Abstract
Incretin-based drugs, such as glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase 4 inhibitors, are now routinely used to treat type 2 diabetes mellitus. These agents regulate glucose metabolism through multiple mechanisms, their use is associated with low rates of hypoglycemia, and they either do not affect body weight (dipeptidyl peptidase 4 inhibitors), or promote weight loss (glucagon-like peptide-1 receptor agonists). The success of exenatide and sitagliptin, the first therapies in their respective drug classes to be based on incretins, has fostered the development of multiple new agents that are currently in late stages of clinical development or awaiting approval. This Review highlights our current understanding of the mechanisms of action of incretin-based drugs, with an emphasis on the emerging clinical profile of new agents.
Key Points
-
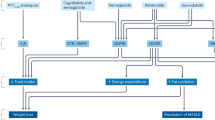
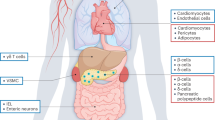
Incretins exert antidiabetic actions in a glucose-dependent manner
-
Glucagon-like peptide 1 receptor (GLP-1R) agonists, but not dipeptidyl peptidase-4 (DPP-4) inhibitors, inhibit gastric emptying and might cause weight loss
-
DPP-4 inhibitors can be administered orally and are well tolerated
-
GLP-1R agonists must be administered by subcutaneous injection and commonly cause nausea
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Kreymann, B., Williams, G., Ghatei, M. A. & Bloom, S. R. Glucagon-like peptide-17–36: a physiological incretin in man. Lancet 2, 1300–1304 (1987).
Deacon, C. F. Therapeutic strategies based on glucagon-like peptide 1. Diabetes 53, 2181–2189 (2004).
Drucker, D. J. The biology of incretin hormones. Cell. Metab. 3, 153–165 (2006).
Drucker, D. J. & Nauck, M. A. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet 368, 1696–1705 (2006).
Deacon, C. F., Johnsen, A. H. & Holst, J. J. Degradation of glucagon-like peptide-1 by human plasma in vitro yields an N-terminally truncated peptide that is a major endogenous metabolite in vivo . J. Clin. Endocrinol. Metab. 80, 952–957 (1995).
Kieffer, T. J., McIntosh, C. H. & Pederson, R. A. Degradation of glucose-dependent insulinotropic polypeptide and truncated glucagon-like peptide 1 in vitro and in vivo by dipeptidyl peptidase IV. Endocrinology 136, 3585–3596 (1995).
Cervera, A. et al. Mechanism of action of exenatide to reduce postprandial hyperglycemia in type 2 diabetes. Am. J. Physiol. Endocrinol. Metab. 294, E846–E852 (2008).
Nauck, M. A. et al. Preserved incretin activity of glucagon-like peptide 17–36 amide but not of synthetic human gastric inhibitory polypeptide in patients with type-2 diabetes mellitus. J. Clin. Invest. 91, 301–307 (1993).
Højberg, P. V. et al. Four weeks of near-normalisation of blood glucose improves the insulin response to glucagon-like peptide-1 and glucose-dependent insulinotropic polypeptide in patients with type 2 diabetes. Diabetologia 52, 199–207 (2008).
Zander, M., Madsbad, S., Madsen, J. L. & Holst, J. J. Effect of 6-week course of glucagon-like peptide 1 on glycaemic control, insulin sensitivity, and β-cell function in type 2 diabetes: a parallel-group study. Lancet 359, 824–830 (2002).
Buse, J. B. et al. Effects of exenatide (exendin-4) on glycemic control over 30 weeks in sulfonylurea-treated patients with type 2 diabetes. Diabetes Care 27, 2628–2635 (2004).
DeFronzo, R. A. et al. Effects of exenatide (exendin-4) on glycemic control and weight over 30 weeks in metformin-treated patients with type 2 diabetes. Diabetes Care 28, 1092–1100 (2005).
Kendall, D. M. et al. Effects of exenatide (exendin-4) on glycemic control over 30 weeks in patients with type 2 diabetes treated with metformin and a sulfonylurea. Diabetes Care 28, 1083–1091 (2005).
Zinman, B. et al. The effect of adding exenatide to a thiazolidinedione in suboptimally controlled type 2 diabetes: a randomized trial. Ann. Intern. Med. 146, 477–485 (2007).
Heine, R. J. et al. Exenatide versus insulin glargine in patients with suboptimally controlled type 2 diabetes: a randomized trial. Ann. Intern. Med. 143, 559–569 (2005).
Nauck, M. A. et al. A comparison of twice-daily exenatide and biphasic insulin aspart in patients with type 2 diabetes who were suboptimally controlled with sulfonylurea and metformin: a non-inferiority study. Diabetologia 50, 259–267 (2007).
Bunck, M. C. et al. β-cell function and glycemic control following one year exenatide therapy, and after 12 week wash out, in patients with type 2 diabetes. Presented at the 68th Annual Meeting of the American Diabetes Association, 2008 June 6–10, San Francisco, US (2008).
Vilsbøll, T. et al. Liraglutide, a long-acting human GLP-1 analog, given as monotherapy significantly improves glycemic control and lowers body weight without risk of hypoglycemia in patients with type 2 diabetes mellitus. Diabetes Care 6, 160–610 (2007).
Seino, Y., Rasmussen, M. F., Zdravkovic, M. & Kaku, K. Dose-dependent improvement in glycemia with once-daily liraglutide without hypoglycemia or weight gain: a double-blind, randomized, controlled trial in Japanese patients with type 2 diabetes. Diabetes Res. Clin. Pract. 81, 161–168 (2008).
Zinman, B. et al. Efficacy and safety of the human GLP-1 analog liraglutide in combination with metformin and TZD in patients with type 2 diabetes mellitus (LEAD-4 Met + TZD). Presented at the 44th Annual Meeting of the European Association for the Study of Diabetes, 2008 September 7–11, Rome, Italy (2008).
Garber, A. et al. Liraglutide versus glimepiride monotherapy for type 2 diabetes (LEAD-3 Mono): a randomised, 52-week, phase III, double-blind, parallel-treatment trial. Lancet 373, 473–481 (2009).
Marre, M. et al. Liraglutide, a once-daily human GLP-1 analogue, added to a sulphonylurea over 26 weeks produces greater improvements in glycaemic and weight control compared with adding rosiglitazone or placebo in subjects with type 2 diabetes (LEAD-1 SU). Diabetic Medicine 14 Jan doi:10.1111/j.1464–54912009.02666.x (2009).
Nauck, M. et al. Efficacy and safety comparison of liraglutide, glimepiride, and placebo, all in combination with metformin in type 2 diabetes mellitus (LEAD-2 Met). Diabetes Care 32, 84–90 (2008).
Russell-Jones, D. et al. Significantly better glycemic control and weight reduction with liraglutide, a once-daily human GLP-1 analog, compared with insulin glargine: all as add-on to metformin and a sulfonylurea in type 2 diabetes. Presented at the 68th Annual meeting of the American Diabetes Association, 6–10 June 2008, San Francisco, USA (2008).
Blonde, L. et al. Liragluide: superiror glycemia control vs exenatide when added to metformin and/or SU in type 2 diabetes. Presented at the Annual meeting of the Canadian Diabetes Association, 16 October 2008, Montreal, Quebec, Canada (2008).
Ratner, R. E. et al. A dose-finding study of the new GLP-1 agonist AVE0010 in type 2 diabetes insufficiently controlled with metformin. Presented at the 68th Annual meeting of the American Diabetes Association, 2008 June 6–10, San Francisco, USA (2008).
Kim, D, et al. Effects of once-weekly dosing of a long-acting release formulation of exenatide on glucose control and body weight in subjects with type 2 diabetes. Diabetes Care 30, 1487–1493 (2007).
Drucker, D. J. et al. Exenatide once weekly versus twice daily for the treatment of type 2 diabetes: a randomised, open-label, non-inferiority study. Lancet 372, 1240–1250 (2008).
Baggio, L. L. et al. A recombinant human glucagon-like peptide (GLP)-1-albumin protein (albugon) mimics peptidergic activation of GLP-1 receptor-dependent pathways coupled with satiety, gastrointestinal motility, and glucose homeostasis. Diabetes 53, 2492–2500 (2004).
Bloom, M. et al. Albugon fusion protein; a long-acting analog of GLP-1 that provides lasting anti-diabetic effect in animals. Presented at the 63rd Annual Meeting of the American Diabetes Association, 2003 June, New Orleans, LO, USA (2003).
Matthews, J. E. et al. Pharmacodynamics, pharmacokinetics, safety and tolerability of albiglutide, a long-acting GLP-1 mimetic, in patients with type 2 diabetes. J. Clin. Endocrinol. Metab. 93, 4810–4817 (2008).
Balena, R. et al. Eight weeks of treatment with the long acting, human GLP-1 analogue R1583 improves glycemic control and lowers body weight in subjects with type 2 diabetes mellitus (T2DM) treated with metformin: a double-blind placebo-controlled phase 2 Study. Presented at the 68th Annual Meeting of the American Diabetes Association, 2008 June 6–10, San Francisco, US (2008).
Ratner, R. et al. safety and tolerability of high doses of the long acting, human GLP-1 analogue R1583 in diabetic subjects treated with metformin: a double-blind, placebo-controlled phase 2 study. Presented at the 68th Annual Meeting of the American Diabetes Association, 2008 June 6–10, San Francisco, USA (2008).
Baggio, L. L. et al. The long-acting albumin-exendin-4 GLP-1R agonist CJC-1134 engages central and peripheral mechanisms regulating glucose homeostasis. Gastroenterology 134, 1137–1147 (2008).
Ahmad, S. R. & Swann, J. Exenatide and rare adverse events. N. Engl. J. Med. 358, 1970–1972 (2008).
Raufman, J.-P. et al. Truncated glucagon-like peptide-1 interacts with exendin receptors on disperced acini from guinea pig pancreas. Identification of a mammalian homologue of the reptilian peptide exendin-4. J. Biol. Chem. 267, 21432–21437 (1992).
Hansotia, T. et al. Double incretin receptor knockout (DIRKO) mice reveal an essential role for the enteroinsular axis in transducing the glucoregulatory actions of DPP-IV inhibitors. Diabetes 53, 1326–1335 (2004).
Aschner, P. et al. Effect of the dipeptidyl peptidase-4 inhibitor sitagliptin as monotherapy on glycemic control in patients with type 2 diabetes. Diabetes Care 29, 2632–2637 (2006).
Charbonnel, B. et al. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor sitagliptin added to ongoing metformin therapy in patients with type 2 diabetes inadequately controlled with metformin alone. Diabetes Care 29, 2638–2643 (2006).
Rosenstock, J. et al. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor sitagliptin added to ongoing pioglitazone therapy in patients with type 2 diabetes: a 24-week, multicenter, randomized, double-blind, placebo-controlled, parallel-group study. Clin. Ther. 28, 1556–1568 (2006).
Goldstein, B. J. et al. Effect of initial combination therapy with sitagliptin, a dipeptidyl peptidase-4 inhibitor, and metformin on glycemic control in patients with type 2 diabetes. Diabetes Care 30, 1979–1987 (2007).
Scott, R. et al. Efficacy and safety of sitagliptin when added to ongoing metformin therapy in patients with type 2 diabetes. Diabetes Obes. Metab. 10, 959–969 (2008).
Raz, I. et al. Efficacy and safety of sitagliptin added to ongoing metformin therapy in patients with type 2 diabetes. Curr. Med. Res. Opin. 24, 537–550 (2008).
Pi-Sunyer, F. X. et al. Efficacy and tolerability of vildagliptin monotherapy in drug-naive patients with type 2 diabetes. Diabetes Res. Clin. Pract. 76, 132–138 (2007).
Rosenstock, J. et al. Comparison of vildagliptin and rosiglitazone monotherapy in patients with type 2 diabetes: a 24-week, double-blind, randomized trial. Diabetes Care 30, 217–223 (2007).
Bolli, G. et al. Efficacy and tolerability of vildagliptin vs. pioglitazone when added to metformin: a 24-week, randomized, double-blind study. Diabetes Obes. Metab. 10, 82–90 (2008).
Lankas, G. R. et al. Dipeptidyl peptidase IV inhibition for the treatment of type 2 diabetes: potential importance of selectivity over dipeptidyl peptidases 8 and 9. Diabetes 54, 2988–2994 (2005).
Chan, J. C. et al. Safety and efficacy of sitagliptin in patients with type 2 diabetes and chronic renal insufficiency. Diabetes Obes. Metab. 10, 545–555 (2008).
Defronzo, R. A. et al. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor alogliptin in patients with type 2 diabetes mellitus and inadequate glycemic control: a randomized, double-blind, placebo-controlled study. Diabetes Care 31, 2315–2317 (2008).
Nauck, M. A. et al. Efficacy and safety of adding the dipeptidyl peptidase-4 inhibitor alogliptin to metformin therapy in patients with type 2 diabetes inadequately controlled with metformin monotherapy: a multicentre, randomised, double-blind, placebo-controlled study. Int. J. Clin. Pract. 63, 46–55 (2009).
Fonseca, V. et al. Addition of vildagliptin to insulin improves glycaemic control in type 2 diabetes. Diabetologia 50, 1148–1155 (2007).
Garcia-Soria, G. et al. The dipeptidyl peptidase-4 inhibitor PHX1149 improves blood glucose control in patients with type 2 diabetes mellitus. Diabetes Obes. Metab. 10, 293–300 (2008).
Ghofaili, K. A. et al. Effect of exenatide on β cell function after islet transplantation in type 1 diabetes. Transplantation 83, 24–28 (2007).
Froud, T. et al. The use of exenatide in islet transplant recipients with chronic allograft dysfunction: safety, efficacy, and metabolic effects. Transplantation 86, 36–45 (2008).
Inzucchi, S. E. & McGuire, D. K. New drugs for the treatment of diabetes: part II: incretin-based therapy and beyond. Circulation 117, 574–584 (2008).
Sokos, G. G. et al. Glucagon-like peptide-1 infusion improves left ventricular ejection fraction and functional status in patients with chronic heart failure. J. Card. Fail. 12, 694–699 (2006).
Nikolaidis, L. A. et al. Effects of glucagon-like peptide-1 in patients with acute myocardial infarction and left ventricular dysfunction after successful reperfusion. Circulation 109, 962–965 (2004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
D. Drucker declared associations with the following companies: Amylin Pharmaceuticals, Arena Pharmaceuticals Inc., Arisaph Pharmaceuticals Inc, Conjuchem Inc., Eli Lilly Inc., Emisphere Technologies Inc., GlaxoSmithKline, Glenmark Pharmaceuticals, Hoffman LaRoche Inc., Isis Pharmaceuticals Inc., Mannkind Inc., Merck Research Laboratories, Metabolex Inc., Novartis Pharmaceuticals, Novo Nordisk Inc., Phenomix Inc., Takeda, Transition Pharmaceuticals (advisor/consultant); Arena Pharmaceuticals Inc, Merck Research Laboratories, Metabolex Inc. and Novo Nordisk Inc (advisor/consultant and research/grant support). J. Lovshin declared no competing interests.
Rights and permissions
About this article
Cite this article
Lovshin, J., Drucker, D. Incretin-based therapies for type 2 diabetes mellitus. Nat Rev Endocrinol 5, 262–269 (2009). https://doi.org/10.1038/nrendo.2009.48
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2009.48
This article is cited by
-
Novel Insight into the Effect of Probiotics in the Regulation of the Most Important Pathways Involved in the Pathogenesis of Type 2 Diabetes Mellitus
Probiotics and Antimicrobial Proteins (2023)
-
DPP-4 inhibitors and GLP-1RAs: cardiovascular safety and benefits
Military Medical Research (2022)
-
A comparative study of the cardioprotective effect of Metformin, Sitagliptin and Dapagliflozin on Isoprenaline induced myocardial infarction in non-diabetic rats
Bulletin of the National Research Centre (2022)
-
Drugs and Endogenous Factors as Protagonists in Neurogenic Stimulation
Stem Cell Reviews and Reports (2022)
-
Semaglutide and Diabetic Retinopathy Risk in Patients with Type 2 Diabetes Mellitus: A Meta-Analysis of Randomized Controlled Trials
Clinical Drug Investigation (2022)