Key Points
-
Pulmonary arterial hypertension (PAH) remains a devastating disease without a cure, despite therapeutic innovations
-
Most patients are diagnosed at a very advanced stage of the disease
-
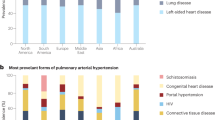
Specific populations of patients, such as those with systemic sclerosis and carriers of PAH-causing mutations (such as in the BMPR2 gene) are at high risk of developing PAH
-
Screening of high-risk populations for PAH is recommended by current guidelines and is an important strategy to improve clinical outcomes
-
Optimal screening algorithms for the early detection of PAH will continue to evolve with ongoing research
Abstract
Pulmonary arterial hypertension (PAH) remains an incurable disease associated with an unacceptably high early mortality, despite advances in therapeutic options. The disease is clinically silent until late in its natural history, when most of the distal pulmonary arteries have been obliterated. Early diagnosis of PAH is associated with improved long-term survival, and screening of at-risk populations is, therefore, a rational strategy to improve outcomes in this condition. Doppler echocardiography is the most widely used screening tool in current clinical practice. The role of evidence-based screening strategies has been clarified by research such as the DETECT study in patients with systemic sclerosis. A multimodal approach, using a range of noninvasive tests, improves the performance of screening algorithms. Right heart catheterization is mandatory to confirm a diagnosis of PAH. Uncertainties exist about the definition and prognostic relevance of pulmonary hypertension during exercise, but accumulating evidence suggests that stress testing of the pulmonary circulation can unmask clinically important early disease. Novel tools for the early detection of pulmonary vascular disease are urgently needed, given the substantial limitations of currently available techniques.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Simonneau, G. et al. Updated clinical classification of pulmonary hypertension. J. Am. Coll. Cardiol. 62 (Suppl.), D34–D41 (2013).
Hoeper, M. M. et al. Definitions and diagnosis of pulmonary hypertension. J. Am. Coll. Cardiol. 62 (Suppl.), D42–D50 (2013).
Thenappan, T. et al. Survival in pulmonary arterial hypertension: a reappraisal of the NIH risk stratification equation. Eur. Respir. J. 35, 1079–1087 (2010).
Humbert, M. et al. Survival in patients with idiopathic, familial, and anorexigen-associated pulmonary arterial hypertension in the modern management era. Circulation 122, 156–163 (2010).
Condliffe, R. et al. Connective tissue disease-associated pulmonary arterial hypertension in the modern treatment era. Am. J. Respir. Crit. Care Med. 179, 151–157 (2009).
Mathai, S. C. et al. Survival in pulmonary hypertension associated with the scleroderma spectrum of diseases: impact of interstitial lung disease. Arthritis Rheum. 60, 569–577 (2009).
Launay, D. et al. Survival in systemic sclerosis-associated pulmonary arterial hypertension in the modern management era. Ann. Rheum. Dis. 72, 1940–1946 (2013).
Ling, Y. et al. Changing demographics, epidemiology, and survival of incident pulmonary arterial hypertension: results from the pulmonary hypertension registry of the United Kingdom and Ireland. Am. J. Respir. Crit. Care Med. 186, 790–796 (2012).
Strange, G. et al. Time from symptoms to definitive diagnosis of idiopathic pulmonary arterial hypertension: the delay study. Pulm. Circ. 3, 89–94 (2013).
Galie, N. et al. Treatment of patients with mildly symptomatic pulmonary arterial hypertension with bosentan (EARLY study): a double-blind, randomised controlled trial. Lancet 371, 2093–2100 (2008).
Benza, R. L. et al. Predicting survival in pulmonary arterial hypertension: insights from the Registry to Evaluate Early and Long-Term Pulmonary Arterial Hypertension Disease Management (REVEAL). Circulation 122, 164–172 (2010).
Humbert, M. et al. Screening for pulmonary arterial hypertension in patients with systemic sclerosis: clinical characteristics at diagnosis and long-term survival. Arthritis Rheum. 63, 3522–3530 (2011).
Rose, G. & Barker, D. J. Epidemiology for the uninitiated. Screening. Br. Med. J. 2, 1417–1418 (1978).
Peacock, A. J., Murphy, N. F., McMurray, J. J., Caballero, L. & Stewart, S. An epidemiological study of pulmonary arterial hypertension. Eur. Respir. J. 30, 104–109 (2007).
Humbert, M. et al. Pulmonary arterial hypertension in France: results from a national registry. Am. J. Respir. Crit. Care Med. 173, 1023–1030 (2006).
Machado, R. D. et al. Mutations of the TGF-beta type II receptor BMPR2 in pulmonary arterial hypertension. Hum. Mutat. 27, 121–132 (2006).
Machado, R. D. et al. BMPR2 haploinsufficiency as the inherited molecular mechanism for primary pulmonary hypertension. Am. J. Hum. Genet. 68, 92–102 (2001).
Larkin, E. K. et al. Longitudinal analysis casts doubt on the presence of genetic anticipation in heritable pulmonary arterial hypertension. Am. J. Respir. Crit. Care Med. 186, 892–896 (2012).
Pope, J. E. et al. Prevalence of elevated pulmonary arterial pressures measured by echocardiography in a multicenter study of patients with systemic sclerosis. J. Rheumatol. 32, 1273–1278 (2005).
Stupi, A. M. et al. Pulmonary hypertension in the CREST syndrome variant of systemic sclerosis. Arthritis Rheum. 29, 515–524 (1986).
Mukerjee, D. et al. Prevalence and outcome in systemic sclerosis associated pulmonary arterial hypertension: application of a registry approach. Ann. Rheum. Dis. 62, 1088–1093 (2003).
Benza, R. L. et al. An evaluation of long-term survival from time of diagnosis in pulmonary arterial hypertension from the REVEAL Registry. Chest 142, 448–456 (2012).
Tyndall, A. J. et al. Causes and risk factors for death in systemic sclerosis: a study from the EULAR Scleroderma Trials and Research (EUSTAR) database. Ann. Rheum. Dis. 69, 1809–1815 (2010).
D'Alto, M. & Mahadevan, V. S. Pulmonary arterial hypertension associated with congenital heart disease. Eur. Respir. Rev. 21, 328–337 (2012).
Duffels, M. G. et al. Pulmonary arterial hypertension in congenital heart disease: an epidemiologic perspective from a Dutch registry. Int. J. Cardiol. 120, 198–204 (2007).
van Riel, A. C. et al. Contemporary prevalence of pulmonary arterial hypertension in adult congenital heart disease following the updated clinical classification. Int. J. Cardiol. 174, 299–305 (2014).
Hadengue, A., Benhayoun, M. K., Lebrec, D. & Benhamou, J. P. Pulmonary hypertension complicating portal hypertension: prevalence and relation to splanchnic hemodynamics. Gastroenterology 100, 520–528 (1991).
Colle, I. O. et al. Diagnosis of portopulmonary hypertension in candidates for liver transplantation: a prospective study. Hepatology 37, 401–409 (2003).
Krowka, M. J. et al. Pulmonary hemodynamics and perioperative cardiopulmonary-related mortality in patients with portopulmonary hypertension undergoing liver transplantation. Liver Transpl. 6, 443–450 (2000).
Murray, K. F., Carithers, R. L. Jr & Aasld . AASLD practice guidelines: evaluation of the patient for liver transplantation. Hepatology 41, 1407–1432 (2005).
Parent, F. et al. A hemodynamic study of pulmonary hypertension in sickle cell disease. N. Engl. J. Med. 365, 44–53 (2011).
Fonseca, G. H., Souza, R., Salemi, V. M., Jardim, C. V. & Gualandro, S. F. Pulmonary hypertension diagnosed by right heart catheterisation in sickle cell disease. Eur. Respir. J. 39, 112–118 (2012).
Simonneau, G. et al. Updated clinical classification of pulmonary hypertension. J. Am. Coll. Cardiol 54 (Suppl.), S43–S54 (2009).
Chitsulo, L., Engels, D., Montresor, A. & Savioli, L. The global status of schistosomiasis and its control. Acta Trop. 77, 41–51 (2000).
Lapa, M. et al. Cardiopulmonary manifestations of hepatosplenic schistosomiasis. Circulation 119, 1518–1523 (2009).
Sitbon, O. et al. Prevalence of HIV-related pulmonary arterial hypertension in the current antiretroviral therapy era. Am. J. Respir. Crit. Care Med. 177, 108–113 (2008).
McLaughlin, V. V. et al. ACCF/AHA 2009 expert consensus document on pulmonary hypertension a report of the American College of Cardiology Foundation Task Force on Expert Consensus Documents and the American Heart Association developed in collaboration with the American College of Chest Physicians; American Thoracic Society, Inc.; and the Pulmonary Hypertension Association. J. Am. Coll. Cardiol. 53, 1573–1619 (2009).
Klings, E. S. et al. An official American Thoracic Society clinical practice guideline: diagnosis, risk stratification, and management of pulmonary hypertension of sickle cell disease. Am. J. Respir. Crit. Care Med. 189, 727–740 (2014).
Schwaiger, J. P., Khanna, D. & Gerry Coghlan, J. Screening patients with scleroderma for pulmonary arterial hypertension and implications for other at-risk populations. Eur. Respir. Rev. 22, 515–525 (2013).
Tedford, R. J. et al. Right ventricular dysfunction in systemic sclerosis-associated pulmonary arterial hypertension. Circ. Heart Fail. 6, 953–963 (2013).
Overbeek, M. J. et al. Right ventricular contractility in systemic sclerosis-associated and idiopathic pulmonary arterial hypertension. Eur. Respir. J. 31, 1160–1166 (2008).
Rich, J. D., Shah, S. J., Swamy, R. S., Kamp, A. & Rich, S. Inaccuracy of Doppler echocardiographic estimates of pulmonary artery pressures in patients with pulmonary hypertension: implications for clinical practice. Chest 139, 988–993 (2011).
Fisher, M. R. et al. Accuracy of Doppler echocardiography in the hemodynamic assessment of pulmonary hypertension. Am. J. Respir. Crit. Care Med. 179, 615–621 (2009).
D'Alto, M. et al. Accuracy and precision of echocardiography versus right heart catheterization for the assessment of pulmonary hypertension. Int. J. Cardiol. 168, 4058–4062 (2013).
Mukerjee, D. et al. Echocardiography and pulmonary function as screening tests for pulmonary arterial hypertension in systemic sclerosis. Rheumatology (Oxford) 43, 461–466 (2004).
Hachulla, E. et al. Early detection of pulmonary arterial hypertension in systemic sclerosis: a French nationwide prospective multicenter study. Arthritis Rheum. 52, 3792–3800 (2005).
Coghlan, J. G. et al. Evidence-based detection of pulmonary arterial hypertension in systemic sclerosis: the DETECT study. Ann. Rheum Dis. 73, 1340–1349 (2014).
Galie, N. et al. Guidelines for the diagnosis and treatment of pulmonary hypertension: the Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS), endorsed by the International Society of Heart and Lung Transplantation (ISHLT). Eur. Heart J. 30, 2493–2537 (2009).
Kitabatake, A. et al. Noninvasive evaluation of pulmonary hypertension by a pulsed Doppler technique. Circulation 68, 302–309 (1983).
Arkles, J. S. et al. Shape of the right ventricular Doppler envelope predicts hemodynamics and right heart function in pulmonary hypertension. Am. J. Respir. Crit. Care Med. 183, 268–276 (2011).
Abbas, A. E. et al. A simple method for noninvasive estimation of pulmonary vascular resistance. J. Am. Coll. Cardiol. 41, 1021–1027 (2003).
D'Alto, M. et al. Echocardiographic prediction of pre- versus postcapillary pulmonary hypertension. J. Am. Soc. Echocardiogr. http://dx.doi.org/10.1016/j.echo.2014.09.004.
Leuchte, H. H. et al. Clinical significance of brain natriuretic peptide in primary pulmonary hypertension. J. Am. Coll. Cardiol. 43, 764–770 (2004).
Fijalkowska, A. et al. Serum N-terminal brain natriuretic peptide as a prognostic parameter in patients with pulmonary hypertension. Chest 129, 1313–1321 (2006).
Nagaya, N. et al. Plasma brain natriuretic peptide as a prognostic indicator in patients with primary pulmonary hypertension. Circulation 102, 865–870 (2000).
Blyth, K. G. et al. NT-proBNP can be used to detect right ventricular systolic dysfunction in pulmonary hypertension. Eur. Respir. J. 29, 737–744 (2007).
Williams, M. H. et al. Role of N-terminal brain natriuretic peptide (N-TproBNP) in scleroderma-associated pulmonary arterial hypertension. Eur. Heart J. 27, 1485–1494 (2006).
Mukerjee, D. et al. Significance of plasma N-terminal pro-brain natriuretic peptide in patients with systemic sclerosis-related pulmonary arterial hypertension. Respir. Med. 97, 1230–1236 (2003).
Allanore, Y. et al. N-terminal pro-brain natriuretic peptide as a diagnostic marker of early pulmonary artery hypertension in patients with systemic sclerosis and effects of calcium-channel blockers. Arthritis Rheum. 48, 3503–3508 (2003).
McLaughlin, V. V. et al. Treatment goals of pulmonary hypertension. J. Am. Coll. Cardiol. 62 (Suppl.), D73–D81 (2013).
Luchner, A. et al. Effect of compensated renal dysfunction on approved heart failure markers: direct comparison of brain natriuretic peptide (BNP) and N-terminal pro-BNP. Hypertension 46, 118–123 (2005).
Steen, V. & Medsger, T. A. Jr. Predictors of isolated pulmonary hypertension in patients with systemic sclerosis and limited cutaneous involvement. Arthritis Rheum. 48, 516–522 (2003).
Thakkar, V. et al. The inclusion of N-terminal pro-brain natriuretic peptide in a sensitive screening strategy for systemic sclerosis-related pulmonary arterial hypertension: a cohort study. Arthritis Res. Ther. 15, R193 (2013).
Borland, C. A place for TL,NO with TL,CO? Eur. Respir. J. 31, 918–919 (2008).
Roughton, F. J. & Forster, R. E. Relative importance of diffusion and chemical reaction rates in determining rate of exchange of gases in the human lung, with special reference to true diffusing capacity of pulmonary membrane and volume of blood in the lung capillaries. J. Appl. Physiol. 11, 290–302 (1957).
Overbeek, M. J. et al. Membrane diffusion- and capillary blood volume measurements are not useful as screening tools for pulmonary arterial hypertension in systemic sclerosis: a case control study. Respir. Res. 9, 68 (2008).
Trip, P. et al. Diffusion capacity and BMPR2 mutations in pulmonary arterial hypertension. Eur. Respir. J. 43, 1195–1198 (2014).
Montani, D. et al. Pulmonary veno-occlusive disease. Eur. Respir. J. 33, 189–200 (2009).
Gunther, S. et al. Computed tomography findings of pulmonary venoocclusive disease in scleroderma patients presenting with precapillary pulmonary hypertension. Arthritis Rheum. 64, 2995–3005 (2012).
Sun, X. G., Hansen, J. E., Oudiz, R. J. & Wasserman, K. Pulmonary function in primary pulmonary hypertension. J. Am. Coll. Cardiol. 41, 1028–1035 (2003).
Arena, R., Lavie, C. J., Milani, R. V., Myers, J. & Guazzi, M. Cardiopulmonary exercise testing in patients with pulmonary arterial hypertension: an evidence-based review. J. Heart Lung Transplant. 29, 159–173 (2010).
Trip, P., Vonk-Noordegraaf, A. & Bogaard, H. J. Cardiopulmonary exercise testing reveals onset of disease and response to treatment in a case of heritable pulmonary arterial hypertension. Pulm. Circ. 2, 387–389 (2012).
Raevens, S. et al. Echocardiography for the detection of portopulmonary hypertension in liver transplant candidates: an analysis of cutoff values. Liver Transpl. 19, 602–610 (2013).
Machado, R. F. et al. Hospitalization for pain in patients with sickle cell disease treated with sildenafil for elevated TRV and low exercise capacity. Blood 118, 855–864 (2011).
Ataga, K. I. et al. Pulmonary hypertension in patients with sickle cell disease: a longitudinal study. Br. J. Haematol. 134, 109–115 (2006).
Gladwin, M. T. et al. Pulmonary hypertension as a risk factor for death in patients with sickle cell disease. N. Engl. J. Med. 350, 886–895 (2004).
Soubrier, F. et al. Genetics and genomics of pulmonary arterial hypertension. J. Am. Coll. Cardiol. 62 (Suppl.), D13–D21 (2013).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
Kovacs, G., Berghold, A., Scheidl, S. & Olschewski, H. Pulmonary arterial pressure during rest and exercise in healthy subjects: a systematic review. Eur. Respir. J. 34, 888–894 (2009).
Valerio, C. J., Schreiber, B. E., Handler, C. E., Denton, C. P. & Coghlan, J. G. Borderline mean pulmonary artery pressure in patients with systemic sclerosis: transpulmonary gradient predicts risk of developing pulmonary hypertension. Arthritis Rheum. 65, 1074–1084 (2013).
Brofman, B. L. et al. Unilateral pulmonary artery occlusion in man; control studies. J. Thorac. Surg. 34, 206–227 (1957).
Reed, C. E., Spinale, F. G. & Crawford, F. A. Jr Effect of pulmonary resection on right ventricular function. Ann. Thorac. Surg. 53, 578–582 (1992).
Sasahara, A. A. Pulmonary vascular responses to thromboembolism. Mod. Concepts Cardiovasc. Dis. 36, 55–60 (1967).
Tolle, J. J., Waxman, A. B., Van Horn, T. L., Pappagianopoulos, P. P. & Systrom, D. M. Exercise-induced pulmonary arterial hypertension. Circulation 118, 2183–2189 (2008).
Kovacs, G. et al. Borderline pulmonary arterial pressure is associated with decreased exercise capacity in scleroderma. Am. J. Respir. Crit. Care Med. 180, 881–886 (2009).
Oudiz, R. J. & Rubin, L. J. Exercise-induced pulmonary arterial hypertension: a new addition to the spectrum of pulmonary vascular diseases. Circulation 118, 2120–2121 (2008).
Lang, I. M. & Madani, M. Update on chronic thromboembolic pulmonary hypertension. Circulation 130, 508–518 (2014).
Hoeper, M. M. et al. Chronic thromboembolic pulmonary hypertension. Lancet Respir. Med. 2, 573–582 (2014).
Hoeper, M. M. et al. Diagnosis, assessment, and treatment of non-pulmonary arterial hypertension pulmonary hypertension. J. Am. Coll. Cardiol. 54, S85–96 (2009).
Naeije, R. et al. Exercise-induced pulmonary hypertension: physiological basis and methodological concerns. Am. J. Respir. Crit. Care Med. 187, 576–583 (2013).
Lewis, G. D. et al. Pulmonary vascular hemodynamic response to exercise in cardiopulmonary diseases. Circulation 128, 1470–1479 (2013).
Bae, S. et al. Baseline characteristics and follow-up in patients with normal haemodynamics versus borderline mean pulmonary arterial pressure in systemic sclerosis: results from the PHAROS registry. Ann. Rheum. Dis. 71, 1335–1342 (2012).
Saggar, R. et al. Brief report: effect of ambrisentan treatment on exercise-induced pulmonary hypertension in systemic sclerosis: a prospective single-center, open-label pilot study. Arthritis Rheum. 64, 4072–4077 (2012).
Bossone, E. et al. Echocardiography in pulmonary arterial hypertension: from diagnosis to prognosis. J. Am. Soc. Echocardiogr. 26, 1–14 (2013).
Grunig, E. et al. Stress Doppler echocardiography in relatives of patients with idiopathic and familial pulmonary arterial hypertension: results of a multicenter European analysis of pulmonary artery pressure response to exercise and hypoxia. Circulation 119, 1747–1757 (2009).
Reichenberger, F. et al. Noninvasive detection of early pulmonary vascular dysfunction in scleroderma. Resp. Med. 103, 1713–1718 (2009).
Collins, N. et al. Abnormal pulmonary vascular responses in patients registered with a systemic autoimmunity database: Pulmonary Hypertension Assessment and Screening Evaluation using stress echocardiography (PHASE-I). Eur. J. Echocardiogr. 7, 439–446 (2006).
Alkotob, M. L. et al. Reduced exercise capacity and stress-induced pulmonary hypertension in patients with scleroderma. Chest 130, 176–181 (2006).
Steen, V. et al. Exercise-induced pulmonary arterial hypertension in patients with systemic sclerosis. Chest 134, 146–151 (2008).
Bossone, E., Rubenfire, M., Bach, D. S., Ricciardi, M. & Armstrong, W. F. Range of tricuspid regurgitation velocity at rest and during exercise in normal adult men: implications for the diagnosis of pulmonary hypertension. J. Am. Coll. Cardiol. 33, 1662–1666 (1999).
Argiento, P. et al. Exercise stress echocardiography of the pulmonary circulation: limits of normal and sex differences. Chest 142, 1158–1165 (2012).
Maeder, M. T., Thompson, B. R., Brunner-La Rocca, H. P. & Kaye, D. M. Hemodynamic basis of exercise limitation in patients with heart failure and normal ejection fraction. J. Am. Coll. Cardiol. 56, 855–863 (2010).
Lau, E. M. et al. Dobutamine stress echocardiography for the assessment of pressure-flow relationships of the pulmonary circulation. Chest 146, 959–966 (2014).
Kuriyama, K. et al. CT-determined pulmonary artery diameters in predicting pulmonary hypertension. Invest. Radiol. 19, 16–22 (1984).
Haimovici, J. B. et al. Relationship between pulmonary artery diameter at computed tomography and pulmonary artery pressures at right-sided heart catheterization. Massachusetts General Hospital Lung Transplantation Program. Acad. Radiol. 4, 327–334 (1997).
Ng, C. S., Wells, A. U. & Padley, S. P. A CT sign of chronic pulmonary arterial hypertension: the ratio of main pulmonary artery to aortic diameter. J. Thorac. Imaging 14, 270–278 (1999).
Moledina, S. et al. Fractal branching quantifies vascular changes and predicts survival in pulmonary hypertension: a proof of principle study. Heart 97, 1245–1249 (2011).
Boxt, L. M., Katz, J., Kolb, T., Czegledy, F. P. & Barst, R. J. Direct quantitation of right and left ventricular volumes with nuclear magnetic resonance imaging in patients with primary pulmonary hypertension. J. Am. Coll. Cardiol. 19, 1508–1515 (1992).
Benza, R., Biederman, R., Murali, S. & Gupta, H. Role of cardiac magnetic resonance imaging in the management of patients with pulmonary arterial hypertension. J. Am. Coll. Cardiol. 52, 1683–1692 (2008).
Sanz, J. et al. Evaluation of pulmonary artery stiffness in pulmonary hypertension with cardiac magnetic resonance. JACC Cardiovasc. Imaging 2, 286–295 (2009).
Ohno, Y. et al. Primary pulmonary hypertension: 3D dynamic perfusion MRI for quantitative analysis of regional pulmonary perfusion. Am. J. Roentgenol. 188, 48–56 (2007).
Roeleveld, R. J. et al. A comparison of noninvasive MRI-based methods of estimating pulmonary artery pressure in pulmonary hypertension. J. Magn. Reson. Imaging 22, 67–72 (2005).
Saba, T. S., Foster, J., Cockburn, M., Cowan, M. & Peacock, A. J. Ventricular mass index using magnetic resonance imaging accurately estimates pulmonary artery pressure. Eur. Respir. J. 20, 1519–1524 (2002).
Laffon, E. et al. A computed method for noninvasive MRI assessment of pulmonary arterial hypertension. J. Appl. Physiol. (1985) 96, 463–468 (2004).
Archer, S. L., Weir, E. K. & Wilkins, M. R. Basic science of pulmonary arterial hypertension for clinicians: new concepts and experimental therapies. Circulation 121, 2045–2066 (2010).
Zhao, L. et al. Heterogeneity in lung 18FDG uptake in pulmonary arterial hypertension: potential of dynamic 18FDG positron emission tomography with kinetic analysis as a bridging biomarker for pulmonary vascular remodeling targeted treatments. Circulation 128, 1214–1224 (2013).
Hagan, G. et al. 18FDG PET imaging can quantify increased cellular metabolism in pulmonary arterial hypertension: A proof-of-principle study. Pulm. Circ. 1, 448–455 (2011).
Ruiter, G. et al. Pulmonary 2-deoxy-2-[18F]-fluoro-D-glucose uptake is low in treated patients with idiopathic pulmonary arterial hypertension. Pulm. Circ. 3, 647–653 (2013).
Lau, E. M. et al. Pulmonary hypertension leads to a loss of gravity dependent redistribution of regional lung perfusion: a SPECT/CT study. Heart 100, 47–53 (2014).
Hakim, M. et al. Volatile organic compounds of lung cancer and possible biochemical pathways. Chem. Rev. 112, 5949–5966 (2012).
Peled, N. et al. Non-invasive breath analysis of pulmonary nodules. J. Thorac. Oncol. 7, 1528–1533 (2012).
Peng, G. et al. Diagnosing lung cancer in exhaled breath using gold nanoparticles. Nat. Nanotechnol. 4, 669–673 (2009).
Cohen-Kaminsky, S. et al. A proof of concept for the detection and classification of pulmonary arterial hypertension through breath analysis with a sensor array. Am. J. Respir. Crit. Care Med. 188, 756–759 (2013).
Wilson, J. M. G. & Jungner, G. Principles and Practice of Screening for Disease (WHO, 1968).
Author information
Authors and Affiliations
Contributions
All the authors researched data for the article. E.M.T.L. wrote the manuscript, and all the authors reviewed and edited the article before submission.
Corresponding author
Ethics declarations
Competing interests
E.M.T.L. has received support from Actelion for speaking engagements. M.H. has relationships with the following companies: Actelion, Aires, Bayer, BMS, GSK, Novartis, Pfizer, and United Therapeutics. In addition to being an investigator in trials involving these companies, relationships include consultancy services and membership of scientific advisory boards. D.S.C. has received support from Actelion, including serving on the speaker bureau and funding for research.
Rights and permissions
About this article
Cite this article
Lau, E., Humbert, M. & Celermajer, D. Early detection of pulmonary arterial hypertension. Nat Rev Cardiol 12, 143–155 (2015). https://doi.org/10.1038/nrcardio.2014.191
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2014.191
This article is cited by
-
Perfusion imaging heterogeneity during NO inhalation distinguishes pulmonary arterial hypertension (PAH) from healthy subjects and has potential as an imaging biomarker
Respiratory Research (2022)
-
Prognostic value of 6-min walk stress echocardiography in patients with interstitial lung disease
Journal of Echocardiography (2021)
-
Reduced pulmonary vascular reserve during stress echocardiography in confirmed pulmonary hypertension and patients at risk of overt pulmonary hypertension
The International Journal of Cardiovascular Imaging (2020)
-
Progress in Understanding, Diagnosing, and Managing Cardiac Complications of Systemic Sclerosis
Current Rheumatology Reports (2019)
-
Global Proteomics Deciphered Novel-Function of Osthole Against Pulmonary Arterial Hypertension
Scientific Reports (2018)