Key Points
-
Despite progress in antiplatelet therapy, a large amount of unmet clinical need continues to exist. According to current estimates, >60 million people in the United States alone have one or more forms of cardiovascular disease and a high proportion of these individuals are at an increased risk of arterial thrombosis.
-
Antithrombotics are the most rapidly growing sector of the cardiovascular market, with sales around US $9.1 billion in 2001 that are forecast to grow to US $22 billion by the year 2007. One of the major factors underpinning this growth is the rapid increase in sales of the antiplatelet drug clopidogrel, which are increasing by an average of 65% per year at present.
-
Although aspirin reduces acute vascular events in approximately 25% of patients with vascular disease, the residual risk remains high, justifying the need to develop more effective antithrombotic agents or use combination antiplatelet therapies.
-
An important lesson that has emerged from numerous antithrombotic trials is that increased antithrombotic potency per se does not necessarily guarantee enhanced clinical benefit and that, in general, potent antithrombotic approaches must be reserved for high-risk patient populations.
-
Bleeding represents the most important factor influencing long-term compliance and overall clinical benefit of antithrombotic therapy. The key concern regarding all existing antithrombotic drugs relates to their therapeutic window. Ideally, antithrombotic approaches should target one or more processes that are crucial for pathological thrombosis, but which are less important for haemostasis. Despite intensive effort, major progress in this area has been limited.
-
Novel antithrombotic approaches are likely to evolve from an improved understanding of the mechanisms regulating thrombus formation. In this context, there have been considerable recent advances, largely gained from the development of novel experimental techniques enabling real-time analysis of arterial thrombus formation in vivo.
-
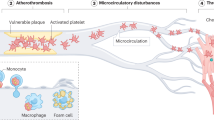
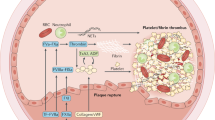
The factors contributing to an exaggerated platelet response at sites of atherosclerotic plaque rupture are multifactorial: the net result of changes in platelet reactivity; plaque thrombogenicity; rheological disturbances and the breakdown of the normal control mechanisms dampening platelet activation.
-
Future advances in antiplatelet therapy are likely to derive from improvements in the understanding of the molecular events regulating thrombogenesis. In this context, the determination of factors specifically promoting an exaggerated platelet response at sites of atherosclerotic plaque rupture could provide a rational basis for the design and development of novel antiplatelet agents with improved efficacy/side-effect profiles.
Abstract
The central importance of platelets in the development of arterial thrombosis and cardiovascular disease is well established. No other single cell type is responsible for as much morbidity and mortality as the platelet and, as a consequence, it represents a major target for therapeutic intervention. The growing awareness of the importance of platelets is reflected in the increasing number of patients receiving antiplatelet therapy, a trend that is likely to continue in the future. There are, however, significant drawbacks with existing therapies, including issues related to limited efficacy and safety. The discovery of a 'magic bullet' that selectively targets pathological thrombus formation without undermining haemostasis remains elusive, although recent progress in unravelling the molecular events regulating thrombosis has provided promising new avenues to solve this long-standing problem.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Fuster, V., Badimon, L., Badimon, J. J. & Chesebro, J. H. The pathogenesis of coronary artery disease and the acute coronary syndromes (1). N. Engl. J. Med. 326, 242–250 (1992).
Falk, E., Shah, P. K. & Fuster, V. Coronary plaque disruption. Circulation 92, 657–671 (1995).
Bhatt, D. L. & Topol, E. J. Scientific and therapeutic advances in antiplatelet therapy. Nature Rev. Drug Discov. 2, 15–28 (2003). A comprehensive and up-to-date review on the current status of antiplatelet therapy.
Schror, K. Antiplatelet drugs. A comparative review. Drugs 50, 7–28 (1995).
Awtry, E. H. & Loscalzo, J. in Platelets (ed. Michelson, A. D.) 745–759 (Academic, San Diego, USA, 2002).
Solet, D. J., Zacharski, L. R. & Plehn, J. F. The role of adenosine 5′-diphosphate receptor blockade in patients with cardiovascular disease. Am. J. Med. 111, 45–53 (2001).
Becker, R. C. Platelet surface physiology and its importance in pharmacotherapy design and development: the adenosine diphosphate receptor antagonists. J. Thromb. Thrombolysis. 10, 35–53 (2000).
Gachet, C. ADP receptors of platelets and their inhibition. Thromb. Haemost. 86, 222–232 (2001).
Sharis, P. J., Cannon, C. P. & Loscalzo, J. The antiplatelet effects of ticlopidine and clopidogrel. Ann. Intern. Med. 129, 394–405 (1998).
Curtin, R., Cox, D. & Fitzgerald, D. in Platelets (ed. Michelson, A. D.) 787–797 (Academic, San Diego, USA, 2002).
Movsesian, M. A. Therapeutic potential of cyclic nucleotide phosphodiesterase inhibitors in heart failure. Expert Opin. Investig. Drugs 9, 963–73 (2000).
Ikeda, Y., Sudo, T. & Kimura, Y. in Platelets (ed. Michelson, A. D.) 817–820 (Academic, San Diego, USA, 2002).
Eisert, W. G. in Platelets (ed. Michelson, A. D.) 803–810 (Academic, San Diego, USA, 2002).
Casserly, I. P. & Topol, E. J. Glycoprotein IIb/IIIa antagonists — from bench to practice. Cell. Mol. Life Sci. 59, 478–500 (2002).
Kam, P. C. & Egan, M. K. Platelet glycoprotein IIb/IIIa antagonists: pharmacology and clinical developments. Anesthesiology 96, 1237–1249 (2002).
Agah, A., Plow, E. F. & Topol, E. J. in Platelets (ed. Michelson, A. D.) 769–781 (Academic, San Diego, USA, 2002).
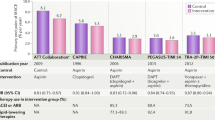
Antithrombotic Trialists' Collaboration. Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ 324, 71–86 (2002). The largest ever meta-analysis of antiplatelet clinical trials demonstrating the importance of antiplatelet therapy in a wide range of cardiovascular diseases.
Birch, S. The Cardiovascular Outlook to 2007 (Datamonitor, PLC, Reuters Business Insight, London, UK, 2002).
A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE). CAPRIE Steering Committee. Lancet 348, 1329–1339 (1996). A landmark study demonstrating the efficacy and safety of clopidogrel relative to aspirin in a diverse group of patients with cardiovascular diseases.
Thebault, J. J., Kieffer, G., Lowe, G. D., Nimmo, W. S. & Cariou, R. Repeated-dose pharmacodynamics of clopidogrel in healthy subjects. Semin. Thromb. Hemost. 25 (Suppl. 2), 9–14 (1999).
Ruggeri, Z. M. Platelets in atherothrombosis. Nature Med. 8, 1227–1234 (2002).
Phillips, D. R., Charo, I. F. & Scarborough, R. M. GPIIb-IIIa: the responsive integrin. Cell 65, 359–362 (1991).
Du, X. & Ginsberg, M. H. Integrin αIIbβ3 and platelet function. Thromb. Haemost. 78, 96–100 (1997).
Plow, E. F., D'Souza, S. E. & Ginsberg, M. H. Ligand binding to GPIIb-IIIa: a status report. Semin. Thromb. Hemost. 18, 324–332 (1992).
Casserly, I. P. & Topol, E. J. Glycoprotein IIb/IIIa antagonists- from bench to practice. Cell. Mol. Life Sci. 59, 478–500 (2002).
Scarborough, R. M., Kleiman, N. S. & Phillips, D. R. Platelet glycoprotein IIb/IIIa antagonists. What are the relevant issues concerning their pharmacology and clinical use? Circulation 100, 437–444 (1999).
Yusuf, S. et al. Effects of clopidogrel in addition to aspirin in patients with acute coronary syndromes without ST-segment elevation. N. Engl. J. Med. 345, 494–502 (2001).
Harrington, R. A. et al. Dose-finding, safety, and tolerability study of an oral platelet glycoprotein IIb/IIIa inhibitor, lotrafiban, in patients with coronary or cerebral atherosclerotic disease. Circulation 102, 728–735 (2000).
Chew, D. P., Bhatt, D. L., Sapp, S. & Topol, E. J. Increased mortality with all platelet glycoprotein IIb/IIIa antagonists: a meta-analysis of phase III multicenter randomized trials. Circulation 103, 201–206 (2001).
Newby, L. K. & McGuire, D. K. Oral platelet glycoprotein IIb/IIIa inhibition. Curr. Cardiol. Rep. 2, 372–377 (2000).
Chew, D. P., Bhatt, D. L., Sapp, S. & Topol, E. J. Increased mortality with oral platelet glycoprotein IIb/IIIa antagonists: a meta-analysis of Phase III multicenter randomized trials. Circulation 103, 201–206 (2001).
Huynh, T., Theroux, P., Bogaty, P., Nasmith, J. & Solymoss, S. Aspirin, warfarin, or the combination for secondary prevention of coronary events in patients with acute coronary syndromes and prior coronary artery bypass surgery. Circulation 103, 3069–3074 (2001).
Fiore, L. D. et al. Department of Veterans Affairs Cooperative Studies Program Clinical Trial comparing combined warfarin and aspirin with aspirin alone in survivors of acute myocardial infarction: primary results of the CHAMP study. Circulation 105, 557–563 (2002).
Randomised double-blind trial of fixed low-dose warfarin with aspirin after myocardial infarction. Coumadin Aspirin Reinfarction Study (CARS) Investigators. Lancet 350, 389–396 (1997).
Falati, S., Gross, P., Merrill-Skoloff, G., Furie, B. C. & Furie, B. Real-time in vivo imaging of platelets, tissue factor and fibrin during arterial thrombus formation in the mouse. Nature Med. 8, 1175–1181 (2002). The first report of the development of a state-of-the-art confocal based imaging technique that enables three-dimensional real-time visualization of thrombus formation in vivo.
Ni, H. et al. Plasma fibronectin promotes thrombus growth and stability in injured arterioles. Proc. Natl Acad. Sci. USA 100, 2415–2419 (2003).
Patel, D. et al. Dynamics of GPIIb/IIIa-mediated platelet-platelet interactions in platelet adhesion/thrombus formation on collagen in vitro as revealed by videomicroscopy. Blood 101, 929–936 (2003).
Kulkarni, S. et al. A revised model of platelet aggregation. J. Clin. Invest. 105, 783–791 (2000).
Ni, H. et al. Persistence of platelet thrombus formation in arterioles of mice lacking both von Willebrand factor and fibrinogen. J. Clin. Invest. 106, 385–392 (2000). This report highlights the existence of a third adhesive ligand promoting platelet aggregation and arterial thrombus formation in mice lacking both vWf and fibrinogen.
Denis, C. et al. A mouse model of severe von Willebrand disease: defects in hemostasis and thrombosis. Proc. Natl Acad. Sci. USA 95, 9524–9529 (1998).
Tronik-Le Roux, D. et al. Thrombasthenic mice generated by replacement of the integrin α(IIb) gene: demonstration that transcriptional activation of this megakaryocytic locus precedes lineage commitment. Blood 96, 1399–1408 (2000).
Smyth, S. S., Reis, E. D., Vaananen, H., Zhang, W. & Coller, B. S. Variable protection of β3-integrin-deficient mice from thrombosis initiated by different mechanisms. Blood 98, 1055–1062 (2001).
Hodivala-Dilke, K. M. et al. β3-integrin-deficient mice are a model for Glanzmann thrombasthenia showing placental defects and reduced survival. J. Clin. Invest. 103, 229–238 (1999).
Yeh, C. H., Chang, M. C., Peng, H. C. & Huang, T. F. Pharmacological characterization and antithrombotic effect of agkistin, a platelet glycoprotein Ib antagonist. Br. J. Pharmacol. 132, 843–850 (2001).
Wu, D., Meiring, M., Kotze, H. F., Deckmyn, H. & Cauwenberghs, N. Inhibition of platelet glycoprotein Ib, glycoprotein IIb/IIIa, or both by monoclonal antibodies prevents arterial thrombosis in baboons. Arterioscler. Thromb. Vasc. Biol. 22, 323–328 (2002).
Ware, J., Russell, S. & Ruggeri, Z. M. Generation and rescue of a murine model of platelet dysfunction: the Bernard-Soulier syndrome. Proc. Natl Acad. Sci. USA 97, 2803–2808 (2000).
Lopez, J. A., Andrews, R. K., Afshar-Kharghan, V. & Berndt, M. C. Bernard-Soulier syndrome. Blood 91, 4397–4418 (1998).
Ramakrishnan, V. et al. Increased thrombin responsiveness in platelets from mice lacking glycoprotein V. Proc. Natl Acad. Sci. USA 96, 13336–13341 (1999).
Moog, S. et al. Platelet glycoprotein V binds to collagen and participates in platelet adhesion and aggregation. Blood 98, 1038–1046 (2001).
Kahn, M. L. et al. Glycoprotein V-deficient platelets have undiminished thrombin responsiveness and do not exhibit a Bernard-Soulier phenotype. Blood 94, 4112–4121 (1999).
Massberg, S. et al. A crucial role of glycoprotein VI for platelet recruitment to the injured arterial wall in vivo. J. Exp. Med. 197, 41–49 (2003).
Kato, K. et al. The contribution of glycoprotein VI to stable platelet adhesion and thrombus formation illustrated by targeted gene deletion. Blood 2003 May 8 [epub ahead of print].
Moroi, M., Jung, S. M., Okuma, M. & Shinmyozu, K. A patient with platelets deficient in glycoprotein VI that lack both collagen-induced aggregation and adhesion. J. Clin. Invest. 84, 1440–1445 (1989).
Konishi, H. et al. Platelets activated by collagen through immunoreceptor tyrosine-based activation motif play pivotal role in initiation and generation of neointimal hyperplasia after vascular injury. Circulation 105, 912–916 (2002).
Nieswandt, B. et al. Glycoprotein VI but not α2β1 integrin is essential for platelet interaction with collagen. EMBO J. 20, 2120–2130 (2001). This study demonstrates the importance of GPVI in promoting platelet adhesion and activation on collagen, and somewhat unexpectedly, the relatively normal haemostatic function of mouse platelets lacking all β1 integrins.
Kehrel, B. et al. Deficiency of intact thrombospondin and membrane glycoprotein Ia in platelets with defective collagen-induced aggregation and spontaneous loss of disorder. Blood 71, 1074–1078 (1988).
Nieuwenhuis, H. K., Akkerman, J. W., Houdijk, W. P. & Sixma, J. J. Human blood platelets showing no response to collagen fail to express surface glycoprotein Ia. Nature 318, 470–472 (1985).
Sadler, J. E. Biochemistry and genetics of von Willebrand factor. Annu. Rev. Biochem. 67, 395–424 (1998).
Sadler, J. E. et al. Impact, diagnosis and treatment of von Willebrand disease. Thromb. Haemost. 84, 160–174 (2000).
Suh, T. T. et al. Resolution of spontaneous bleeding events but failure of pregnancy in fibrinogen-deficient mice. Genes Dev. 9, 2020–2033 (1995).
Holmback, K., Danton, M. J., Suh, T. T., Daugherty, C. C. & Degen, J. L. Impaired platelet aggregation and sustained bleeding in mice lacking the fibrinogen motif bound by integrin αIIbβ3. EMBO J. 15, 5760–5771 (1996).
Myers, D., Jr. et al. Selectins influence thrombosis in a mouse model of experimental deep venous thrombosis. J. Surg. Res. 108, 212–221 (2002).
Falati, S. et al. Accumulation of tissue factor into developing thrombi in vivo is dependent upon microparticle P-selectin glycoprotein ligand 1 and platelet P-selectin. J. Exp. Med. 197, 1585–1598 (2003).
Subramaniam, M. et al. Defects in hemostasis in P-selectin-deficient mice. Blood 87, 1238–1242 (1996).
Vollmar, B., Schmits, R., Kunz, D. & Menger, M. D. Lack of in vivo function of CD31 in vascular thrombosis. Thromb. Haemost. 85, 160–164 (2001).
Falati, S. et al. Opposing effects of P-selectin and PECAM–1 on arterial thrombus growth and stability in vivo. J. Thromb. Haemost. (Suppl. 1), AOC054 (2003).
Mahooti, S. et al. PECAM-1 (CD31) expression modulates bleeding time in vivo. Am. J. Pathol. 157, 75–81 (2000).
Weiss, E. J., Hamilton, J. R., Lease, K. E. & Coughlin, S. R. Protection against thrombosis in mice lacking PAR3. Blood 100, 3240–3244 (2002).
Sambrano, G. R., Weiss, E. J., Zheng, Y. W., Huang, W. & Coughlin, S. R. Role of thrombin signalling in platelets in haemostasis and thrombosis. Nature 413, 1146–1148 (2001). The first demonstration of the importance of platelet thrombin receptors in normal haemostasis and thrombosis.
Fabre, J. E. et al. Decreased platelet aggregation, increased bleeding time and resistance to thromboembolism in P2Y1-deficient mice. Nature Med. 5, 1199–1202 (1999). This paper, and reference 71, describe the first report of P2Y 1 receptor targeting in mouse platelets. These studies demonstrate an important role for this receptor in normal haemostasis and thrombosis. Importantly, the bleeding diathesis in these mice was less severe than that observed in mice lacking the P2Y 12 receptor.
Leon, C. et al. Defective platelet aggregation and increased resistance to thrombosis in purinergic P2Y(1) receptor-null mice. J. Clin. Invest. 104, 1731–1737 (1999).
Leon, C. et al. Key role of the P2Y(1) receptor in tissue factor-induced thrombin-dependent acute thromboembolism: studies in P2Y(1)-knockout mice and mice treated with a P2Y(1) antagonist. Circulation 103, 718–723 (2001).
Foster, C. J. et al. Molecular identification and characterization of the platelet ADP receptor targeted by thienopyridine antithrombotic drugs. J. Clin. Invest. 107, 1591–1598 (2001).
Hollopeter, G. et al. Identification of the platelet ADP receptor targeted by antithrombotic drugs. Nature 409, 202–207 (2001).
Hechler, B. et al. A role of the fast ATP-gated P2X1 cation channel in thrombosis of small arteries in vivo. J. Exp. Med. 198, 661–667 (2003).
Oury, C. et al. A natural dominant negative P2X1 receptor due to deletion of a single amino acid residue. J. Biol. Chem. 275, 22611–22614 (2000).
Thomas, D. W. et al. Coagulation defects and altered hemodynamic responses in mice lacking receptors for thromboxane A2. J. Clin. Invest. 102, 1994–2001 (1998).
Hirata, T., Ushikubi, F., Kakizuka, A., Okuma, M. & Narumiya, S. Two thromboxane A2 receptor isoforms in human platelets. Opposite coupling to adenylyl cyclase with different sensitivity to Arg60 to Leu mutation. J. Clin. Invest. 97, 949–956 (1996).
Hirata, T. et al. Arg60 to Leu mutation of the human thromboxane A2 receptor in a dominantly inherited bleeding disorder. J. Clin. Invest. 94, 1662–1667 (1994).
Ma, H. et al. Increased bleeding tendency and decreased susceptibility to thromboembolism in mice lacking the prostaglandin E receptor subtype EP(3). Circulation 104, 1176–1180 (2001).
Hirsch, E. et al. Resistance to thromboembolism in PI3Kγ-deficient mice. FASEB J. 15, 2019–2021 (2001).
Law, D. A. et al. Integrin cytoplasmic tyrosine motif is required for outside-in αIIbβ3 signalling and platelet function. Nature 401, 808–811 (1999).
Abtahian, F. et al. Regulation of blood and lymphatic vascular separation by signaling proteins SLP-76 and Syk. Science 299, 247–251 (2003).
Clements, J. L. et al. Fetal hemorrhage and platelet dysfunction in SLP-76-deficient mice. J. Clin. Invest. 103, 19–25 (1999).
Judd, B. A. et al. Hematopoietic reconstitution of SLP-76 corrects hemostasis and platelet signaling through αIIbβ3 and collagen receptors. Proc. Natl Acad. Sci. USA 97, 12056–12061 (2000).
Wang, D. et al. Phospholipase Cγ2 is essential in the functions of B cell and several Fc receptors. Immunity 13, 25–35 (2000).
Mangin, P. et al. A PLCγ2-independent platelet collagen aggregation requiring functional association of GPVI and integrin α(2)β(1). FEBS Lett. 542, 53–59 (2003).
Johnson, E. N., Brass, L. F. & Funk, C. D. Increased platelet sensitivity to ADP in mice lacking platelet-type 12-lipoxygenase. Proc. Natl Acad. Sci. USA 95, 3100–3105 (1998).
Langenbach, R. et al. Prostaglandin synthase 1 gene disruption in mice reduces arachidonic acid-induced inflammation and indomethacin-induced gastric ulceration. Cell 83, 483–492 (1995).
Lagarde, M., Byron, P. A., Vargaftig, B. B. & Dechavanne, M. Impairment of platelet thromboxane A2 generation and of the platelet release reaction in two patients with congenital deficiency of platelet cyclo-oxygenase. Br. J. Haematol. 38, 251–266 (1978).
Malmsten, C., Hamberg, M., Svensson, J. & Samuelsson, B. Physiological role of an endoperoxide in human platelets: hemostatic defect due to platelet cyclo-oxygenase deficiency. Proc. Natl Acad. Sci. USA 72, 1446–1450 (1975).
Horellou, M. H. et al. Familial and constitutional bleeding disorder due to platelet cyclo-oxygenase deficiency. Am. J. Hematol. 14, 1–9 (1983).
Offermanns, S., Toombs, C. F., Hu, Y. H. & Simon, M. I. Defective platelet activation in G α(q)-deficient mice. Nature 389, 183–186 (1997). The first demonstration on the fundamental importance of G q and PLC-mediated phosphoinositide turnover in promoting the haemostatic function of platelets.
Kelleher, K. L., Matthaei, K. I. & Hendry, I. A. Targeted disruption of the mouse Gz-α gene: a role for Gz in platelet function? Thromb. Haemost. 85, 529–532 (2001).
Law, D. A. et al. Genetic and pharmacological analyses of Syk function in αIIbβ3 signaling in platelets. Blood 93, 2645–2652 (1999).
Massberg, S. et al. Increased adhesion and aggregation of platelets lacking cyclic guanosine 3′,5′-monophosphate kinase I. J. Exp. Med. 189, 1255–1264 (1999).
Angelillo-Scherrer, A. et al. Deficiency or inhibition of Gas6 causes platelet dysfunction and protects mice against thrombosis. Nature Med. 7, 215–221 (2001). Demonstrates an important role for Gas6 in amplifying platelet activation in response to multiple stimuli. Mice deficient in Gas6 demonstrate defective thrombus formation without prolongation of bleeding time.
Andre, P. et al. CD40L stabilizes arterial thrombi by a β3 integrin-dependent mechanism. Nature Med. 8, 247–252 (2002). This study demonstrates an important role for sCD40L in stabilizing platelet thrombus formation in vivo , through direct binding to integrin αIIbβ3.
Ruggeri, Z. M. von Willebrand factor. J. Clin. Invest. 99, 559–564 (1997).
George, J. N., Sadler, J. E. & Lammle, B. Platelets: thrombotic thrombocytopenic purpura. Hematology (Am. Soc. Hematol. Educ. Program) 315–334 (2002).
Ruggeri, Z. M. Old concepts and new developments in the study of platelet aggregation. J. Clin. Invest. 105, 699–701 (2000).
Savage, B., Almus-Jacobs, F. & Ruggeri, Z. M. Specific synergy of multiple substrate-receptor interactions in platelet thrombus formation under flow. Cell 94, 657–666 (1998).
Savage, B., Saldivar, E. & Ruggeri, Z. M. Initiation of platelet adhesion by arrest onto fibrinogen or translocation on von Willebrand factor. Cell 84, 289–297 (1996). A seminal study highlighting the dynamic nature of platelet adhesion on a vWf substrate under physiologically relevant shear conditions.
Nievelstein, P. F., D'Alessio, P. A. & Sixma, J. J. Fibronectin in platelet adhesion to human collagen types I and III. Use of nonfibrillar and fibrillar collagen in flowing blood studies. Arteriosclerosis 8, 200–206 (1988).
Bonnefoy, A., Harsfalvi, J., Pfliegler, G., Fauvel-Lafeve, F. & Legrand, C. The subendothelium of the HMEC-1 cell line supports thrombus formation in the absence of von Willebrand factor and collagen types I, III and VI. Thromb. Haemost. 85, 552–559 (2001).
Beumer, S. et al. Platelet adhesion to fibronectin in flow: the importance of von Willebrand factor and glycoprotein Ib. Blood 86, 3452–3460 (1995).
Beumer, S., IJesseldijk, M. J., de Groot, P. G. & Sixma, J. J. Platelet adhesion to fibronectin in flow: dependence on surface concentration and shear rate, role of platelet membrane glycoproteins GP IIb/IIIa and VLA-5, and inhibition by heparin. Blood 84, 3724–3733 (1994).
Houdijk, W. P., Sakariassen, K. S., Nievelstein, P. F. & Sixma, J. J. Role of factor VIII-von Willebrand factor and fibronectin in the interaction of platelets in flowing blood with monomeric and fibrillar human collagen types I and III. J. Clin. Invest. 75, 531–540 (1985).
Moroi, M. et al. Analysis of platelet adhesion to a collagen-coated surface under flow conditions: the involvement of glycoprotein VI in the platelet adhesion. Blood 88, 2081–2092 (1996).
Turitto, V. T., Weiss, H. J., Zimmerman, T. S. & Sussman, I. I. Factor VIII/von Willebrand factor in subendothelium mediates platelet adhesion. Blood 65, 823–831 (1985).
Weiss, H. J. et al. Fibrinogen-independent platelet adhesion and thrombus formation on subendothelium mediated by glycoprotein IIb–IIIa complex at high shear rate. J. Clin. Invest. 83, 288–297 (1989).
Ikeda, Y. et al. The role of von Willebrand factor and fibrinogen in platelet aggregation under varying shear stress. J. Clin. Invest. 87, 1234–1240 (1991).
Ruggeri, Z. M., Dent, J. A. & Saldivar, E. Contribution of distinct adhesive interactions to platelet aggregation in flowing blood. Blood 94, 172–178 (1999).
Tsuji, S. et al. Real-time analysis of mural thrombus formation in various platelet aggregation disorders: distinct shear-dependent roles of platelet receptors and adhesive proteins under flow. Blood 94, 968–975 (1999).
Nieswandt, B. & Watson, S. P. Platelet collagen interaction: is GPVI the central receptor? Blood 2003 Mar 20 [epub ahead of print].
van Zanten, G. H. et al. Increased platelet deposition on atherosclerotic coronary arteries. J. Clin. Invest. 93, 615–632 (1994).
Gibbins, J. M., Okuma, M., Farndale, R., Barnes, M. & Watson, S. P. Glycoprotein VI is the collagen receptor in platelets which underlies tyrosine phosphorylation of the Fc receptor γ-chain. FEBS Lett. 413, 255–259 (1997).
Tsuji, M., Ezumi, Y., Arai, M. & Takayama, H. A novel association of Fc receptor γ-chain with glycoprotein VI and their co-expression as a collagen receptor in human platelets. J. Biol. Chem. 272, 23528–23531 (1997).
Nieswandt, B. et al. Expression and function of the mouse collagen receptor glycoprotein VI is strictly dependent on its association with the FcRγ chain. J. Biol. Chem. 275, 23998–24002 (2000).
Berlanga, O. et al. The Fc receptor γ-chain is necessary and sufficient to initiate signalling through glycoprotein VI in transfected cells by the snake C-type lectin, convulxin. Eur. J. Biochem. 269, 2951–2960 (2002).
Goto, S. et al. Involvement of glycoprotein VI in platelet thrombus formation on both collagen and von Willebrand factor surfaces under flow conditions. Circulation 106, 266–272 (2002).
Nieswandt, B. et al. Long-term antithrombotic protection by in vivo depletion of platelet glycoprotein VI in mice. J. Exp. Med. 193, 459–469 (2001).
Kato, K. et al. Impaired thrombosis formation in a murine model of glycoprotein VI deficiency. J. Thromb. Haemost. (Suppl. 1), AOC147 (2003).
Jackson, S. P., Nesbitt, W. S. & Kulkarni, S. Signalling events underlying thromus formation. J. Thromb. Haemost. 1, 1602–1612 (2003).
Nieuwenhuis, H. K., Sakariassen, K. S., Houdijk, W. P., Nievelstein, P. F. & Sixma, J. J. Deficiency of platelet membrane glycoprotein Ia associated with a decreased platelet adhesion to subendothelium: a defect in platelet spreading. Blood 68, 692–695 (1986).
Kroll, M. H. & Schafer, A. I. Biochemical mechanisms of platelet activation. Blood 74, 1181–1195 (1989).
Fressinaud, E., Sakariassen, K. S., Rothschild, C., Baumgartner, H. R. & Meyer, D. Shear rate-dependent impairment of thrombus growth on collagen in nonanticoagulated blood from patients with von Willebrand disease and hemophilia A. Blood 80, 988–994 (1992).
Inauen, W., Baumgartner, H. R., Bombeli, T., Haeberli, A. & Straub, P. W. Dose- and shear rate-dependent effects of heparin on thrombogenesis induced by rabbit aorta subendothelium exposed to flowing human blood. Arteriosclerosis 10, 607–615 (1990).
Gast, A., Tschopp, T. B. & Baumgartner, H. R. Thrombin plays a key role in late platelet thrombus growth and/or stability. Effect of a specific thrombin inhibitor on thrombogenesis induced by aortic subendothelium exposed to flowing rabbit blood. Arterioscler. Thromb. 14, 1466–1474 (1994).
Alevriadou, B. R. et al. Real-time analysis of shear-dependent thrombus formation and its blockade by inhibitors of von Willebrand factor binding to platelets. Blood 81, 1263–1276 (1993).
Barstad, R. M. et al. Reduced effect of aspirin on thrombus formation at high shear and disturbed laminar blood flow. Thromb. Haemost. 75, 827–832 (1996).
Roald, H. E. et al. Modulation of thrombotic responses in moderately stenosed arteries by cigarette smoking and aspirin ingestion. Arterioscler. Thromb. 14, 617–621 (1994).
Barstad, R. M., Roald, H. E., Cui, Y., Turitto, V. T. & Sakariassen, K. S. A perfusion chamber developed to investigate thrombus formation and shear profiles in flowing native human blood at the apex of well-defined stenoses. Arterioscler. Thromb. 14, 1984–1991 (1994).
Coughlin, S. R. How the protease thrombin talks to cells. Proc. Natl Acad. Sci. USA 96, 11023–11027 (1999).
Cattaneo, M. & Gachet, C. The platelet ADP receptors. Haematologica 86, 346–348 (2001).
Gachet, C. Platelet activation by ADP: the role of ADP antagonists. Ann. Med. 32 (Suppl. 1), 15–20 (2000).
Goldsack, N. R., Chambers, R. C., Dabbagh, K. & Laurent, G. J. Thrombin. Int. J. Biochem. Cell Biol. 30, 641–646 (1998).
Coughlin, S. R. & Camerer, E. PARticipation in inflammation. J. Clin. Invest. 111, 25–27 (2003).
Gachet, C. Identification, characterization, and inhibition of the platelet ADP receptors. Int. J. Hematol. 74, 375–381 (2001).
Kahn, M. L. et al. A dual thrombin receptor system for platelet activation. Nature 394, 690–694 (1998).
Rekhter, M. D. Collagen synthesis in atherosclerosis: too much and not enough. Cardiovasc. Res. 41, 376–384 (1999).
Toschi, V. et al. Tissue factor modulates the thrombogenicity of human atherosclerotic plaques. Circulation 95, 594–599 (1997).
Badimon, J. J. et al. Local inhibition of tissue factor reduces the thrombogenicity of disrupted human athersclerotic plaques. Effects of tissue factor pathway inhibitor on plaque thrombogenicity under flow conditions. Circulation 99, 1780–1787 (1999).
Siess, W. Athero- and thrombogenic actions of lysophosphatidic acid and sphingosine-1-phosphate. Biochim. Biophys. Acta 1582, 204–215 (2002).
Siess, W. et al. Lysophosphatidic acid mediates the rapid activation of platelets and endothelial cells by mildly oxidized low density lipoprotein and accumulates in human atherosclerotic lesions. Proc. Natl Acad. Sci. USA 96, 6931–6936 (1999).
Libby, P. Coronary artery injury and the biology of atherosclerosis: Inflammation, thrombosis and stabilization. Am. J. Cardiol. 86 (Suppl.), 3J–9J (2000).
Mallat, Z. et al. Shed membrane microparticles with procoagulant potential in human atherosclerotic plaques. A role for apoptosis in plaque thrombogenicity. Circulation 99, 348–353 (1999).
Trip, M. D., Cats, V. M., van Capelle, F. J. L. & Vreeken, J. Platelet hyperreactivity and prognosis in survivors of myocardial infarction. N. Engl. J. Med. 322, 1549–1554 (1990).
Hirsh, J. Hyperactive platelets and complications of coronary artery disease. N. Engl. J. Med. 316, 1543–1544 (1987).
Roffi, M. et al. Platelet glycoprotein IIb/IIIa inhibitors reduce mortality in diabetic patients with non-ST-segment-elevation acute coronary syndromes. Circulation 104, 2767–2771 (2001).
Lincoff, A. M. Important triad in cardiovascular medicine: diabetes, coronary intervention, and platelet glycoprotein IIb/IIIa receptor blockade. Circulation 107, 1556–1559 (2003).
Kjeldsen, S. E., Rostrup, M., Gjesdal, K. & Eide, I. The epinephrine-blood platelet connection with special reference to essential hypertension. Am. Heart J. 122, 330–336 (1991).
Hamet, P., Skuherska, R., Pang, S. C. & Tremblay, J. Abnormalities of platelet function in hypertension and diabetes. Hypertension 7, II135–II142 (1985).
Opper, C. et al. Increased number of high sensitive platelets in hypercholesterolemia, cardiovascular diseases, and after incubation with cholesterol. Atherosclerosis 277, 15225–15228 (1995).
Terres, W., Becker, P. & Rosenberg, A. Changes in cardiovascular risk profile during the cessation of smoking. Am. J. Med. 97, 242–249 (1994).
Terres, W., Weber, K., Kupper, W. & Bleifeld, W. Age, cardiovascular risk factors and coronary heart disease as determinants of platelet function in men. A multivariate approach. Thromb. Res. 62, 649–661 (1991).
Renaud, S. C., Beswick, A. D., Fehily, A. M., Sharp, D. S. & Elwood, P. C. Alcohol and platelet aggregation: the Caerphilly Prospective Heart Disease Study. Am. J. Clin. Nutr. 55, 1012–1017 (1992).
Mori, T. A., Beilin, L. J., Burke, V., Morris, J. & Ritchie, J. Interactions between dietary fat, fish, and fish oils and their effects on platelet function in men at risk of cardiovascular disease. Arterioscler. Thromb. Vasc. Biol. 17, 279–286 (1997).
Santoso, S. Platelet polymorphisms in thrombotic disorders. Transfus. Clin. Biol. 8, 261–266 (2001).
Gonzalez-Conejero, R. et al. Polymorphisms of platelet membrane glycoprotein Ib associated with arterial thrombotic disease. Blood 92, 2771–2776 (1998).
Baker, R. I. et al. Platelet glycoprotein Ibα Kozak polymorphism is associated with an increased risk of ischemic stroke. Blood 98, 36–40 (2001).
Jilma-Stohlawetz, P. et al. Glycoprotein Ib polymorphisms influence platelet plug formation under high shear rates. Br. J. Haematol. 120, 652–655 (2003).
Burr, D., Doss, H., Cooke, G. E. & Goldschmidt-Clermont, P. J. A meta-analysis of studies on the association of the platelet PlA polymorphism of glycoprotein IIIa and risk of coronary heart disease. Stat. Med. 22, 1741–1760 (2003).
Kunicki, T. J. & Ruggeri, Z. M. Platelet collagen receptors and risk prediction in stroke and coronary artery disease. Circulation 104, 1451–1453 (2001).
Long, Q., Xu, X. Y., Ramnarine, K. V. & Hoskins, P. Numerical investigation of physiologically realistic pulsatile flow through arterial stenosis. J. Biomech. 34, 1229–1242 (2001).
Wootton, D. M. & Ku, D. N. Fluid mechanics of vascular systems, diseases, and thrombosis. Annu. Rev. Biomed. Eng. 1, 299–329 (1999).
Deplano, V. & Siouffi, M. Experimental and numerical study of pulsatile flows through stenosis: wall shear stress analysis. J. Biomech. 32, 1081–1090 (1999).
Moritz, M. W., Reimers, R. C., Baker, R. K., Sutera, S. P. & Joist, J. H. Role of cytoplasmic and releasable ADP in platelet aggregation induced by laminar shear stress. J. Lab. Clin. Med. 101, 537–544 (1983).
Moake, J. L., Turner, N. A., Stathopoulos, N. A., Nolasco, L. & Hellums, J. D. Shear-induced platelet aggregation can be mediated by vWF released from platelets, as well as by exogenous large or unusually large vWF multimers, requires adenosine diphosphate, and is resistant to aspirin. Blood 71, 1366–1374 (1988).
Chow, T. W., Hellums, J. D., Moake, J. L. & Kroll, M. H. Shear stress-induced von Willebrand factor binding to platelet glycoprotein Ib initiates calcium influx associated with aggregation. Blood 80, 113–120 (1992).
Jen, C. J. & McIntire, L. V. Characteristics of shear-induced aggregation in whole blood. J. Lab. Clin. Med. 103, 115–124 (1984).
Konstantopoulos, K. et al. Flow cytometric studies of platelet responses to shear stress in whole blood. Biorheology 32, 73–93 (1995).
Hardwick, R. A., Hellums, J. D., Peterson, D. M., Moake, J. L. & Olson, J. D. The effect of PGI2 and theophylline on the response of platelets subjected to shear stress. Blood 58, 678–681 (1981).
Zambrowicz, B. P. & Sands, A. T. Knockouts model the 100 best-selling drugs — will they model the next 100? Nature Rev. Drug Discov. 2, 38–51 (2003).
Gurevitz, O. et al. S-nitrosoderivative of a recombinant fragment of von Willebrand factor (S-nitroso-AR545C) inhibits thrombus formation in guinea pig carotid artery thrombosis model. Thromb. Haemost. 84, 912–917 (2000).
Gurevitz, O. et al. Recombinant von Willebrand factor fragment AR545C inhibits platelet aggregation and enhances thrombolysis with rtPA in a rabbit thrombosis model. Arterioscler. Thromb. Vasc. Biol. 18, 200–207 (1998).
Kageyama, S. et al. Anti-thrombotic effects and bleeding risk of AJvW-2, a monoclonal antibody against human von Willebrand factor. Br. J. Pharmacol. 122, 165–171 (1997).
Azzam, K., Cisse-Thiam, M. & Drouet, L. The antithrombotic effect of aurin tricarboxylic acid in the guinea pig is not solely due to its interaction with the von Willebrand factor-GPIb axis. Thromb. Haemost. 75, 203–210 (1996).
Chang, M. C., Lin, H. K., Peng, H. C. & Huang, T. F. Antithrombotic effect of crotalin, a platelet membrane glycoprotein Ib antagonist from venom of Crotalus atrox. Blood 91, 1582–1589 (1998).
Kageyama, S., Yamamoto, H., Nakazawa, H. & Yoshimoto, R. Anti-human vWF monoclonal antibody, AJvW-2 Fab, inhibits repetitive coronary artery thrombosis without bleeding time prolongation in dogs. Thromb. Res. 101, 395–404 (2001).
Marcus, A. J. Thrombosis and inflammation as multicellular processes: significance of cell-cell interactions. Semin. Hematol. 31, 261–269 (1994).
Simon, D. I. et al. Platelet glycoprotein ibα is a counterreceptor for the leukocyte integrin Mac-1 (CD11b/CD18). J. Exp. Med. 192, 193–204 (2000).
Romo, G. M. et al. The glycoprotein Ib–IX–V complex is a platelet counterreceptor for P-selectin. J. Exp. Med. 190, 803–814 (1999).
Libby, P. Inflammation in atherosclerosis. Nature 420, 868–874 (2002).
Libby, P., Ridker, P. M. & Maseri, A. Inflammation and atherosclerosis. Circulation 105, 1135–1143 (2002).
Cauwenberghs, N. et al. Antithrombotic effect of platelet glycoprotein Ib-blocking monoclonal antibody Fab fragments in nonhuman primates. Arterioscler. Thromb. Vasc. Biol. 20, 1347–1353 (2000).
Cadroy, Y. et al. Relative antithrombotic effects of monoclonal antibodies targeting different platelet glycoprotein-adhesive molecule interactions in nonhuman primates. Blood 83, 3218–3224 (1994).
Takahashi, R., Sekine, N. & Nakatake, T. Influence of monoclonal antiplatelet glycoprotein antibodies on in vitro human megakaryocyte colony formation and proplatelet formation. Blood 93, 1951–198 (1999).
Alimardani, G., Guichard, J., Fichelson, S. & Cramer, E. M. Pathogenic effects of anti-glycoprotein Ib antibodies on megakaryocytes and platelets. Thromb. Haemost. 88, 1039–1046 (2002).
Wu, D. et al. Inhibition of the von Willebrand (VWF)-collagen interaction by an antihuman VWF monoclonal antibody results in abolition of in vivo arterial platelet thrombus formation in baboons. Blood 99, 3623–3628 (2002).
Ojio, S. et al. Considerable time from the onset of plaque rupture and/or thrombi until the onset of acute myocardial infarction in humans: coronary angiographic findings within 1 week before the onset of infarction. Circulation 102, 2063–2069 (2000). An important study providing angiographic evidence that the majority of patients presenting with an acute myocardial infarction have plaque rupture and superimposed non-occlusive thrombus formation, up to one week before hospital presentation.
Smith, J. B. & Willis, A. L. Aspirin selectively inhibits prostaglandin production in human platelets. Nature New Biol. 231, 235–237 (1971).
Inhibition of vascular and platelet prostaglandin synthesis by aspirin. N. Engl. J. Med. 309, 670–672 (1983).
Randomized trial of ridogrel, a combined thromboxane A2 synthase inhibitor and thromboxane A2/prostaglandin endoperoxide receptor antagonist, versus aspirin as adjunct to thrombolysis in patients with acute myocardial infarction. The Ridogrel Versus Aspirin Patency Trial (RAPT). Circulation 89, 588–595 (1994).
Michaux, C. et al. Terbogrel, a dual-acting agent for thromboxane receptor antagonism and thromboxane synthase inhibition. Acta Crystallogr. C 56, 1265–1266 (2000).
Soyka, R., Guth, B. D., Weisenberger, H. M., Luger, P. & Muller, T. H. Guanidine derivatives as combined thromboxane A2 receptor antagonists and synthase inhibitors. J. Med. Chem. 42, 1235–1249 (1999).
Langleben, D. et al. Effects of the thromboxane synthetase inhibitor and receptor antagonist terbogrel in patients with primary pulmonary hypertension. Am. Heart J. 143, E4 (2002).
Mangin, P. et al. Signaling role for PLCγ 2 in platelet glycoprotein Ibα calcium flux and cytoskeletal reorganization involvement of a pathway distinct from FcR γ-chain and cγ RIIA. J. Biol. Chem. 2003 Jun 17 [epub ahead of print].
Andre, P., Nannizzi-Alaimo, L., Prasad, S. K. & Phillips, D. R. Platelet-derived CD40L: the switch-hitting player of cardiovascular disease. Circulation 106, 896–899 (2002).
Mangano, D. T. Aspirin and mortality from coronary bypass surgery. N. Engl. J. Med. 347, 1309–1317 (2002).
Grotta, J. C. & Alexandrov, A. V. Preventing stroke: is preventing microemboli enough? Circulation 103, 2321–2322 (2001).
Acknowledgements
The authors wish to thank Drs Zaverio Ruggeri, Christian Gachet, Hatem Salem and Francois Lanza, as well as other members of the authors' laboratory for valuable comments.
Author information
Authors and Affiliations
Corresponding author
Related links
Related links
DATABASES
LocusLink
Online Mendelian Inheritance in Man
FURTHER INFORMATION
Glossary
- BLEEDING TIME
-
The time taken for cessation of bleeding from a standardized injury, which is used to test for platelet abnormalities (acquired and congenital) and also for von Willebrand's disease.
- THERAPEUTIC WINDOW
-
The dose (or concentration) range between the minimum effective dose (concentration) and the minimum toxic dose (concentration).
- RHEOLOGICAL ENVIRONMENT
-
The blood-flow characteristics operating within defined areas of the vasculature.
- THROMBOTIC THROMBOCYTOPAENIC PURPURA
-
A rare microvascular thrombotic disorder associated with low platelet count, fever, neurological and renal impairment and micro-angiopathic haemolytic anaemia.
- DIATHESIS
-
A permanent (hereditary or acquired) condition that predisposes the affected individual to certain diseases.
- THROMBOCYTOPAENIA
-
A decrease in platelet count, defined in humans as less than 150 × 109 per l, which results in the potential for increased bleeding.
- PHARMACOPHORE
-
The ensemble of steric and electronic features that are necessary to ensure optimal interactions with a specific biological target structure and to trigger (or to block) its biological response.
- CAROTID ENDARTERECTOMY
-
A surgical procedure that removes atherosclerotic plaque from the walls of the carotid arteries in an attempt to reduce the risk of stroke or transient ischaemic attack.
- PERCUTANEOUS CORONARY INTERVENTION
-
A group of techniques that can relieve coronary artery narrowing. The techniques include balloon angioplasty, laser angioplasty and implantation of intracoronary stents and other catheter devices.
Rights and permissions
About this article
Cite this article
Jackson, S., Schoenwaelder, S. Antiplatelet therapy: in search of the 'magic bullet'. Nat Rev Drug Discov 2, 775–789 (2003). https://doi.org/10.1038/nrd1198
Issue Date:
DOI: https://doi.org/10.1038/nrd1198
This article is cited by
-
Platelet CFTR inhibition enhances arterial thrombosis via increasing intracellular Cl− concentration and activation of SGK1 signaling pathway
Acta Pharmacologica Sinica (2022)
-
Design, synthesis and evaluation of new chromone-derived aminophosphonates as potential acetylcholinesterase inhibitor
Molecular Diversity (2021)
-
In vitro assessment and phase I randomized clinical trial of anfibatide a snake venom derived anti-thrombotic agent targeting human platelet GPIbα
Scientific Reports (2021)
-
The chimeric monoclonal antibody MHCSZ-123 against human von Willebrand factor A3 domain inhibits high-shear arterial thrombosis in a Rhesus monkey model
Journal of Hematology & Oncology (2017)
-
A novel ruthenium (II)-derived organometallic compound, TQ-6, potently inhibits platelet aggregation: Ex vivo and in vivo studies
Scientific Reports (2017)