Abstract
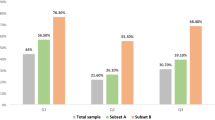
Approximately 120,000 young women are diagnosed with cancer every year in the USA. Many will have treatment that can reduce their fertility, although few will learn this fact before their treatment commences. This presents a tremendous quality of life issue post-treatment, as evidenced in this Perspectives by a personal account from a 23-year-old woman diagnosed with breast cancer. Clinicians must increase awareness about patients' desires for motherhood and awareness about their individual reproductive potential. We demonstrate novel evidence about the wide variability in ovarian reserve in women of similar age, using assessment by antral follicle count. We show how a unified approach between oncology and fertility teams can help patients better understand their risk of treatment-related infertility, as well as how to take effective measures to mitigate it. Finally, we present options for fertility preservation, based on the time point at which consultation occurs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
NCI Fast Stats. Statistics stratified by age. Surveillance Epidemiology and End Results (SEER) [online], (2006).
Loprinzi, C. L., Wolf, S. L., Barton, D. L. & Laack, N. N. Symptom management in premenopausal patients with breast cancer. Lancet Oncol. 9, 993–1001 (2008).
Tschudin, S. & Bitzer, J. Psychological aspects of fertility preservation in men and women affected by cancer and other life-threatening diseases. Hum. Reprod. Update 15, 587–597 (2009).
Forman, E. J., Anders, C. K. & Behera, M. A. A nationwide survey of oncologists regarding treatment-related infertility and fertility preservation in female cancer patients. Fertil. Steril. doi:10.1016/j.fertnstert.2009.10.008.
Quinn, G. P. et al. Physician referral for fertility preservation in oncology patients: a national study of practice behaviors. J. Clin. Oncol. 27, 5952–5957 (2009).
Sonmezer, M. & Oktay, K. Fertility preservation in female cancer patients. Hum. Reprod. Update 10, 251–266 (2004).
Albright, F., Smith, P. H. & Fraser, R. A syndrome characterized by primary ovarian insufficiency and decreased stature. Am. J. Med. Sci. 204, 625–648 (1942).
Nelson, L. M. Primary ovarian insufficiency. N. Engl. J. Med. 360, 606–614 (2009).
Schover, L. R. Patient attitudes toward fertility preservation. Pediatr. Blood Cancer 53, 281–284 (2009).
Carter, J. et al. Gynecologic cancer treatment and the impact of cancer-related infertility. Gynecol. Oncol. 97, 90–95 (2005).
Patel, A. et al. Reproductive health assessment for women with cancer: a pilot study. Am. J. Obstet. Gynecol. 201, 191e1–191e4 (2009).
Bines, J., Oleske, D. M. & Cobleigh, M. A. Ovarian function in premenopausal women treated with adjuvant chemotherapy for breast cancer. J. Clin. Oncol. 14, 1718–1729 (1996).
Hansen, K. R. et al. A new model of reproductive aging: the decline in ovarian non-growing follicle number from birth to menopause. Hum. Reprod. 23, 699–708 (2008).
Rosen, M. et al. Antral follicle count: absence of significant midlife decline. Fertil. Steril. doi:10.1016/j.fertnstert.2009.12.045.
Scheffer, G. J. et al. Antral follicle counts by transvaginal ultrasoundography are related to age in women with proven natural fertility. Fertil. Steril. 72, 845–851 (1999).
Rosen, M. P. et al. Is antral follicle count a genetic trait? Menopause 17, 109–113 (2009).
Schuh-Huerta, S. M. et al Genetic determinants of ovarian aging assessed by antral follicle count. Fertil. Steril. 90, S265–S266 (2008).
Lutchman Singh, K. et al. Predictors of ovarian reserve in young women with breast cancer. Br. J. Cancer. 96, 1808–1816 (2007).
Anders, C. et al. A pilot study of predictive markers of chemotherapy-related amenorrhea among premenopausal women with early stage breast cancer. Cancer Invest. 26, 286–295 (2008).
Larsen, E. C., Muller, J., Rechnitzer, C., Schmiegelow, K. & Andersen, A. N. Diminished ovarian reserve in female childhood cancer survivors with regular menstrual cycles and basal FSH <10 IU/l. Hum. Reprod. 18, 417–422 (2003).
Lie Fong, S. et al. Assessment of ovarian reserve in adult childhood cancer survivors using anti-Mullerian hormone. Hum. Reprod. 24, 982–990 (2009).
Su, H. I. et al. Antimullerian hormone and inhibin B are hormone measures of ovarian function in late reproductive-aged breast cancer survivors. Cancer 116, 592–599 (2010).
Gibreel, A., Maheshwari, A., Bhattacharya, S. & Johnson, N. P. Ultrasound tests of ovarian reserve; a systematic review of accuracy in predicting fertility outcomes. Hum. Fertil. (Camb.) 12, 95–106 (2009).
de Boer, E. J. et al. A low number of retrieved oocytes at in vitro fertilization is predictive of early menopause. Fertil. Steril. 77, 978–985 (2002).
Azim, A. & Oktay, K. Letrozole for ovulation induction and fertility preservation by embryo cryopreservation in young women with endometrial carcinoma. Fertil. Steril. 88, 657–664 (2007).
Azim, A. A., Costantini-Ferrando, M. & Oktay, K. Safety of fertility preservation by ovarian stimulation with letrozole and gonadotropins in patients with breast cancer: a prospective controlled study. J. Clin. Oncol. 26, 2630–2635 (2008).
Lohrisch, C. et al. Impact on survival of time from definitive surgery to initiation of adjuvant chemotherapy for early-stage breast cancer. J. Clin. Oncol. 24, 4888–4894 (2006).
von Wolff, M. et al. Ovarian stimulation to cryopreserve fertilized oocytes in cancer patients can be started in the luteal phase. Fertil. Steril. 92, 1360–1365 (2009).
Madrigrano, A., Westphal, L. & Wapnir, I. Egg retrieval with cryopreservation does not delay breast cancer treatment. Am. J. Surg. 194, 477–481 (2007).
Lee, S. J. et al. American Society of Clinical Oncology recommendations on fertility preservation in cancer patients. J. Clin. Oncol. 24, 2917–2931 (2006).
Georgescu, E. S., Goldberg, J. M., du Plessis, S. S. & Agarwal, A. Present and future fertility preservation strategies for female cancer patients. Obstet. Gynecol. Surv. 63, 725–732 (2008).
Oktay, K., Cil, A. P. & Bang, H. Efficiency of oocyte cryopreservation: a meta-analysis. Fertil. Steril. 86, 70–80 (2006).
Cao, Y. & Chian, R. Fertility preservation with immature and in vitro matured oocytes. Sem. Reprod. Med. 27, 456–464.
Meirow, D. et al. Pregnancy after transplantation of cryopreserved ovarian tissue in a patient with ovarian failure after chemotherapy. N. Engl. J. Med. 353, 318–321 (2005).
Reinblatt, S. L. & Buckett, W. In vitro maturation for patients with polycystic ovary syndrome. Semin. Reprod. Med. 26, 121–126 (2008).
Badaway, A., Elnashar, A., El-Ashry, M. & Shahat, M. Gonadotropin-releasing hormone agonists for prevention of chemotherapy-induced ovarian damage: prospective randomized study. Fertil. Steril. 91, 694–697 (2009).
Oktay, K. & Sönmezer, M. Questioning GnRH analogs for gonadal protection in cancer patients. Fertil. Steril. 92, e32 (2009).
Behringer, K. et al. No protection of the ovarian follicle pool with the use of GnRH-analogues or oral contraceptives in young women treated with escalated BEACOPP for advanced-stage Hodgkin lymphoma. Final results of a phase II clinical trial from the German Hodgkin Study Group. Ann. Oncol. doi:10.1093/annonc/mdq066.
Ginsburg, E. S. et al. In vitro fertilization for cancer patients and survivors. Fertil. Steril. 75, 705–710 (2001).
von Wolff, M. et al. Cryopreservation and autotransplantation of human ovarian tissue prior to cytotoxic therapy—a technique in its infancy but already successful in fertility preservation. Eur. J. Cancer 45, 1547–1553 (2009).
Acknowledgements
This project was supported by NIH/NICHD and NIH/NIA Grant Number R01 HD044876 and by NIH/NCRR UCSF-CTSI Grant Number UL1 RR024131. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH. Mr Letourneau's work was supported by a grant from the UCSF PACCTR Medical Student Research Fellowship. We would like to thank our patient for her generous and heartfelt submission. Without her work this manuscript would not have been possible.
Author information
Authors and Affiliations
Contributions
M. E. Melisko provided a substantial contribution to discussions of the content and to review and/or editing of the manuscript before submission. M. I. Cedar researched the data for the article and provided a substantial contribution to discussions of the content. J. M. Letourneau and M. P. Rosen contributed equally to research, discussion of content and writing the article and to review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Letourneau, J., Melisko, M., Cedars, M. et al. A changing perspective: improving access to fertility preservation. Nat Rev Clin Oncol 8, 56–60 (2011). https://doi.org/10.1038/nrclinonc.2010.133
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2010.133
This article is cited by
-
Oocyte cryopreservation review: outcomes of medical oocyte cryopreservation and planned oocyte cryopreservation
Reproductive Biology and Endocrinology (2022)
-
Update Knowledge Assessment and Influencing Predictor of Female Fertility Preservation in Oncologists
Current Medical Science (2022)
-
Deliveries following fertility preservation by ovarian tissue cryopreservation without autotransplantation—what should be expected?
Journal of Assisted Reproduction and Genetics (2019)
-
Human umbilical cord stem cell conditioned medium versus serum-free culture medium in the treatment of cryopreserved human ovarian tissues in in-vitro culture: a randomized controlled trial
Stem Cell Research & Therapy (2017)
-
Cancer survivors of gynecologic malignancies are at risk for decreased opportunity for fertility preservation
Contraception and Reproductive Medicine (2017)