Key Points
-
Increased antimalarial drug discovery efforts are urgently needed. The goal must be to develop safe and affordable new drugs to counter the spread of malaria parasites resistant to existing agents. Here, we review different in vitro and in vivo screens for antimalarial drug discovery and recommend a streamlined process for evaluating new compounds on a critical path from drug discovery to development.
-
Malaria caused by Plasmodium falciparum remains one of the most important diseases of the developing world, killing 1–3 million people and causing disease in 300–500 million people each year. Malaria-endemic regions of the world are faced with an unprecedented situation in which the only affordable treatment options, historically based on the use of chloroquine or sulphadoxine-pyrimethamine, are rapidly losing therapeutic efficacy.
-
Ideally, new drugs for uncomplicated P. falciparum malaria must be efficacious against drug resistant strains, provide cure within a reasonable time (ideally three days or less) to ensure good compliance, be safe, be suitable for small children and pregnant women, have appropriate formulations for oral use, and above all be affordable. Drug discovery necessarily requires trade-offs among desired drug features, but for the treatment of malaria in the developing world, the provision of affordable, orally active treatments that are safe for children is, for practical purposes, mandatory.
-
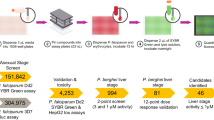
In vitro screens for compound activity are performed with P. falciparum strains of differing drug-susceptibility profiles that can be readily cultured in human erythrocytes. The most common screen involves measuring the uptake of 3H-hypoxanthine (which is taken up by the parasite for purine salvage and DNA synthesis) during a 48–72 hour period. This enables a determination of the level of P. falciparum growth inhibition as a function of the concentration of the test compound.
-
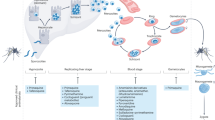
In vivo evaluation of antimalarial compounds typically begins with the use of rodent malaria parasites, either P. berghei, P. yoelii, P. chabaudi or P. vinckei. P. berghei is the most commonly used model. The most widely used initial screen is a four-day suppressive test, where the efficacy of four daily doses of compounds is measured by comparison of blood parasitaemia (on day four after infection) and mouse survival time in treated and untreated mice. Active compounds can be progressed through several secondary tests.
-
This review also provides an example of a critical path for an antimalarial drug discovery effort, lists targets currently being pursued, summarizes ongoing drug discovery and developments efforts and provides online links to informative websites.
Abstract
Increased efforts in antimalarial drug discovery are urgently needed. The goal must be to develop safe and affordable new drugs to counter the spread of malaria parasites that are resistant to existing agents. Drug efficacy, pharmacology and toxicity are important parameters in the selection of compounds for development, yet little attempt has been made to review and standardize antimalarial drug-efficacy screens. Here, we suggest different in vitro and in vivo screens for antimalarial drug discovery and recommend a streamlined process for evaluating new compounds on the path from drug discovery to development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Greenwood, B. & Mutabingwa, T. Malaria in 2002. Nature 415, 670–672 (2002).
White, N. J. The treatment of malaria. N. Engl. J. Med. 335, 800–806 (1996).
Pagola, S., Stephens, P. W., Bohle, D. S., Kosar, A. D. & Madsen, S. K. The structure of malaria pigment β-haematin. Nature 404, 307–310 (2000).
Ursos, L. M. & Roepe, P. D. Chloroquine resistance in the malarial parasite, Plasmodium falciparum. Med. Res. Rev. 22, 465–491 (2002).
Wellems, T. E. & Plowe, C. V. Chloroquine-resistant malaria. J. Infect. Dis. 184, 770–776 (2001).
Trape, J. F. The public health impact of chloroquine resistance in Africa. Am. J. Trop. Med. Hyg. 64, 12–17 (2001).
Plowe, C. V. Monitoring antimalarial drug resistance: making the most of the tools at hand. J. Exp. Biol. 206, 3745–3752 (2003).
Olliaro, P. et al. Systematic review of amodiaquine treatment in uncomplicated malaria. Lancet 348, 1196–1201 (1996).
Winstanley, P. Modern chemotherapeutic options for malaria. Lancet Infect. Dis. 1, 242–250 (2001). A useful review of current options for the treatment of malaria in the developing world.
Lang, T. & Greenwood, B. The development of Lapdap, an affordable new treatment for malaria. Lancet Infect. Dis. 3, 162–168 (2003). Describes the development of the newest approved antimalarial drug, an affordable combination regimen developed specifically for the treatment of drug-resistant infections in Africa.
Ridley, R. G. Medical need, scientific opportunity and the drive for antimalarial drugs. Nature 415, 686–693 (2002). Summarizes the current antimalarial treatment situation and promising new targets for chemotherapy.
Nwaka, S. & Ridley, R. G. Virtual drug discovery and development for neglected diseases through public–private partnerships. Nature Rev. Drug Discov. 2, 919–928 (2003). Reviews the public–private partnership model for virtual drug discovery and development for malaria and other neglected diseases.
Guerin, P. J. et al. Malaria: current status of control, diagnosis, treatment, and a proposed agenda for research and development. Lancet Infect. Dis. 2, 564–573 (2002). A summary of current state-of-the-art management of malaria in the developing world.
Dorsey, G., Vlahos, J., Kamya, M. R., Staedke, S. G. & Rosenthal, P. J. Prevention of increasing rates of treatment failure by combining sulfadoxine-pyrimethamine with artesunate or amodiaquine for the sequential treatment of malaria. J. Infect. Dis. 188, 1231–1238 (2003).
Looareesuwan, S., Chulay, J. D., Canfield, C. J. & Hutchinson, D. B. Malarone (atovaquone and proguanil hydrochloride): a review of its clinical development for treatment of malaria. Malarone Clinical Trials Study Group. Am. J. Trop. Med. Hyg. 60, 533–541 (1999). This paper is a detailed account of the development of one of the newest available antimalarial drugs. Note, however, that Malarone is much too expensive for widescale use in developing countries.
Staedke, S. G. et al. Amodiaquine, sulfadoxine/ pyrimethamine, and combination therapy for treatment of uncomplicated falciparum malaria in Kampala, Uganda: a randomised trial. Lancet 358, 368–374 (2001).
Schellenberg, D. et al. The safety and efficacy of sulfadoxine-pyrimethamine, amodiaquine, and their combination in the treatment of uncomplicated Plasmodium falciparum malaria. Am. J. Trop. Med. Hyg. 67, 17–23 (2002).
Dorsey, G. et al. Sulfadoxine/pyrimethamine alone or with amodiaquine or artesunate for treatment of uncomplicated malaria: a longitudinal randomised trial. Lancet 360, 2031–2038 (2002).
White, N. J. & Pongtavornpinyo, W. The de novo selection of drug-resistant malaria parasites. Proc. R. Soc. Lond. B 270, 545–554 (2003).
Nosten, F. et al. Effects of artesunate-mefloquine combination on incidence of Plasmodium falciparum malaria and mefloquine resistance in western Thailand: a prospective study. Lancet 356, 297–302 (2000).
White, N. J. et al. Averting a malaria disaster. Lancet 353, 1965–1967 (1999). A call to arms regarding the urgent need for new therapies for malaria in Africa.
Adjuik, M. et al. Amodiaquine-artesunate versus amodiaquine for uncomplicated Plasmodium falciparum malaria in African children: a randomised, multicentre trial. Lancet 359, 1365–1372 (2002).
van Vugt, M. et al. Artemether-lumefantrine for the treatment of multidrug-resistant falciparum malaria. Trans. R. Soc. Trop. Med. Hyg. 94, 545–548 (2000).
Rosenthal, P. J., Sijwali, P. S., Singh, A. & Shenai, B. R. Cysteine proteases of malaria parasites: targets for chemotherapy. Curr. Pharm. Des. 8, 1659–1672 (2002).
Chakrabarti, D. et al. Protein farnesyltransferase and protein prenylation in Plasmodium falciparum. J. Biol. Chem. 277, 42066–42073 (2002).
Rotella, D. P. Osteoporosis: challenges and new opportunities for therapy. Curr. Opin. Drug Discov. Devel. 5, 477–486 (2002).
Gelb, M. H. & Hol, W. G. Parasitology. Drugs to combat tropical protozoan parasites. Science 297, 343–344 (2002).
Ralph, S. A., D'Ombrain, M. C. & McFadden, G. I. The apicoplast as an antimalarial drug target. Drug Resist. Update 4, 145–151 (2001).
Waller, R. F. et al. Nuclear-encoded proteins target to the plastid in Toxoplasma gondii and Plasmodium falciparum. Proc. Natl. Acad. Sci. USA 95, 12352–12357 (1998).
Jomaa, H. et al. Inhibitors of the nonmevalonate pathway of isoprenoid biosynthesis as antimalarial drugs. Science 285, 1573–1576 (1999). This study is an excellent example (as is reference 32) of how an improved understanding of the molecular biology and biochemistry of Plasmodium parasites, coupled to pragmatic chemistry, can identify novel antimalarial compounds.
Missinou, M. A. et al. Fosmidomycin for malaria. Lancet 360, 1941–1942 (2002).
Surolia, N. & Surolia, A. Triclosan offers protection against blood stages of malaria by inhibiting enoyl-ACP reductase of Plasmodium falciparum. Nature Med. 7, 167–173 (2001).
Perozzo, R. et al. Structural elucidation of the specificity of the antibacterial agent triclosan for malarial enoyl ACP reductase. J. Biol. Chem. 277, 13106–13114 (2002).
Waller, K. L., Lee, S. & Fidock, D. A. in Genomes and the Molecular Cell Biology of Malaria Parasites (eds Waters, A. P. & Janse, C. J.) 501–540 (Horizon, New York, 2004).
Biagini, G. A., O'Neill, P. M., Nzila, A., Ward, S. A. & Bray, P. G. Antimalarial chemotherapy: young guns or back to the future? Trends Parasitol. 19, 479–487 (2003).
Gardner, M. J. et al. Genome sequence of the human malaria parasite Plasmodium falciparum. Nature 419, 498–511 (2002).
Kissinger, J. C. et al. The Plasmodium genome database. Nature 419, 490–492 (2002).
Wu, Y., Kirkman, L. A. & Wellems, T. E. Transformation of Plasmodium falciparum malaria parasites by homologous integration of plasmids that confer resistance to pyrimethamine. Proc. Natl. Acad. Sci. USA 93, 1130–1134 (1996).
Fidock, D. A. & Wellems, T. E. Transformation with human dihydrofolate reductase renders malaria parasites insensitive to WR99210 but does not affect the intrinsic activity of proguanil. Proc. Natl. Acad. Sci. USA 94, 10931–10936 (1997).
van Dijk, M. R., Janse, C. J. & Waters, A. P. Expression of a Plasmodium gene introduced into subtelomeric regions of Plasmodium berghei chromosomes. Science 271, 662–665 (1996).
Crabb, B. S. et al. Targeted gene disruption shows that knobs enable malaria-infected red cells to cytoadhere under physiological shear stress. Cell 89, 287–296 (1997).
Sidhu, A. B. S., Verdier-Pinard, D. & Fidock, D. A. Chloroquine resistance in Plasmodium falciparum malaria parasites conferred by pfcrt mutations. Science 298, 210–213 (2002).
Waters, A. P., Thomas, A. W., van Dijk, M. R. & Janse, C. J. Transfection of malaria parasites. Methods 13, 134–147 (1997).
Peters, W. & Robinson, B. L. in Handbook of Animal Models of Infection (eds. Zak., O. & Sande, M.) 757–773 (Academic, London, 1999).
Rathod, P. K., Ganesan, K., Hayward, R. E., Bozdech, Z. & DeRisi, J. L. DNA microarrays for malaria. Trends Parasitol. 18, 39–45 (2002).
Bozdech, Z. et al. The transcriptome of the intraerythrocytic developmental cycle of Plasmodium falciparum. PLoS Biol. 1, E5 (2003).
Le Roch, K. G. et al. Discovery of gene function by expression profiling of the malaria parasite life cycle. Science 301, 1503–1508 (2003).
Lasonder, E. et al. Analysis of the Plasmodium falciparum proteome by high-accuracy mass spectrometry. Nature 419, 537–542 (2002).
Florens, L. et al. A proteomic view of the Plasmodium falciparum life cycle. Nature 419, 520–526 (2002).
Greenbaum, D. C. et al. A role for the protease falcipain 1 in host cell invasion by the human malaria parasite. Science 298, 2002–2006 (2002).
Noedl, H., Wongsrichanalai, C. & Wernsdorfer, W. H. Malaria drug-sensitivity testing: new assays, new perspectives. Trends Parasitol. 19, 175–181 (2003). Summarizes of P. falciparum in vitro drug sensitivity tests.
Makler, M. T. & Hinrichs, D. J. Measurement of the lactate dehydrogenase activity of Plasmodium falciparum as an assessment of parasitemia. Am. J. Trop. Med. Hyg. 48, 205–210 (1993).
van der Heyde, H. C., Elloso, M. M., vande Waa, J., Schell, K. & Weidanz, W. P. Use of hydroethidine and flow cytometry to assess the effects of leukocytes on the malarial parasite Plasmodium falciparum. Clin. Diagn. Lab. Immunol. 2, 417–425 (1995).
ter Kuile, F., White, N. J., Holloway, P., Pasvol, G. & Krishna, S. Plasmodium falciparum: in vitro studies of the pharmacodynamic properties of drugs used for the treatment of severe malaria. Exp. Parasitol. 76, 85–95 (1993).
Ofulla, A. O. et al. Determination of fifty percent inhibitory concentrations (IC50) of antimalarial drugs against Plasmodium falciparum parasites in a serum-free medium. Am. J. Trop. Med. Hyg. 51, 214–218 (1994).
Gluzman, I. Y., Schlesinger, P. H. & Krogstad, D. J. Inoculum effect with chloroquine and Plasmodium falciparum. Antimicrob. Agents Chemother. 31, 32–36 (1987).
Berenbaum, M. C. A method for testing for synergy with any number of agents. J. Infect. Dis. 137, 122–130 (1978).
Ohrt, C., Willingmyre, G. D., Lee, P., Knirsch, C. & Milhous, W. Assessment of azithromycin in combination with other antimalarial drugs against Plasmodium falciparum in vitro. Antimicrob. Agents Chemother. 46, 2518–2524 (2002).
Canfield, C. J., Pudney, M. & Gutteridge, W. E. Interactions of atovaquone with other antimalarial drugs against Plasmodium falciparum in vitro. Exp. Parasitol. 80, 373–381 (1995). A good example of methodologies used in the identification of a new successful drug combination.
White, N. J. Delaying antimalarial drug resistance with combination chemotherapy. Parassitologia 41, 301–308 (1999).
Ponnudurai, T., Meuwissen, J. H., Leeuwenberg, A. D., Verhave, J. P. & Lensen, A. H. The production of mature gametocytes of Plasmodium falciparum in continuous cultures of different isolates infective to mosquitoes. Trans. R. Soc. Trop. Med. Hyg. 76, 242–250 (1982).
Templeton, T. J., Kaslow, D. C. & Fidock, D. A. Developmental arrest of the human malaria parasite Plasmodium falciparum within the mosquito midgut via CTRP gene disruption. Mol. Microbiol. 36, 1–9 (2000).
Moreno, A., Badell, E., Van Rooijen, N. & Druilhe, P. Human malaria in immunocompromised mice: new in vivo model for chemotherapy studies. Antimicrob. Agents Chemother. 45, 1847–1853 (2001).
Childs, G. E. et al. Comparison of in vitro and in vivo antimalarial activities of 9-phenanthrenecarbinols. Ann. Trop. Med. Parasitol. 78, 13–20 (1984).
Peters, W. et al. The chemotherapy of rodent malaria. XXVII. Studies on mefloquine (WR 142,490). Ann. Trop. Med. Parasitol. 71, 407–418 (1977).
Peters, W., Robinson, B. L. & Ellis, D. S. The chemotherapy of rodent malaria. XLII. Halofantrine and halofantrine resistance. Ann. Trop. Med. Parasitol. 81, 639–646 (1987).
Vennerstrom, J. L. et al. Synthesis and antimalarial activity of sixteen dispiro-1,2,4, 5-tetraoxanes: alkyl-substituted 7,8,15,16-tetraoxadispiro[5.2.5.2]hexadecanes. J. Med. Chem. 43, 2753–2758 (2000).
Posner, G. H. et al. Orally active, antimalarial, anticancer, artemisinin-derived trioxane dimers with high stability and efficacy. J. Med. Chem. 46, 1060–1065 (2003).
Peters, W. & Robinson, B. L. The chemotherapy of rodent malaria. LVI. Studies on the development of resistance to natural and synthetic endoperoxides. Ann. Trop. Med. Parasitol. 93, 325–329 (1999).
Sanni, L. A., Fonseca, L. F. & Langhorne, J. Mouse models for erythrocytic-stage malaria. Methods Mol. Med. 72, 57–76 (2002).
Wengelnik, K. et al. A class of potent antimalarials and their specific accumulation in infected erythrocytes. Science 295, 1311–1314 (2002).
Singh, A. et al. Critical role of amino acid 23 in mediating activity and specificity of vinckepain-2, a papain-family cysteine protease of rodent malaria parasites. Biochem. J. 368, 273–281 (2002).
Gysin, J. in Malaria: Parasite Biology, Pathogenesis and Protection. (ed. Sherman, I.) 419–441 (ASM, Washington DC, 1998).
Alanine, A., Nettekoven, M., Roberts, E. & Thomas, A. W. Lead generation — enhancing the success of drug discovery by investing in the hit to lead process. Comb. Chem. High Throughput Screen. 6, 51–66 (2003).
Bleicher, K. H., Bohm, H. J., Muller, K. & Alanine, A. I. Hit and lead generation: beyond high-throughput screening. Nature Rev. Drug Discov. 2, 369–378 (2003).
Di, L. & Kerns, E. H. Profiling drug-like properties in discovery research. Curr. Opin. Chem. Biol. 7, 402–408 (2003).
Kuo, M. R. et al. Targeting tuberculosis and malaria through inhibition of enoyl reductase: compound activity and structural data. J. Biol. Chem. 278, 20851–20859 (2003).
Yuvaniyama, J. et al. Insights into antifolate resistance from malarial DHFR-TS structures. Nature Struct. Biol. 10, 357–365 (2003).
Lipinski, C. A., Lombardo, F., Dominy, B. W. & Feeney, P. J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 46, 3–26 (2001).
Venkatesh, S. & Lipper, R. A. Role of the development scientist in compound lead selection and optimization. J. Pharm. Sci. 89, 145–154 (2000).
Molzon, J. The common technical document: the changing face of the New Drug Application. Nature Rev. Drug Discov. 2, 71–74 (2003).
Nzila, A. M. et al. Molecular evidence of greater selective pressure for drug resistance exerted by the long-acting antifolate pyrimethamine/sulfadoxine compared with the shorter-acting chlorproguanil/dapsone on Kenyan Plasmodium falciparum. J. Infect. Dis. 181, 2023–2028 (2000).
Mutabingwa, T. et al. Chlorproguanil-dapsone for treatment of drug-resistant falciparum malaria in Tanzania. Lancet 358, 1218–1223 (2001).
Jiang, L., Lee, P. C., White, J. & Rathod, P. K. Potent and selective activity of a combination of thymidine and 1843U89, a folate-based thymidylate synthase inhibitor, against Plasmodium falciparum. Antimicrob. Agents Chemother. 44, 1047–1050 (2000).
Razakantoanina, V., Nguyen Kim, P. P. & Jaureguiberry, G. Antimalarial activity of new gossypol derivatives. Parasitol. Res. 86, 665–668 (2000).
Bracchi-Ricard, V. et al. Characterization of an eukaryotic peptide deformylase from Plasmodium falciparum. Arch. Biochem. Biophys. 396, 162–170 (2001).
Banumathy, G., Singh, V., Pavithra, S. R. & Tatu, U. Heat shock protein 90 function is essential for Plasmodium falciparum growth in human erythrocytes. J. Biol. Chem. 278, 18336–18345 (2003).
Davioud-Charvet, E. et al. A prodrug form of a Plasmodium falciparum glutathione reductase inhibitor conjugated with a 4-anilinoquinoline. J. Med. Chem. 44, 4268–4276 (2001).
Woodard, C. L. et al. Oxindole-based compounds are selective inhibitors of Plasmodium falciparum cyclin dependent protein kinases. J. Med. Chem. 46, 3877–3882 (2003).
Eckstein-Ludwig, U. et al. Artemisinins target the SERCA of Plasmodium falciparum. Nature 424, 957–961 (2003).
Gero, A. M. et al. New malaria chemotherapy developed by utilization of a unique parasite transport system. Curr. Pharm. Des. 9, 867–877 (2003).
Joet, T., Eckstein-Ludwig, U., Morin, C. & Krishna, S. Validation of the hexose transporter of Plasmodium falciparum as a novel drug target. Proc. Natl Acad. Sci. USA 100, 7476–7479 (2003).
De, D., Krogstad, F. M., Byers, L. D. & Krogstad, D. J. Structure–activity relationships for antiplasmodial activity among 7-substituted 4-aminoquinolines. J. Med. Chem. 41, 4918–4926 (1998).
Stocks, P. A. et al. Novel short chain chloroquine analogues retain activity against chloroquine resistant K1 Plasmodium falciparum. J. Med. Chem. 45, 4975–4983 (2002).
Francis, S. E. et al. Molecular characterization and inhibition of a Plasmodium falciparum aspartic hemoglobinase. EMBO J. 13, 306–317 (1994).
Haque, T. S. et al. Potent, low-molecular-weight non-peptide inhibitors of malarial aspartyl protease plasmepsin II. J. Med. Chem. 42, 1428–1440 (1999).
Rosenthal, P. J. in Antimalarial Chemotherapy: Mechanisms of Action, Resistance, and New Directions in Drug Discovery (ed. Rosenthal, P. J.) 325–345 (Humana, Totawa, New Jersey, 2001).
Shenai, B. R. et al. Structure–activity relationships for inhibition of cysteine protease activity and development of Plasmodium falciparum by peptidyl vinyl sulfones. Antimicrob. Agents Chemother. 47, 154–160 (2003).
Vennerstrom, J. L. et al. Synthesis and antimalarial activity of sixteen dispiro-1,2,4, 5- tetraoxanes: alkyl-substituted 7,8,15,16-tetraoxadispiro[5.2.5.2]hexadecanes. J. Med. Chem. 43, 2753–2758 (2000).
Borstnik, K., Paik, I. H. & Posner, G. H. Malaria: new chemotherapeutic peroxide drugs. Mini Rev. Med. Chem. 2, 573–583 (2002).
Vaidya, A. B. in Antimalarial Chemotherapy: Mechanisms of Action, Resistance, and New Directions in Drug Discovery (ed. Rosenthal, P. J.) 203–218 (Humana, Totowa, New Jersey, 2001).
Clough, B. & Wilson, R. J. M. in Antimalarial Chemotherapy: Mechanisms of Action, Resistance, and New Directions in Drug Discovery (ed. Rosenthal, P. J.) 265–286 (Humana, Totawa, New Jersey, 2001).
McLeod, R. et al. Triclosan inhibits the growth of Plasmodium falciparum and Toxoplasma gondii by inhibition of apicomplexan Fab I. Int. J. Parasitol. 31, 109–113 (2001).
Ohkanda, J. et al. Peptidomimetic inhibitors of protein farnesyltransferase show potent antimalarial activity. Bioorg. Med. Chem. Lett. 11, 761–764 (2001).
Blackman, M. J. Proteases involved in erythrocyte invasion by the malaria parasite: function and potential as chemotherapeutic targets. Curr. Drug Targets 1, 59–83 (2000).
Gupta, S., Thapar, M. M., Mariga, S. T., Wernsdorfer, W. H. & Bjorkman, A. Plasmodium falciparum: in vitro interactions of artemisinin with amodiaquine, pyronaridine, and chloroquine. Exp. Parasitol. 100, 28–35 (2002).
Chawira, A. N., Warhurst, D. C., Robinson, B. L. & Peters, W. The effect of combinations of qinghaosu (artemisinin) with standard antimalarial drugs in the suppressive treatment of malaria in mice. Trans. R. Soc. Trop. Med. Hyg. 81, 554–558 (1987).
Peters, W. & Robinson, B. L. The chemotherapy of rodent malaria. LV. Interactions between pyronaridine and artemisinin. Ann. Trop. Med. Parasitol. 91, 141–145 (1997).
Acknowledgements
The authors wish to thank the MMV staff for organizing the antimalarial drug screening meeting that prompted the writing of this article. The contribution of all attendees at that meeting (F. Buckner, D. Gargallo, W. Milhous, H. Matile, M. Bendig, Kevin Bauer, S. Kamchonwongpaisan, M.-A. Mouries, L. Riopel and C. Craft) who were not authors on this review is appreciated. J. Sacchettini and M. Kuo are gratefully acknowledged for contributing Figure 5.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- APICOMPLEXA
-
Lower eukaryotic, obligate intracellular parasites characterized by the presence of apical organelles involved in host cell invasion. Includes parasitic protozoa responsible for malaria (Plasmodium), toxoplasmosis (Toxoplasma), babesiosis (Babesia) and coccidiosis (Eimeria).
- GAMETOCYTES
-
The sexual haploid stages produced in the blood that are infectious for the mosquito vector. Once inside the mosquito midgut, gametocytes transform into male or female gametes that can undergo fusion, genetic recombination and meiosis.
- SPOROZOITES
-
The haploid parasite forms that reside in the mosquito salivary glands and that are infectious for the human host, where they rapidly invade hepatocytes and transform into liver stage parasites.
- EFFICACY
-
A quantitative index of drug action, in this case related to suppression of malarial infection either in vitro or in vivo.
- ANTIFOLATES
-
Drugs that target the folate biosynthesis pathway. Antimalarial antifolate drugs target dihydrofolate reductase or dihydropteroate synthase.
- INTERMITTENT PREVENTIVE TREATMENT
-
Entails the administration of full therapeutic doses of a drug at defined intervals. Envisaged to confer a degree of sustained prophylactic protection in the most vulnerable populations, that is, young children and pregnant women.
- CHEMOPROPHYLAXIS
-
Drug treatment designed to prevent future occurrences of disease.
- POTENCY
-
An expression of the activity of a compound, in terms of the concentration required to produce a desired effect (for example, 50% inhibition of parasite growth).
- ENDEMICITY
-
An expression of the seasonality and degree of transmission in a malaria-afflicted region.
- PARASITAEMIA
-
A quantitative measure of the percentage of erythrocytes that are parasitized.
- PHARMACOKINETICS
-
The study of absorption, distribution, metabolism and elimination of drugs in a higher organism.
- RECRUDESCENCE
-
The reappearance of parasites or symptoms, in the case of parasitological or clinical recrudescence, in the days following drug treatment. This is a result of incomplete clearance of the infection.
- CYSTEINE PROTEASES
-
A class of parasite enzymes involved in crucial processes during parasite development.
- PROTEIN FARNESYL TRANSFERASES
-
These mediate attachment of the prenyl groups farnesyl and geranylgeranyl to specific eukaryotic cell proteins.
- HAEMOZOIN
-
An ordered crystalline assembly of β-haematin moieties produced following haemoglobin digestion in the parasite's food vacuole.
- FOOD VACUOLE
-
An acidic organelle of erythrocytic parasites in which haemoglobin degradation takes place.
- ORTHOLOGUE
-
A gene (or protein) with similar function to a gene (or protein) in a related species.
- HAEMATOCRIT
-
The proportion of the volume of a sample of blood that is represented by red blood cells.
- IC50
-
The drug concentration that produces a 50% inhibition of P. falciparum growth in vitro. This is frequently determined by calculating the concentration that produces a 50% reduction in uptake of 3H-hypoxanthine.
- GIEMSA
-
A nucleic acid stain used to visually distinguish Plasmodium parasites from the surrounding cells.
- SYNCHRONIZED CULTURES
-
Cultures of parasites at the same or similar stage of intracellular development.
- INOCULUM
-
A substance or organism that is introduced into surroundings suited to cell growth. In this case this refers to introduction of a defined number of parasitized erythrocytes into in vitro culture or a susceptible rodent.
- ISOBOLOGRAM
-
A graphical representation of growth inhibition data for two compounds, plotted as fractional IC50 values on an X–Y axis, permitting determination of whether the two compounds are synergistic, additive or antagonistic.
- ANTAGONISM
-
An interaction between agents in which one partially or completely inhibits the effect of the other.
- SCHIZOGONY
-
The intracellular process whereby multinucleated Plasmodium parasites differentiate to form multiple infectious forms (merozoites), which then egress from the infected cell in order to invade uninfected erythrocytes.
- ED50
-
The drug concentration that produces a 50% reduction in parasitaemia in vivo, typically in a rodent malaria model.
- BIOAVAILABILITY
-
The degree to which a drug is available to the body. This is influenced by how much the substance is absorbed and circulated.
- SEQUESTRATION
-
The ability of P. falciparum to sequester in capillary beds as a result of binding of the parasitized erythrocyte to endothelial cell surface receptors. This binding is mediated by parasite-encoded variant antigens presented at the erythrocyte surface.
- LEAD OPTIMIZATION
-
The process of chemically optimizing a compound to improve antimicrobial activity and pharmacological properties.
- LEAD IDENTIFICATION
-
The process of identifying an acceptable lead compound with potent in vitro and in vivo activity.
- PHARMACODYNAMICS
-
The study of the mechanisms of actions of a drug, and the relationship between how much drug is in the body and its effects.
Rights and permissions
About this article
Cite this article
Fidock, D., Rosenthal, P., Croft, S. et al. Antimalarial drug discovery: efficacy models for compound screening. Nat Rev Drug Discov 3, 509–520 (2004). https://doi.org/10.1038/nrd1416
Issue Date:
DOI: https://doi.org/10.1038/nrd1416
This article is cited by
-
Solubility enhancing lipid-based vehicles for artemether and lumefantrine destined for the possible treatment of induced malaria and inflammation: in vitro and in vivo evaluations
Beni-Suef University Journal of Basic and Applied Sciences (2024)
-
Synergistic antimalarial treatment of Plasmodium berghei infection in mice with dihydroartemisinin and Gymnema inodorum leaf extract
BMC Complementary Medicine and Therapies (2023)
-
Development of a human malaria-on-a-chip disease model for drug efficacy and off-target toxicity evaluation
Scientific Reports (2023)
-
Cuscuta reflexa Possess Potent Inhibitory Activity Against Human Malaria Parasite: An In Vitro and In Vivo Study
Current Microbiology (2023)
-
Evaluation of anti-malarial potency of new pyrazole-hydrazine coupled to Schiff base derivatives
Malaria Journal (2022)