Abstract
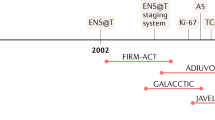
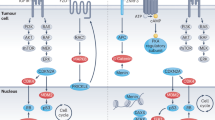
Adrenocortical carcinoma is a rare heterogeneous neoplasm with an incompletely understood pathogenesis and a poor prognosis. Previous studies have identified overexpression of insulin-like growth factor 2 (IGF-2) and constitutive activation of β-catenin as key factors involved in the development of adrenocortical carcinoma. Most patients present with steroid hormone excess, for example Cushing syndrome or virilization, or abdominal mass effects, but a growing proportion of patients with adrenocortical carcinoma (currently >15%) is initially diagnosed incidentally. No general consensus on the diagnostic and therapeutic measures for adrenocortical carcinoma exists, but collaborative efforts, such as international conferences and networks, including the European Network for the Study of Adrenal Tumors (ENSAT), have substantially advanced the field. In patients with suspected adrenocortical carcinoma, a thorough endocrine and imaging work-up is recommended to guide the surgical approach aimed at complete resection of the tumor. To establish an adequate basis for treatment decisions, pathology reports include the Weiss score to assess malignancy, the resection status and the Ki67 index. As recurrence is frequent, close follow-up initially every 3 months is mandatory. Most patients benefit from adjuvant mitotane treatment. In metastatic disease, mitotane is the cornerstone of initial treatment, and cytotoxic drugs should be added in case of progression. Results of a large phase III trial in advanced adrenocortical carcinoma are anticipated for 2011 and will hopefully establish a benchmark therapy. New targeted therapies, for example, IGF-1 receptor inhibitors, are under investigation and may soon improve current treatment options.
Key Points
-
Overexpression of insulin-like growth factor 2 and constitutive activation of β-catenin are key molecular alterations in adrenocortical carcinoma
-
Detailed presurgical endocrine and imaging work-up and an expert surgeon are key prerequisites for a complete resection that offers the best chance of cure
-
Even after radical resection, recurrence rate is high and, therefore, most patients benefit from adjuvant treatment strategies (mitotane with or without radiotherapy)
-
Mitotane is the most effective single drug for adrenocortical carcinoma, but drug monitoring is important and management of adverse events is demanding
-
In advanced disease not amenable to surgery, mitotane is given as monotherapy or in combination with cytotoxic chemotherapy (either etoposide combined with doxorubicin and cisplatin or streptozotocin)
-
To facilitate progress in the treatment of adrenocortical carcinoma, patients should be enrolled in clinical trials
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Abecassis, M., McLoughlin, M. J., Langer, B. & Kudlow, J. E. Serendipitous adrenal masses: prevalence, significance, and management. Am. J. Surg. 149, 783–788 (1985).
Grumbach, M. M. et al. Management of the clinically inapparent adrenal mass (“incidentaloma”). Ann. Intern. Med. 138, 424–429 (2003).
Bovio, S. et al. Prevalence of adrenal incidentaloma in a contemporary computerized tomography series. J. Endocrinol. Invest. 29, 298–302 (2006).
Song, J. H., Chaudhry, F. S. & Mayo-Smith, W. W. The incidental adrenal mass on CT: prevalence of adrenal disease in 1,049 consecutive adrenal masses in patients with no known malignancy. AJR Am. J. Roentgenol. 190, 1163–1168 (2008).
Cutler, S. J., Young, J. L. & Connelly, R. R. (Eds) Third national cancer survey: incidenca data (U. S. Dept of Health, Education, and Welfare, Public Health Service, National Institutes of Health, National Cancer Institute, Bethesda, 1975).
Kebebew, E., Reiff, E., Duh, Q. Y., Clark, O. H. & McMillan, A. Extent of disease at presentation and outcome for adrenocortical carcinoma: have we made progress? World J. Surg. 30, 872–878 (2006).
Golden, S. H., Robinson, K. A., Saldanha, I., Anton, B. & Ladenson, P. W. Clinical review: Prevalence and incidence of endocrine and metabolic disorders in the United States: a comprehensive review. J. Clin. Endocrinol. Metab. 94, 1853–1878 (2009).
Koschker, A. C., Fassnacht, M., Hahner, S., Weismann, D. & Allolio, B. Adrenocortical carcinoma—improving patient care by establishing new structures. Exp. Clin. Endocrinol. Diabetes 114, 45–51 (2006).
Ribeiro, R. C. et al. An inherited p53 mutation that contributes in a tissue-specific manner to pediatric adrenal cortical carcinoma. Proc. Natl Acad. Sci. USA 98, 9330–9335 (2001).
Pinto, E. M. et al. Founder effect for the highly prevalent R337H mutation of tumor suppressor p53 in Brazilian patients with adrenocortical tumors. Arq. Bras. Endocrinol. Metabol. 48, 647–650 (2004).
Hutter, A. M. Jr & Kayhoe, D. E. Adrenal cortical carcinoma. Clinical features of 138 patients. Am. J. Med. 41, 572–580 (1966).
Bilimoria, K. Y. et al. Adrenocortical carcinoma in the United States: treatment utilization and prognostic factors. Cancer 113, 3130–3136 (2008).
Wooten, M. D. & King, D. K. Adrenal cortical carcinoma. Epidemiology and treatment with mitotane and a review of the literature. Cancer 72, 3145–3155 (1993).
Universität Würzburg Offizielle Homepage des Deutsche Nebennierenkarzinom-Registers [online], www.nebennierenkarzinom.de/ (2010).
Sidhu, S. et al. Comparative genomic hybridization analysis of adrenocortical tumors. J. Clin. Endocrinol. Metab. 87, 3467–3474 (2002).
Kjellman, M. et al. Genotyping of adrenocortical tumors: very frequent deletions of the MEN1 locus in 11q13 and of a 1-centimorgan region in 2p16. J. Clin. Endocrinol. Metab. 84, 730–735 (1999).
Gicquel, C. et al. Molecular markers and long-term recurrences in a large cohort of patients with sporadic adrenocortical tumors. Cancer Res. 61, 6762–6767 (2001).
DeChiara, T. M., Robertson, E. J. & Efstratiadis, A. Parental imprinting of the mouse insulin-like growth factor II gene. Cell 64, 849–859 (1991).
Wiedemann, H. R. et al. The proteus syndrome. Partial gigantism of the hands and/or feet, nevi, hemihypertrophy, subcutaneous tumors, macrocephaly or other skull anomalies and possible accelerated growth and visceral affections. Eur. J. Pediatr. 140, 5–12 (1983).
Boulle, N., Logié, A., Gicquel, C., Perin, L. & Le Bouc, Y. Increased levels of insulin-like growth factor II (IGF-II) and IGF-binding protein-2 are associated with malignancy in sporadic adrenocortical tumors. J. Clin. Endocrinol. Metab. 83, 1713–1720 (1998).
Gicquel, C. et al. Structural and functional abnormalities at 11p15 are associated with the malignant phenotype in sporadic adrenocortical tumors: study on a series of 82 tumors. J. Clin. Endocrinol. Metab. 82, 2559–2565 (1997).
Logié, A. et al. Autocrine role of IGF-II in proliferation of human adrenocortical carcinoma NCI H295R cell line. J. Mol. Endocrinol. 23, 23–32 (1999).
Kikuchi, A. Tumor formation by genetic mutations in the components of the Wnt signaling pathway. Cancer Sci. 94, 225–229 (2003).
Naylor, E. W. & Gardner, E. J. Adrenal adenomas in a patient with Gardner's syndrome. Clin. Genet. 20, 67–73 (1981).
Tissier, F. et al. Mutations of beta-catenin in adrenocortical tumors: activation of the Wnt signaling pathway is a frequent event in both benign and malignant adrenocortical tumors. Cancer Res. 65, 7622–7627 (2005).
Tadjine, M., Lampron, A., Ouadi, L. & Bourdeau, I. Frequent mutations of beta-catenin gene in sporadic secreting adrenocortical adenomas. Clin. Endocrinol. (Oxf.) 68, 264–270 (2008).
Berthon, A. et al. Constitutive beta-catenin activation induces adrenal hyperplasia and promotes adrenal cancer development. Hum. Mol. Genet. 19, 1561–1576 (2010).
Luo, X., Ikeda, Y. & Parker, K. L. A cell-specific nuclear receptor is essential for adrenal and gonadal development and sexual differentiation. Cell 77, 481–490 (1994).
Hammer, G. D., Parker, K. L. & Schimmer, B. P. Minireview: transcriptional regulation of adrenocortical development. Endocrinology 146, 1018–1024 (2005).
Schimmer, B. P. & White, P. C. Minireview: steroidogenic factor 1: its roles in differentiation, development, and disease. Mol. Endocrinol. 24, 1322–1337 (2010).
Figueiredo, B. C. et al. Amplification of the steroidogenic factor 1 gene in childhood adrenocortical tumors. J. Clin. Endocrinol. Metab. 90, 615–619 (2005).
Pianovski, M. A. et al. Mortality rate of adrenocortical tumors in children under 15 years of age in Curitiba, Brazil. Pediatr. Blood Cancer 47, 56–60 (2006).
Almeida, M. Q. et al. Steroidogenic factor 1 overexpression and gene amplification are more frequent in adrenocortical tumors from children than from adults. J. Clin. Endocrinol. Metab. 95, 1458–1462 (2010).
Sbiera, S. et al. High diagnostic and prognostic value of steroidogenic factor-1 expression in adrenal tumors. J. Clin. Endocrinol. Metab. 95, E161–E171 (2010).
Doghman, M. et al. Increased steroidogenic factor-1 dosage triggers adrenocortical cell proliferation and cancer. Mol. Endocrinol. 21, 2968–2987 (2007).
Lichtenauer, U. D. et al. Pre-B-cell transcription factor 1 and steroidogenic factor 1 synergistically regulate adrenocortical growth and steroidogenesis. Endocrinology 148, 693–704 (2007).
Doghman, M. et al. Inhibition of adrenocortical carcinoma cell proliferation by steroidogenic factor-1 inverse agonists. J. Clin. Endocrinol. Metab. 94, 2178–2183 (2009).
Hotta, M. & Baird, A. Differential effects of transforming growth factor type beta on the growth and function of adrenocortical cells in vitro. Proc. Natl Acad. Sci. USA 83, 7795–7799 (1986).
Feige, J. J. et al. Transforming growth factor beta 1: an autocrine regulator of adrenocortical steroidogenesis. Endocr. Res. 17, 267–279 (1991).
Feige, J. J., Vilgrain, I., Brand, C., Bailly, S. & Souchelnitskiy, S. Fine tuning of adrenocortical functions by locally produced growth factors. J. Endocrinol. 158, 7–19 (1998).
de Fraipont, F. et al. Gene expression profiling of human adrenocortical tumors using complementary deoxyribonucleic Acid microarrays identifies several candidate genes as markers of malignancy. J. Clin. Endocrinol. Metab. 90, 1819–1829 (2005).
Bernini, G. P. et al. Angiogenesis in human normal and pathologic adrenal cortex. J. Clin. Endocrinol. Metab. 87, 4961–4965 (2002).
Kolomecki, K., Stepien, H., Bartos, M. & Kuzdak, K. Usefulness of VEGF, MMP-2, MMP-3 and TIMP-2 serum level evaluation in patients with adrenal tumours. Endocr. Regul. 35, 9–16 (2001).
Adam, P. et al. Epidermal growth factor receptor in adrenocortical tumors: analysis of gene sequence, protein expression and correlation with clinical outcome. Mod. Pathol. 23, 1596–1604 (2010).
Hisada, M., Garber, J. E., Fung, C. Y., Fraumeni, J. F. Jr & Li, F. P. Multiple primary cancers in families with Li-Fraumeni syndrome. J. Natl Cancer Inst. 90, 606–611 (1998).
Wagner, J. et al. High frequency of germline p53 mutations in childhood adrenocortical cancer. J. Natl Cancer Inst. 86, 1707–1710 (1994).
Ohgaki, H., Kleihues, P. & Heitz, P. U. p53 mutations in sporadic adrenocortical tumors. Int. J. Cancer 54, 408–410 (1993).
Reincke, M. et al. p53 mutations in human adrenocortical neoplasms: immunohistochemical and molecular studies. J. Clin. Endocrinol. Metab. 78, 790–794 (1994).
Libè, R. et al. Somatic TP53 mutations are relatively rare among adrenocortical cancers with the frequent 17p13 loss of heterozygosity. Clin. Cancer Res. 13, 844–850 (2007).
Soon, P. S., McDonald, K. L., Robinson, B. G. & Sidhu, S. B. Molecular markers and the pathogenesis of adrenocortical cancer. Oncologist 13, 548–561 (2008).
Beuschlein, F., Fassnacht, M., Klink, A., Allolio, B. & Reincke, M. ACTH-receptor expression, regulation and role in adrenocortial tumor formation. Eur. J. Endocrinol. 144, 199–206 (2001).
Giordano, T. J. et al. Distinct transcriptional profiles of adrenocortical tumors uncovered by DNA microarray analysis. Am. J. Pathol. 162, 521–531 (2003).
de Reyniès, A. et al. Gene expression profiling reveals a new classification of adrenocortical tumors and identifies molecular predictors of malignancy and survival. J. Clin. Oncol. 27, 1108–1115 (2009).
Giordano, T. J. et al. Molecular classification and prognostication of adrenocortical tumors by transcriptome profiling. Clin. Cancer Res. 15, 668–676 (2009).
Szabó, P. M. et al. Meta-analysis of adrenocortical tumour genomics data: novel pathogenic pathways revealed. Oncogene 29, 3163–3172 (2010).
Icard, P. et al. Adrenocortical carcinomas: surgical trends and results of a 253-patient series from the French Association of Endocrine Surgeons study group. World J. Surg. 25, 891–897 (2001).
Crucitti, F., Bellantone, R., Ferrante, A., Boscherini, M. & Crucitti, P. The Italian Registry for Adrenal Cortical Carcinoma: analysis of a multiinstitutional series of 129 patients. The ACC Italian Registry Study Group. Surgery 119, 161–170 (1996).
Kendrick, M. L. et al. Adrenocortical carcinoma: surgical progress or status quo? Arch. Surg. 136, 543–549 (2001).
Schulick, R. D. & Brennan, M. F. Adrenocortical carcinoma. World J. Urol. 17, 26–34 (1999).
Dackiw, A. P., Lee, J. E., Gagel, R. F. & Evans, D. B. Adrenal cortical carcinoma. World J. Surg. 25, 914–926 (2001).
Abiven, G. et al. Clinical and biological features in the prognosis of adrenocortical cancer: poor outcome of cortisol-secreting tumors in a series of 202 consecutive patients. J. Clin. Endocrinol. Metab. 91, 2650–2655 (2006).
Stewart, P. M., Walker, B. R., Holder, G., O'Halloran, D. & Shackleton, C. H. 11 beta-Hydroxysteroid dehydrogenase activity in Cushing's syndrome: explaining the mineralocorticoid excess state of the ectopic adrenocorticotropin syndrome. J. Clin. Endocrinol. Metab. 80, 3617–3620 (1995).
Seccia, T. M., Fassina, A., Nussdorfer, G. G., Pessina, A. C. & Rossi, G. P. Aldosterone-producing adrenocortical carcinoma: an unusual cause of Conn's syndrome with an ominous clinical course. Endocr. Relat. Cancer 12, 149–159 (2005).
Hyodo, T., Megyesi, K., Kahn, C. R., McLean, J. P. & Friesen, H. G. Adrenocortical carcinoma and hypoglycemia: evidence for production of nonsuppressible insulin-like activity by the tumor. J. Clin. Endocrinol. Metab. 44, 1175–1184 (1977).
Wajchenberg, B. et al. Adrenocortical carcinoma: clinical and laboratory observations. Cancer 88, 711–736 (2000).
Luton, J. P. et al. Clinical features of adrenocortical carcinoma, prognostic factors, and the effect of mitotane therapy. N. Engl. J. Med. 322, 1195–1201 (1990).
Allolio, B. & Fassnacht, M. Clinical review: Adrenocortical carcinoma: clinical update. J. Clin. Endocrinol. Metab. 91, 2027–2037 (2006).
Libè, R., Fratticci, A. & Bertherat, J. Adrenocortical cancer: pathophysiology and clinical management. Endocr. Relat. Cancer 14, 13–28 (2007).
European Network for the Study of Adrenal Tumours Adrenocortical carcinomas [online], http://www.ensat.org/acc.htm (2010).
Fassnacht, M. & Allolio, B. Clinical management of adrenocortical carcinoma. Best Pract. Res. Clin. Endocrinol. Metab. 23, 273–289 (2009).
Boland, G. W. et al. Characterization of adrenal masses using unenhanced CT: an analysis of the CT literature. AJR Am. J. Roentgenol. 171, 201–204 (1998).
Hamrahian, A. H. et al. Clinical utility of noncontrast computed tomography attenuation value (hounsfield units) to differentiate adrenal adenomas/hyperplasias from nonadenomas: Cleveland Clinic experience. J. Clin. Endocrinol. Metab. 90, 871–877 (2005).
Caoili, E. M. et al. Adrenal masses: characterization with combined unenhanced and delayed enhanced CT. Radiology 222, 629–633 (2002).
Ilias, I., Sahdev, A., Reznek, R. H., Grossman, A. B. & Pacak, K. The optimal imaging of adrenal tumours: a comparison of different methods. Endocr. Relat. Cancer 14, 587–599 (2007).
Heinz-Peer, G., Memarsadeghi, M. & Niederle, B. Imaging of adrenal masses. Curr. Opin. Urol. 17, 32–38 (2007).
Szolar, D. H. et al. Adrenocortical carcinomas and adrenal pheochromocytomas: mass and enhancement loss evaluation at delayed contrast-enhanced CT. Radiology 234, 479–485 (2005).
Park, B. K., Kim, C. K., Kim, B. & Lee, J. H. Comparison of delayed enhanced CT and chemical shift MR for evaluating hyperattenuating incidental adrenal masses. Radiology 243, 760–765 (2007).
Hönigschnabl, S. et al. How accurate is MR imaging in characterisation of adrenal masses: update of a long-term study. Eur. J. Radiol. 41, 113–122 (2002).
Groussin, L. et al. 18F-Fluorodeoxyglucose positron emission tomography for the diagnosis of adrenocortical tumors: a prospective study in 77 operated patients. J. Clin. Endocrinol. Metab. 94, 1713–1722 (2009).
Hahner, S. et al. [123 I]Iodometomidate for molecular imaging of adrenocortical cytochrome P450 family 11B enzymes. J. Clin. Endocrinol. Metab. 93, 2358–2365 (2008).
Khan, T. S. et al. 11C-metomidate PET imaging of adrenocortical cancer. Eur. J. Nucl. Med. Mol. Imaging 30, 403–410 (2003).
Hennings, J. et al. [11C]metomidate positron emission tomography of adrenocortical tumors in correlation with histopathological findings. J. Clin. Endocrinol. Metab. 91, 1410–1414 (2006).
Sasano, H. et al. Transcription factor adrenal 4 binding protein as a marker of adrenocortical malignancy. Hum. Pathol. 26, 1154–1156 (1995).
Hough, A. J., Hollifield, J. W., Page, D. L. & Hartmann, W. H. Prognostic factors in adrenal cortical tumors. A mathematical analysis of clinical and morphologic data. Am. J. Clin. Pathol. 72, 390–399 (1979).
van Slooten, H., Schaberg, A., Smeenk, D. & Moolenaar, A. J. Morphologic characteristics of benign and malignant adrenocortical tumors. Cancer 55, 766–773 (1985).
Weiss, L. M., Medeiros, L. J. & Vickery, A. L. Jr. Pathologic features of prognostic significance in adrenocortical carcinoma. Am. J. Surg. Pathol. 13, 202–206 (1989).
Weiss, L. M. Comparative histologic study of 43 metastasizing and nonmetastasizing adrenocortical tumors. Am. J. Surg. Pathol. 8, 163–169 (1984).
Morimoto, R. et al. Immunohistochemistry of a proliferation marker Ki67/MIB1 in adrenocortical carcinomas: Ki67/MIB1 labeling index is a predictor for recurrence of adrenocortical carcinomas. Endocr. J. 55, 49–55 (2008).
Terzolo, M. et al. Immunohistochemical assessment of Ki-67 in the differential diagnosis of adrenocortical tumors. Urology 57, 176–182 (2001).
Fassina, A. S., Borsato, S. & Fedeli, U. Fine needle aspiration cytology (FNAC) of adrenal masses. Cytopathology 11, 302–311 (2000).
Saeger, W. et al. High diagnostic accuracy of adrenal core biopsy: results of the German and Austrian adrenal network multicenter trial in 220 consecutive patients. Hum. Pathol. 34, 180–186 (2003).
Lumachi, F. et al. Fine-needle aspiration cytology of adrenal masses in noncancer patients: clinicoradiologic and histologic correlations in functioning and nonfunctioning tumors. Cancer 93, 323–329 (2001).
Lumachi, F. et al. Role and cost-effectiveness of adrenal imaging and image-guided FNA cytology in the management of incidentally discovered adrenal tumours. Anticancer Res. 25, 4559–4562 (2005).
Quayle, F. J. et al. Needle biopsy of incidentally discovered adrenal masses is rarely informative and potentially hazardous. Surgery 142, 497–502 (2007).
Fassnacht, M. et al. Limited prognostic value of the 2004 International Union Against Cancer staging classification for adrenocortical carcinoma: proposal for a revised TNM classification. Cancer 115, 243–250 (2009).
Lee, J. E. et al. Surgical management, DNA content, and patient survival in adrenal cortical carcinoma. Surgery 118, 1090–1098 (1995).
Lughezzani, G. et al. The European Network for the Study of Adrenal Tumors staging system is prognostically superior to the international union against cancer-staging system: a North American validation. Eur. J. Cancer 46, 713–719 (2010).
Schteingart, D. E. et al. Management of patients with adrenal cancer: recommendations of an international consensus conference. Endocr. Relat. Cancer 12, 667–680 (2005).
Porpiglia, F. et al. Retrospective evaluation of the outcome of open versus laparoscopic adrenalectomy for stage I and II adrenocortical cancer. Eur. Urol. 57, 873–878 (2010).
Brix, D. et al. Laparoscopic versus open adrenalectomy for adrenocortical carcinoma: surgical and oncologic outcome in 152 patients. Eur. Urol. 58, 609–615 (2010).
Miller, B. S. et al. Laparoscopic resection is inappropriate in patients with known or suspected adrenocortical carcinoma. World J. Surg. 34, 1380–1385 (2010).
Leboulleux, S. et al. Adrenocortical carcinoma: is the surgical approach a risk factor of peritoneal carcinomatosis? Eur. J. Endocrinol. 162, 1147–1153 (2010).
Murphy, M. M. et al. Trends in adrenalectomy: a recent national review. Surg. Endosc. 24, 2518–2526 (2010).
Terzolo, M. et al. Adjuvant mitotane treatment for adrenocortical carcinoma. N. Engl. J. Med. 356, 2372–2380 (2007).
Huang, H. & Fojo, T. Adjuvant mitotane for adrenocortical cancer—a recurring controversy. J. Clin. Endocrinol. Metab. 93, 3730–3732 (2008).
Terzolo, M., Fassnacht, M., Ciccone, G., Allolio, B. & Berruti, A. Adjuvant mitotane for adrenocortical cancer--working through uncertainty. J. Clin. Endocrinol. Metab. 94, 1879–1880 (2009).
Berruti, A. et al. Adjuvant therapy in patients with adrenocortical carcinoma: a position of an international panel. J. Clin. Oncol. 28, e401–e402 (2010).
Fassnacht, M. et al. Efficacy of adjuvant radiotherapy of the tumor bed on local recurrence of adrenocortical carcinoma. J. Clin. Endocrinol. Metab. 91, 4501–4504 (2006).
Polat, B. et al. Radiotherapy in adrenocortical carcinoma. Cancer 115, 2816–2823 (2009).
Sabolch, A. et al. Adjuvant and definitive radiotherapy for adrenocortical carcinoma. Int. J. Radiat. Oncol. Biol. Phys. doi:10.1016/j.ijrobp.2010.04.030.
Fassnacht, M. et al. Improved survival in patients with stage II adrenocortical carcinoma followed up prospectively by specialized centers. J. Clin. Endocrinol. Metab. 95, 4925–4932 (2010).
US National Institutes of Health Clinicaltrials.gov [online], http://clinicaltrials.gov/ct2/show/NCT00777244 (2010).
Bauditz, J., Quinkler, M. & Wermke, W. Radiofrequency thermal ablation of hepatic metastases of adrenocortical cancer—a case report and review of the literature. Exp. Clin. Endocrinol. Diabetes 117, 316–319 (2009).
Wood, B. J., Abraham, J., Hvizda, J. L., Alexander, H. R. & Fojo, T. Radiofrequency ablation of adrenal tumors and adrenocortical carcinoma metastases. Cancer 97, 554–560 (2003).
Jensen, J. C., Pass, H. I., Sindelar, W. F. & Norton, J. A. Recurrent or metastatic disease in select patients with adrenocortical carcinoma. Aggressive resection vs chemotherapy. Arch. Surg. 126, 457–461 (1991).
Pommier, R. F. & Brennan, M. F. An eleven-year experience with adrenocortical carcinoma. Surgery 112, 963–970 (1992).
Bellantone, R. et al. Role of reoperation in recurrence of adrenal cortical carcinoma: results from 188 cases collected in the Italian National Registry for Adrenal Cortical Carcinoma. Surgery 122, 1212–1218 (1997).
Schulick, R. D. & Brennan, M. F. Long-term survival after complete resection and repeat resection in patients with adrenocortical carcinoma. Ann. Surg. Oncol. 6, 719–726 (1999).
Erdogan, I. et al. Impact of surgery on clinical outcome in patients with recurrence of adrenocortical carcinoma. Endocrine Abstracts (10th European Congress of Endocrinology), 20, P194 (2009).
Khan, T. S. et al. Streptozocin and o,p'DDD in the treatment of adrenocortical cancer patients: long-term survival in its adjuvant use. Ann. Oncol. 11, 1281–1287 (2000).
First International Randomized Trial in locally advanced and Metastatic Adrenocortical Cancer Treatment (FIRM-ACT) [online], http://www.firm-act.org/ (2010).
Berruti, A. et al. Etoposide, doxorubicin and cisplatin plus mitotane in the treatment of advanced adrenocortical carcinoma: a large prospective phase II trial. Endocr. Relat. Cancer 12, 657–666 (2005).
Hahner, S. & Fassnacht, M. Mitotane for adrenocortical carcinoma treatment. Curr. Opin. Investig. Drugs 6, 386–394 (2005).
Daffara, F. et al. Prospective evaluation of mitotane toxicity in adrenocortical cancer patients treated adjuvantly. Endocr. Relat. Cancer 15, 1043–1053 (2008).
Sperone, P. et al. Gemcitabine plus metronomic 5-fluorouracil or capecitabine as a second-/third-line chemotherapy in advanced adrenocortical carcinoma: a multicenter phase II study. Endocr. Relat. Cancer 17, 445–453 (2010).
Hermsen, I. G., Groenen, Y. E., Dercksen, M. W., Theuws, J. & Haak, H. R. Response to radiation therapy in adrenocortical carcinoma. J. Endocrinol. Invest. doi:10.3275/6904.
Miller, J. W. & Crapo, L. The medical treatment of Cushing's syndrome. Endocr. Rev. 14, 443–458 (1993).
Schulte, H. M., Benker, G., Reinwein, D., Sippell, W. G. & Allolio, B. Infusion of low dose etomidate: correction of hypercortisolemia in patients with Cushing's syndrome and dose-response relationship in normal subjects. J. Clin. Endocrinol. Metab. 70, 1426–1430 (1990).
Fassnacht, M. et al. New mechanisms of adrenostatic compounds in a human adrenocortical cancer cell line. Eur. J. Clin. Invest. 30 (Suppl. 3), 76–82 (2000).
Johanssen, S. & Allolio, B. Mifepristone (RU 486) in Cushing's syndrome. Eur. J. Endocrinol. 157, 561–569 (2007).
Castinetti, F. et al. Merits and pitfalls of mifepristone in Cushing's syndrome. Eur. J. Endocrinol. 160, 1003–1010 (2009).
Quinkler, M. et al. Treatment of advanced adrenocortical carcinoma with erlotinib plus gemcitabine. J. Clin. Endocrinol. Metab. 93, 2057–2062 (2008).
Wortmann, S. et al. Bevacizumab plus capecitabine as a salvage therapy in advanced adrenocortical carcinoma. Eur. J. Endocrinol. 162, 349–356 (2010).
Fassnacht, M., Kreissl, M. C., Weismann, D. & Allolio, B. New targets and therapeutic approaches for endocrine malignancies. Pharmacol. Ther. 123, 117–141 (2009).
Berruti, A. et al. Emerging drugs for adrenocortical carcinoma. Expert Opin. Emerg. Drugs 13, 497–509 (2008).
Carden, C. P. et al. Phase I study of intermittent dosing of OSI-906, a dual tyrosine kinase inhibitor of insulin-like growth factor-1 receptor (IGF-1R) and insulin receptor (IR) in patients with advanced solid tumors. J. Clin. Oncol. 28 (Suppl.), abstr 2530 (2010).
Venkatesh, S., Hickey, R. C., Sellin, R. V., Fernandez, J. F. & Samaan, N. A. Adrenal cortical carcinoma. Cancer 64, 765–769 (1989).
Vassilopoulou-Sellin, R. & Schultz, P. N. Adrenocortical carcinoma. Clinical outcome at the end of the 20th century. Cancer 92, 1113–1121 (2001).
Stojadinovic, A. et al. Adrenocortical carcinoma: clinical, morphologic, and molecular characterization. J. Clin. Oncol. 20, 941–950 (2002).
Assié, G. et al. Prognostic parameters of metastatic adrenocortical carcinoma. J. Clin. Endocrinol. Metab. 92, 148–154 (2007).
Volante, M. et al. Matrix metalloproteinase type 2 expression in malignant adrenocortical tumors: Diagnostic and prognostic significance in a series of 50 adrenocortical carcinomas. Mod. Pathol. 19, 1563–1569 (2006).
Fenske, W. et al. Glucose transporter GLUT1 expression is an stage-independent predictor of clinical outcome in adrenocortical carcinoma. Endocr. Relat. Cancer 16, 919–928 (2009).
Soon, P. S. et al. Microarray gene expression and immunohistochemistry analyses of adrenocortical tumors identify IGF2 and Ki-67 as useful in differentiating carcinomas from adenomas. Endocr. Relat. Cancer 16, 573–583 (2009).
Ronchi, C. L. et al. Expression of excision repair cross complementing group 1 and prognosis in adrenocortical carcinoma patients treated with platinum-based chemotherapy. Endocr. Relat. Cancer 16, 907–918 (2009).
Malandrino, P. et al. Prognostic markers of survival after combined mitotane- and platinum-based chemotherapy in metastatic adrenocortical carcinoma (ACC). Endocr. Relat. Cancer 17, 797–807 (2010).
Acknowledgements
Work described in this Review was supported by grants of the Deutsche Krebshilfe (grant #107111 to M. Fassnacht), the German Ministry of Research BMBF (grant #01KG0501 to M. Fassnacht and B. Allolio) and the German Research Foundation DFG (grant #FA466/3-1 to M. Fassnacht). C. P. Vega, University of California, Irvine, CA, is the author of and is solely responsible for the content of the learning objectives, questions and answers of the Medscape, LLC-accredited continuing medical education activity associated with this article.
Author information
Authors and Affiliations
Contributions
All authors researched the data for the article, provided a substantial contribution to discussions of the content, wrote the review and reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
M. Fassnacht and B. Allolio are participating as investigators in clinical trials on the pharmacokinetics of mitotane sponsored by HRA Pharma (France) and on the efficacy of OSI-906 in advanced ACC sponsored by OSI Pharma (USA). The other authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Fassnacht, M., Libé, R., Kroiss, M. et al. Adrenocortical carcinoma: a clinician's update. Nat Rev Endocrinol 7, 323–335 (2011). https://doi.org/10.1038/nrendo.2010.235
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2010.235
This article is cited by
-
Management of endocrine surgical disorders during COVID-19 pandemic: expert opinion for non-surgical options
Updates in Surgery (2022)
-
Adrenal cortex renewal in health and disease
Nature Reviews Endocrinology (2021)
-
Neutrophil‐to‐Lymphocyte Ratio (NLR) and Platelet‐to‐Lymphocyte Ratio (PLR) as Possible Prognostic Markers for Patients Undergoing Resection of Adrenocortical Carcinoma
World Journal of Surgery (2021)
-
Sum of High-Risk Gene Mutation (SHGM): A Novel Attempt to Assist Differential Diagnosis for Adrenocortical Carcinoma with Benign Adenoma, Based on Detection of Mutations of Nine Target Genes
Biochemical Genetics (2021)
-
A phase 1 study of nevanimibe HCl, a novel adrenal-specific sterol O-acyltransferase 1 (SOAT1) inhibitor, in adrenocortical carcinoma
Investigational New Drugs (2020)