotrophin regulation of neural circuit development and function. Nat. Rev. Neurosci. 14, 7–23 (2013).
Caroni, P., Donato, F. & Muller, D. Structural plasticity upon learning: regulation and functions. Nat. Rev. Neurosci. 13, 478–490 (2012).
Wiltgen, B.J., Brown, R.A., Talton, L.E. & Silva, A.J. New circuits for old memories: the role of the neocortex in consolidation. Neuron 44, 101–108 (2004).
Frankland, P.W. & Bontempi, B. The organization of recent and remote memories. Nat. Rev. Neurosci. 6, 119–130 (2005).
Krettek, J.E. & Price, J.L. Amygdaloid projections to subcortical structures within the basal forebrain and brainstem in the rat and cat. J. Comp. Neurol. 178, 225–254 (1978).
Gray, T.S., Carney, M.E. & Magnuson, D.J. Direct projections from the central amygdaloid nucleus to the hypothalamic paraventricular nucleus: possible role in stress-induced adrenocorticotropin release. Neuroendocrinology 50, 433–446 (1989).
Kirov, S.A., Sorra, K.E. & Harris, K.M. Slices have more synapses than perfusion-fixed hippocampus from both young and mature rats. J. Neurosci. 19, 2876–2886 (1999).
Chklovskii, D.B., Mel, B.W. & Svoboda, K. Cortical rewiring and information storage. Nature 431, 782–788 (2004).
Acknowledgements
We thank N. Xu for help with chronic window surgery, Q. Hu for confocal imaging, Y. Kong and B. Zhang for electron microscopy, L. Han for behavioral analysis, L. Zhou for slice recording, and Y. Dan for critical comments and suggestions. This work was supported by grants from Ministry of Science and Technology (973 Program, 2011CBA00400, M.-m.P.) and Chinese Academy of Sciences (Strategic Priority Research Program, XDB02020001, M.-m.P.), and a SIBS-SA scholarship to Y.Y.
Author information
Authors and Affiliations
Contributions
Y.Y., D.-q.L. and M.-m.P. designed the experiments. Y.Y. and D.-q.L. performed the experiments and analyzed the data. W.H. performed in utero electroporation experiments. Y.S. and J.D. performed the electrophysiology experiments. Y.Z. guided spine data analysis. Y.Y., D.-q.L. Y.Z. and M.-m.P. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 ACx plasticity is required for fear conditioning.
(a) Freezing time of conditioned and control mice before CS presentation and during CS presentation in a new context. Student’s t-test, p> 0.5 for baseline, p< 0.0001 during CS. (b) Diagram showing bilateral infusion of drugs in ACx. (c) Freezing responses in recall test for mice bilaterally infused with different doses of APV at 30 min before fear conditioning. Student’s t-test, p> 0.05 for 0.5-μg and 1-μg group, p< 0.0001 for 2-μg and 10-μg group. (d) Freezing responses for mice bilaterally infused with APV (2 μg) and muscimol immediately after fear conditioning. Student’s t-test, p = 0.028 for APV, p = 0.009 for muscimol. Error bar, s.e.m.
Supplementary Figure 2 Drug spread in ACx.
Coronal slices of mouse brain showing the spread of 1 μl of FITC (Molecular Weight: 389), at 30 min after infusion. Scale bar: 500μm.
Supplementary Figure 3 Coexpression of ChR2 and hM4D in LA for slice recording.
(a) Image showing ChR2 (red) and hM4D (green) expressing neurons in LA. Scale bar: 100 μm. (b) Zoom-in images showing co-expression of ChR2 and hM4D. Scale bar: 10 μm. (c) Proportion of ChR2/hM4D expressing neurons.
Supplementary Figure 4 Virus expression of hM4D and eArch3.0 in LA.
(a) Left: Image showing expression of AAV-hM4D in LA. Scale bar: 500μm. Right: Image showing hM4D-expressing LA axons in ACx. Scale bar: 100 μm. (b) Freezing responses for mice expressing hM4D in LA neurons in two recall tests. Mice were first tested when CNO was infused into ACx. The same mice were tested again 24 h later when saline was infused into ACx. Each line represents data from one mouse. Paired t-test, p= 0.032. (c) Left: Image showing expression of AAV-eArch3.0 in LA. Scale bar: 500 μm. Right: Image showing eArch3.0-expressing LA axons in ACx. Scale bar: 100 μm.
Supplementary Figure 5 Criteria for bouton identification.
(a, b) Fluorescent intensity was measured along identified axons. Bright swellings were identified as boutons when the peak intensity was over 3 fold that of the axon shaft (blue dotted line). (c, d) When changing the threshold to 2 fold (green solid line in B), results of bouton counting were not significantly different using the two different criteria. Each data point represents results from one image stack (>100 boutons each).
Supplementary Figure 6 Retrograde tracing using cholera toxin subunit B (CTB) showed that neurons in LA, MG and ACC project to ACx.
(a) CTB was injected into the superficial layers of ACx. Scale: 100 μm. (b-e) Retrograde labeled neurons were found in the contralateral ACx (b), MG (c), LA (d) and frontal cortex including Cg1, Cg2 and M2 (e). Scale: 100 μm.
Supplementary Figure 7 Correlation of bouton/spine formation with freezing responses.
Correlation analyses showed weak correlation between LA axon bouton formation and freezing responses, and between ACx spine formation and freezing responses.
Supplementary Figure 8 No changes in the spine dynamics in the apical dendrites of ACx L2/3 neurons.
(a) Image showing that L2/3 neurons were labeled with tdTomato using in-utero electroporation. Scale: 500 μm. (b) Zoom-in view of the labeled L2/3 neurons. Scale: 100 μm. (c) Example images obtained by repeated imaging of the same apical dendrites of ACx L2/3 neurons in control and conditioned mice. Green and red arrows, newly formed and eliminated spines, respectively, as compared to -1d. Scale bar, 2 μm. (d) Percentages of spine formation and elimination at apical dendrites of L2/3 neurons in control and conditioned mice at 2h (control: n = 7; conditioned: n = 6) and 3d (control: n = 8; conditioned: n = 6). Mann-Whitney U-test, p> 0.4 for all comparison. Error bar, s.e.m.
Supplementary Figure 9 Example images demonstrating separation of GFP and YFP signals.
(a) Signals obtained with 535/50 bandpass filter, both GFP and YFP signals were collected. (b) Signals obtained with 495/40 bandpass filter. Only GFP signals were collected. (c) YFP-only signals were obtained by subtracting GFP signals from GFP+YFP signals. (d) Merged image of GFP (green) and post-processing YFP (red) signals. Scale bar: 2 μm.
Supplementary Figure 10 Formation rate of boutons/spines in labeled synaptic pairs 2 h after conditioning.
(a) Percentages of newly formed boutons/spines in labeled LA-ACx synaptic pairs (red circles), as compared to those randomly selected (black circles) boutons/spines in the same animal, in conditioned (n = 6) and control (n = 4) mice at 2h after conditioning. Paired t-test, p = 0.03 for bouton formation in conditioned group. p> 0.3 for other comparison.(d, e) Similar to (c), except that labeled synaptic pairs were in MG-ACx (conditioned, n = 4; control, n = 5; p> 0.2) or ACC-ACx connections (conditioned, n = 4; control, n = 4; p> 0.2 for bouton, p = 0.02 for spine in conditioned, p = 0.0021 for spine in control group.).
Supplementary Figure 11 Examples showing that new synapses were mostly made by adding new boutons to existing spines or new spines to existing boutons.
(a) Example showing a new spine (red) growing onto an existing bouton (green). (b) Example showing a new bouton growing onto an existing spine. (c) Example showing de novo formation of a pairs of bouton and spine. (d) Example showing a new bouton growing onto an existing spine with a presynaptic partner labeled. (e) Example showing a new spine growing onto an existing bouton with a postsynaptic partner labeled. (f) Example showing a new spine replacing an existing postsynaptic partner of an existing bouton. Scale bar: 1 μm.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–11 (PDF 1491 kb)
Rights and permissions
About this article
Cite this article
Yang, Y., Liu, Dq., Huang, W. et al. Selective synaptic remodeling of amygdalocortical connections associated with fear memory. Nat Neurosci 19, 1348–1355 (2016). https://doi.org/10.1038/nn.4370
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.4370
This article is cited by
-
Generalized extinction of fear memory depends on co-allocation of synaptic plasticity in dendrites
Nature Communications (2023)
-
Contextual Fear Learning and Extinction in the Primary Visual Cortex of Mice
Neuroscience Bulletin (2023)
-
Neural Integration of Audiovisual Sensory Inputs in Macaque Amygdala and Adjacent Regions
Neuroscience Bulletin (2023)
-
Joint reconstruction of neuron and ultrastructure via connectivity consensus in electron microscope volumes
BMC Bioinformatics (2022)
-
Learning binds new inputs into functional synaptic clusters via spinogenesis
Nature Neuroscience (2022)