Key Points
-
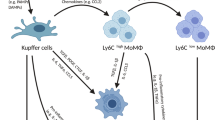
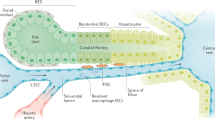
Liver macrophages comprise Kupffer cells — which are self-maintaining, non-migratory tissue-resident phagocytes that originate from yolk sac-derived precursors during embryogenesis — and monocyte-derived macrophages.
-
Kupffer cells are essential for hepatic and systemic homeostasis, as they contribute to metabolism, scavenge bacteria and cellular debris, and induce immunological tolerance.
-
Following their activation by danger signals, Kupffer cells modulate inflammation and recruit immune cells — including large numbers of monocytes — to the liver.
-
Kupffer cells and monocyte-derived macrophages rapidly adapt their phenotypes in response to local signals, which determine their ability to aggravate or cease liver injury.
-
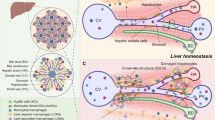
Liver macrophages are crucial in the pathogenesis of acute and chronic liver diseases, in which they orchestrate inflammation, fibrosis, angiogenesis and tumour progression, as well as tissue repair and tumour surveillance.
-
Evidence from animal models and early clinical trials in humans indicates that targeting pathogenic liver macrophages might be a promising therapeutic approach in acute and chronic liver diseases.
Abstract
Macrophages represent a key cellular component of the liver, and are essential for maintaining tissue homeostasis and ensuring rapid responses to hepatic injury. Our understanding of liver macrophages has been revolutionized by the delineation of heterogeneous subsets of these cells. Kupffer cells are a self-sustaining, liver-resident population of macrophages and can be distinguished from the monocyte-derived macrophages that rapidly accumulate in the injured liver. Specific environmental signals further determine the polarization and function of hepatic macrophages. These cells promote the restoration of tissue integrity following liver injury or infection, but they can also contribute to the progression of liver diseases, including hepatitis, fibrosis and cancer. In this Review, we highlight novel findings regarding the origin, classification and function of hepatic macrophages, and we discuss their divergent roles in the healthy and diseased liver.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Guilliams, M. et al. Unsupervised high-dimensional analysis aligns dendritic cells across tissues and species. Immunity 45, 669–684 (2016).
Lopez, B. G., Tsai, M. S., Baratta, J. L., Longmuir, K. J. & Robertson, R. T. Characterization of Kupffer cells in livers of developing mice. Comp. Hepatol. 10, 2 (2011).
Davies, L. C., Jenkins, S. J., Allen, J. E. & Taylor, P. R. Tissue-resident macrophages. Nat. Immunol. 14, 986–995 (2013).
Tacke, F. & Zimmermann, H. W. Macrophage heterogeneity in liver injury and fibrosis. J. Hepatol. 60, 1090–1096 (2014).
Scott, C. L. et al. Bone marrow-derived monocytes give rise to self-renewing and fully differentiated Kupffer cells. Nat. Commun. 7, 10321 (2016). This work identifies CLEC4F as a unique Kupffer cell marker in mice and demonstrates the ability of monocyte-derived macrophages to acquire a Kupffer cell-like phenotype after full Kupffer cell depletion.
von Kupffer, K. W. in Über Sternzellen der Leber. Arch. Mikroskop Anat. 12, 353–358 (in German) (1876). This paper provides a historic description of 'Kupffer stellate cells' as unrecognized non-parenchymal cells in the liver that we now know include both hepatic macrophages (Kupffer cells) and hepatic stellate cells.
Gammella, E., Buratti, P., Cairo, G. & Recalcati, S. Macrophages: central regulators of iron balance. Metallomics 6, 1336–1345 (2014).
Theurl, M. et al. Kupffer cells modulate iron homeostasis in mice via regulation of hepcidin expression. J. Mol. Med. (Berl.) 86, 825–835 (2008).
Naito, M., Hasegawa, G., Ebe, Y. & Yamamoto, T. Differentiation and function of Kupffer cells. Med. Electron Microsc. 37, 16–28 (2004).
Willekens, F. L. et al. Liver Kupffer cells rapidly remove red blood cell-derived vesicles from the circulation by scavenger receptors. Blood 105, 2141–2145 (2005).
Kristiansen, M. et al. Identification of the haemoglobin scavenger receptor. Nature 409, 198–201 (2001).
Wang, Y. et al. Plasma cholesteryl ester transfer protein is predominantly derived from Kupffer cells. Hepatology 62, 1710–1722 (2015).
Gomez Perdiguero, E. et al. Tissue-resident macrophages originate from yolk-sac-derived erythro-myeloid progenitors. Nature 518, 547–551 (2015).
Hoeffel, G. et al. C-Myb+ erythro-myeloid progenitor-derived fetal monocytes give rise to adult tissue-resident macrophages. Immunity 42, 665–678 (2015).
Sheng, J., Ruedl, C. & Karjalainen, K. Fetal HSCs versus EMP2s. Immunity 43, 1025 (2015).
Kim, K. W., Zhang, N., Choi, K. & Randolph, G. J. Homegrown macrophages. Immunity 45, 468–470 (2016).
Mass, E. et al. Specification of tissue-resident macrophages during organogenesis. Science 353, aaf4238 (2016). This study identifies CX 3 CR1+ and ID3+ progenitors that infiltrate the fetal liver during embryogenesis and give rise to self-maintaining Kupffer cells.
Soucie, E. L. et al. Lineage-specific enhancers activate self-renewal genes in macrophages and embryonic stem cells. Science 351, aad5510 (2016).
Lavin, Y. et al. Tissue-resident macrophage enhancer landscapes are shaped by the local microenvironment. Cell 159, 1312–1326 (2014).
Heymann, F. et al. Liver inflammation abrogates immunological tolerance induced by Kupffer cells. Hepatology 62, 279–291 (2015). This study shows that while Kupffer cells maintain liver tolerance during homeostasis, Kupffer cells and monocyte-derived macrophages switch to an immunogenic phenotype in the setting of chronic liver injury.
You, Q., Cheng, L., Kedl, R. M. & Ju, C. Mechanism of T cell tolerance induction by murine hepatic Kupffer cells. Hepatology 48, 978–990 (2008).
Mossanen, J. C. et al. Chemokine (C-C motif) receptor 2-positive monocytes aggravate the early phase of acetaminophen-induced acute liver injury. Hepatology 64, 1667–1682 (2016).
Theurl, I. et al. On-demand erythrocyte disposal and iron recycling requires transient macrophages in the liver. Nat. Med. 22, 945–951 (2016).
Eaves, C. J. Hematopoietic stem cells: concepts, definitions, and the new reality. Blood 125, 2605–2613 (2015).
Auffray, C. et al. CX3CR1+ CD115+ CD135+ common macrophage/DC precursors and the role of CX3CR1 in their response to inflammation. J. Exp. Med. 206, 595–606 (2009).
Liu, K. et al. In vivo analysis of dendritic cell development and homeostasis. Science 324, 392–397 (2009).
Fogg, D. K. et al. A clonogenic bone marrow progenitor specific for macrophages and dendritic cells. Science 311, 83–87 (2006).
Hettinger, J. et al. Origin of monocytes and macrophages in a committed progenitor. Nat. Immunol. 14, 821–830 (2013).
Dai, X. M. et al. Targeted disruption of the mouse colony-stimulating factor 1 receptor gene results in osteopetrosis, mononuclear phagocyte deficiency, increased primitive progenitor cell frequencies, and reproductive defects. Blood 99, 111–120 (2002).
Marra, F. & Tacke, F. Roles for chemokines in liver disease. Gastroenterology 147, 577–594.e1 (2014).
Stutchfield, B. M. et al. CSF1 restores innate immunity after liver injury in mice and serum levels indicate outcomes of patients with acute liver failure. Gastroenterology 149, 1896–1909.e14 (2015).
Varol, C., Mildner, A. & Jung, S. Macrophages: development and tissue specialization. Annu. Rev. Immunol. 33, 643–675 (2015).
Yona, S. et al. Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity 38, 79–91 (2013).
Varol, C. et al. Monocytes give rise to mucosal, but not splenic, conventional dendritic cells. J. Exp. Med. 204, 171–180 (2007).
Hanna, R. N. et al. The transcription factor NR4A1 (Nur77) controls bone marrow differentiation and the survival of Ly6C− monocytes. Nat. Immunol. 12, 778–785 (2011).
Dal-Secco, D. et al. A dynamic spectrum of monocytes arising from the in situ reprogramming of CCR2+ monocytes at a site of sterile injury. J. Exp. Med. 212, 447–456 (2015). This study shows that CCR2+CX 3 CR1− infiltrating monocytes transition into pro-repair CCR2−CX 3 CR1+ macrophages directly at the site of sterile liver injury.
Ingersoll, M. A. et al. Comparison of gene expression profiles between human and mouse monocyte subsets. Blood 115, e10–e19 (2010).
Antoniades, C. G. et al. Secretory leukocyte protease inhibitor: a pivotal mediator of anti-inflammatory responses in acetaminophen-induced acute liver failure. Hepatology 59, 1564–1576 (2014).
Antoniades, C. G. et al. Source and characterization of hepatic macrophages in acetaminophen-induced acute liver failure in humans. Hepatology 56, 735–746 (2012).
Antoniades, C. G. et al. Reduced monocyte HLA-DR expression: a novel biomarker of disease severity and outcome in acetaminophen-induced acute liver failure. Hepatology 44, 34–43 (2006).
Liaskou, E. et al. Monocyte subsets in human liver disease show distinct phenotypic and functional characteristics. Hepatology 57, 385–398 (2013).
Zimmermann, H. W. et al. Functional contribution of elevated circulating and hepatic non-classical CD14+CD16+ monocytes to inflammation and human liver fibrosis. PLoS ONE 5, e11049 (2010).
Naik, S. H. et al. Development of plasmacytoid and conventional dendritic cell subtypes from single precursor cells derived in vitro and in vivo. Nat. Immunol. 8, 1217–1226 (2007).
Onai, N. et al. A clonogenic progenitor with prominent plasmacytoid dendritic cell developmental potential. Immunity 38, 943–957 (2013).
Trombetta, E. S. & Mellman, I. Cell biology of antigen processing in vitro and in vivo. Annu. Rev. Immunol. 23, 975–1028 (2005).
David, B. A. et al. Combination of mass cytometry and imaging analysis reveals origin, location, and functional repopulation of liver myeloid cells in mice. Gastroenterology 151, 1176–1191 (2016). This study describes an unbiased technical approach for reclassifying hepatic myeloid cell populations in healthy mouse liver.
Miller, J. C. et al. Deciphering the transcriptional network of the dendritic cell lineage. Nat. Immunol. 13, 888–899 (2012).
Eckert, C., Klein, N., Kornek, M. & Lukacs-Kornek, V. The complex myeloid network of the liver with diverse functional capacity at steady state and in inflammation. Front. Immunol. 6, 179 (2015).
Heymann, F. et al. Long term intravital multiphoton microscopy imaging of immune cells in healthy and diseased liver using CXCR6.Gfp reporter mice. J. Vis. Exp. http://dx.doi.org/10.3791/52607 (2015).
Zigmond, E. et al. Infiltrating monocyte-derived macrophages and resident Kupffer cells display different ontogeny and functions in acute liver injury. J. Immunol. 193, 344–353 (2014).
Heymann, F. & Tacke, F. Immunology in the liver — from homeostasis to disease. Nat. Rev. Gastroenterol. Hepatol. 13, 88–110 (2016).
Karlmark, K. R. et al. Hepatic recruitment of the inflammatory Gr1+ monocyte subset upon liver injury promotes hepatic fibrosis. Hepatology 50, 261–274 (2009). This is the first paper to demonstrate that infiltrating LY6C+ monocytes stimulate the progression of liver fibrosis.
Reid, D. T. et al. Kupffer cells undergo fundamental changes during the development of experimental NASH and are critical in initiating liver damage and inflammation. PLoS ONE 11, e0159524 (2016).
Carlin, L. M. et al. Nr4a1-dependent Ly6Clow monocytes monitor endothelial cells and orchestrate their disposal. Cell 153, 362–375 (2013).
Xue, J. et al. Transcriptome-based network analysis reveals a spectrum model of human macrophage activation. Immunity 40, 274–288 (2014). This study uses transcriptomic profiling of human monocyte-derived macrophages that were exposed to various stimuli to identify multiple unique macrophage polarization clusters other than the classical M1 and M2 polarization phenotypes.
Beattie, L. et al. Bone marrow-derived and resident liver macrophages display unique transcriptomic signatures but similar biological functions. J. Hepatol. 65, 758–768 (2016).
Wang, J. & Kubes, P. A reservoir of mature cavity macrophages that can rapidly invade visceral organs to affect tissue repair. Cell 165, 668–678 (2016). This work shows that GATA6+ peritoneal macrophages can invade the subcapsular region of the liver following sterile injury and inflammation.
Bartneck, M. et al. Histidine-rich glycoprotein promotes macrophage activation and inflammation in chronic liver disease. Hepatology 63, 1310–1324 (2016).
Tu, Z. et al. TLR-dependent cross talk between human Kupffer cells and NK cells. J. Exp. Med. 205, 233–244 (2008).
Bouchery, T. et al. ILC2s and T cells cooperate to ensure maintenance of M2 macrophages for lung immunity against hookworms. Nat. Commun. 6, 6970 (2015).
Hams, E. et al. IL-25 and type 2 innate lymphoid cells induce pulmonary fibrosis. Proc. Natl Acad. Sci. USA 111, 367–372 (2014).
Barron, L. & Wynn, T. A. Fibrosis is regulated by Th2 and Th17 responses and by dynamic interactions between fibroblasts and macrophages. Am. J. Physiol. Gastrointest. Liver Physiol. 300, G723–G728 (2011).
Okabe, Y. & Medzhitov, R. Tissue biology perspective on macrophages. Nat. Immunol. 17, 9–17 (2016).
Zannetti, C. et al. Characterization of the inflammasome in human Kupffer cells in response to synthetic agonists and pathogens. J. Immunol. 197, 356–367 (2016).
Wree, A. & Marra, F. The inflammasome in liver disease. J. Hepatol. 65, 1055–1056 (2016).
Kubes, P. & Mehal, W. Z. Sterile inflammation in the liver. Gastroenterology 143, 1158–1172 (2012).
Toki, Y. et al. Extracellular ATP induces P2X7 receptor activation in mouse Kupffer cells, leading to release of IL-1β, HMGB1, and PGE2, decreased MHC class I expression and necrotic cell death. Biochem. Biophys. Res. Commun. 458, 771–776 (2015).
McDonald, B. et al. Intravascular danger signals guide neutrophils to sites of sterile inflammation. Science 330, 362–366 (2010).
Amaral, S. S. et al. Altered responsiveness to extracellular ATP enhances acetaminophen hepatotoxicity. Cell Commun. Signal. 11, 10 (2013).
Kim, H. Y., Kim, S. J. & Lee, S. M. Activation of NLRP3 and AIM2 inflammasomes in Kupffer cells in hepatic ischemia/reperfusion. FEBS J. 282, 259–270 (2015).
Kamo, N. et al. ASC/caspase-1/IL-1β signaling triggers inflammatory responses by promoting HMGB1 induction in liver ischemia/reperfusion injury. Hepatology 58, 351–362 (2013).
Huang, H. et al. Histones activate the NLRP3 inflammasome in Kupffer cells during sterile inflammatory liver injury. J. Immunol. 191, 2665–2679 (2013).
Tsuchiya, K. et al. The adaptor ASC exacerbates lethal Listeria monocytogenes infection by mediating IL-18 production in an inflammasome-dependent and -independent manner. Eur. J. Immunol. 44, 3696–3707 (2014).
Wree, A. et al. NLRP3 inflammasome activation results in hepatocyte pyroptosis, liver inflammation, and fibrosis in mice. Hepatology 59, 898–910 (2014).
Vandanmagsar, B. et al. The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat. Med. 17, 179–188 (2011).
Schneider, K. M. et al. CX3CR1 is a gatekeeper for intestinal barrier integrity in mice: limiting steatohepatitis by maintaining intestinal homeostasis. Hepatology 62, 1405–1416 (2015).
Miele, L. et al. Increased intestinal permeability and tight junction alterations in nonalcoholic fatty liver disease. Hepatology 49, 1877–1887 (2009).
Storek, K. M. & Monack, D. M. Bacterial recognition pathways that lead to inflammasome activation. Immunol. Rev. 265, 112–129 (2015).
Triantafilou, M., Hughes, T. R., Morgan, B. P. & Triantafilou, K. Complementing the inflammasome. Immunology 147, 152–164 (2016).
McHedlidze, T. et al. Interleukin-33-dependent innate lymphoid cells mediate hepatic fibrosis. Immunity 39, 357–371 (2013).
Arshad, M. I., Piquet-Pellorce, C. & Samson, M. IL-33 and HMGB1 alarmins: sensors of cellular death and their involvement in liver pathology. Liver Int. 32, 1200–1210 (2012).
Noel, G. et al. Ablation of interaction between IL-33 and ST2+ regulatory T cells increases immune cell-mediated hepatitis and activated NK cells liver infiltration. Am. J. Physiol. Gastrointest. Liver Physiol. 311, G313–G323 (2016).
Vannella, K. M. et al. Combinatorial targeting of TSLP, IL-25, and IL-33 in type 2 cytokine-driven inflammation and fibrosis. Sci. Transl Med. 8, 337ra65 (2016).
Hoque, R., Farooq, A., Ghani, A., Gorelick, F. & Mehal, W. Z. Lactate reduces liver and pancreatic injury in Toll-like receptor- and inflammasome-mediated inflammation via GPR81-mediated suppression of innate immunity. Gastroenterology 146, 1763–1774 (2014).
Yan, J., Li, S. & Li, S. The role of the liver in sepsis. Int. Rev. Immunol. 33, 498–510 (2014).
Wong, C. H., Jenne, C. N., Petri, B., Chrobok, N. L. & Kubes, P. Nucleation of platelets with blood-borne pathogens on Kupffer cells precedes other innate immunity and contributes to bacterial clearance. Nat. Immunol. 14, 785–792 (2013).
Surewaard, B. G. et al. Identification and treatment of the Staphylococcus aureus reservoir in vivo. J. Exp. Med. 213, 1141–1151 (2016).
Bleriot, C. et al. Liver-resident macrophage necroptosis orchestrates type 1 microbicidal inflammation and type-2-mediated tissue repair during bacterial infection. Immunity 42, 145–158 (2015).
Jawhara, S. et al. Integrin αXβ2 is a leukocyte receptor for Candida albicans and is essential for protection against fungal infections. J. Immunol. 189, 2468–2477 (2012).
Jalan, R. et al. Bacterial infections in cirrhosis: a position statement based on the EASL Special Conference 2013. J. Hepatol. 60, 1310–1324 (2014).
Arroyo, V. et al. Acute-on-chronic liver failure: a new syndrome that will re-classify cirrhosis. J. Hepatol. 62, S131–S143 (2015).
Claria, J. et al. Systemic inflammation in decompensated cirrhosis: characterization and role in acute-on-chronic liver failure. Hepatology 64, 1249–1264 (2016).
Sole, C. et al. Characterization of inflammatory response in acute-on-chronic liver failure and relationship with prognosis. Sci. Rep. 6, 32341 (2016).
Laursen, T. L. et al. The soluble mannose receptor is released from the liver in cirrhotic patients, but is not associated with bacterial translocation. Liver Int. http://dx.doi.org/10.1111/liv.13262 (2016).
Bernsmeier, C. et al. Patients with acute-on-chronic liver failure have increased numbers of regulatory immune cells expressing the receptor tyrosine kinase MERTK. Gastroenterology 148, 603–615.e14 (2015).
Bernal, W. & Wendon, J. Acute liver failure. N. Engl. J. Med. 369, 2525–2534 (2013).
Erhardt, A., Biburger, M., Papadopoulos, T. & Tiegs, G. IL-10, regulatory T cells, and Kupffer cells mediate tolerance in concanavalin A-induced liver injury in mice. Hepatology 45, 475–485 (2007).
You, Q. et al. Role of hepatic resident and infiltrating macrophages in liver repair after acute injury. Biochem. Pharmacol. 86, 836–843 (2013).
Zhai, Y., Busuttil, R. W. & Kupiec-Weglinski, J. W. Liver ischemia and reperfusion injury: new insights into mechanisms of innate-adaptive immune-mediated tissue inflammation. Am. J. Transplant. 11, 1563–1569 (2011).
Movita, D. et al. Inflammatory monocytes recruited to the liver within 24 hours after virus-induced inflammation resemble Kupffer cells but are functionally distinct. J. Virol. 89, 4809–4817 (2015).
Heymann, F. et al. Hepatic macrophage migration and differentiation critical for liver fibrosis is mediated by the chemokine receptor C-C motif chemokine receptor 8 in mice. Hepatology 55, 898–909 (2012).
Nakamoto, N. et al. CCR9+ macrophages are required for acute liver inflammation in mouse models of hepatitis. Gastroenterology 142, 366–376 (2012).
Karlmark, K. R. et al. The fractalkine receptor CX3CR1 protects against liver fibrosis by controlling differentiation and survival of infiltrating hepatic monocytes. Hepatology 52, 1769–1782 (2010).
Dambach, D. M., Watson, L. M., Gray, K. R., Durham, S. K. & Laskin, D. L. Role of CCR2 in macrophage migration into the liver during acetaminophen-induced hepatotoxicity in the mouse. Hepatology 35, 1093–1103 (2002).
Wehr, A. et al. Chemokine receptor CXCR6-dependent hepatic NK T cell accumulation promotes inflammation and liver fibrosis. J. Immunol. 190, 5226–5236 (2013).
Bartocci, A. et al. Macrophages specifically regulate the concentration of their own growth factor in the circulation. Proc. Natl Acad. Sci. USA 84, 6179–6183 (1987).
Huebener, P. et al. The HMGB1/RAGE axis triggers neutrophil-mediated injury amplification following necrosis. J. Clin. Invest. 125, 539–550 (2015).
Zhang, J. et al. CCL2–CCR2 signaling promotes hepatic ischemia/reperfusion injury. J. Surg. Res. 202, 352–362 (2016).
Williams, C. D. et al. Neutrophil activation during acetaminophen hepatotoxicity and repair in mice and humans. Toxicol. Appl. Pharmacol. 275, 122–133 (2014).
Holt, M. P., Cheng, L. & Ju, C. Identification and characterization of infiltrating macrophages in acetaminophen-induced liver injury. J. Leukoc. Biol. 84, 1410–1421 (2008).
Ohkubo, H. et al. VEGFR1-positive macrophages facilitate liver repair and sinusoidal reconstruction after hepatic ischemia/reperfusion injury. PLoS ONE 9, e105533 (2014).
Weiskirchen, R. & Tacke, F. Liver fibrosis: from pathogenesis to novel therapies. Dig. Dis. 34, 410–422 (2016).
Younossi, Z. M. et al. The economic and clinical burden of nonalcoholic fatty liver disease in the United States and Europe. Hepatology 64, 1577–1586 (2016).
Tilg, H., Moschen, A. R. & Szabo, G. Interleukin-1 and inflammasomes in alcoholic liver disease/acute alcoholic hepatitis and nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. Hepatology 64, 955–965 (2016).
Huang, W. et al. Depletion of liver Kupffer cells prevents the development of diet-induced hepatic steatosis and insulin resistance. Diabetes 59, 347–357 (2010).
Hirsova, P. et al. Lipid-induced signaling causes release of inflammatory extracellular vesicles from hepatocytes. Gastroenterology 150, 956–967 (2016).
Robert, O. et al. Decreased expression of the glucocorticoid receptor–GILZ pathway in Kupffer cells promotes liver inflammation in obese mice. J. Hepatol. 64, 916–924 (2016).
Wan, J. et al. M2 Kupffer cells promote M1 Kupffer cell apoptosis: a protective mechanism against alcoholic and nonalcoholic fatty liver disease. Hepatology 59, 130–142 (2014).
Lodder, J. et al. Macrophage autophagy protects against liver fibrosis in mice. Autophagy 11, 1280–1292 (2015).
Wang, M. et al. Chronic alcohol ingestion modulates hepatic macrophage populations and functions in mice. J. Leukoc. Biol. 96, 657–665 (2014).
Miura, K., Yang, L., van Rooijen, N., Ohnishi, H. & Seki, E. Hepatic recruitment of macrophages promotes nonalcoholic steatohepatitis through CCR2. Am. J. Physiol. Gastrointest. Liver Physiol. 302, G1310–G1321 (2012).
Zhang, X. et al. CXC chemokine receptor 3 promotes steatohepatitis in mice through mediating inflammatory cytokines, macrophages and autophagy. J. Hepatol. 64, 160–170 (2016).
Tomita, K. et al. CXCL10-mediates macrophage, but not other innate immune cells-associated inflammation in murine nonalcoholic steatohepatitis. Sci. Rep. 6, 28786 (2016).
Wu, R., Nakatsu, G., Zhang, X. & Yu, J. Pathophysiological mechanisms and therapeutic potentials of macrophages in non-alcoholic steatohepatitis. Expert Opin. Ther. Targets 20, 615–626 (2016).
Kazankov, K. et al. The macrophage activation marker sCD163 is associated with morphological disease stages in patients with non-alcoholic fatty liver disease. Liver Int. 36, 1549–1557 (2016).
Kazankov, K. et al. The macrophage activation marker sCD163 is associated with changes in NAFLD and metabolic profile during lifestyle intervention in obese children. Pediatr. Obes. 10, 226–233 (2015).
Wan, J., Benkdane, M., Alons, E., Lotersztajn, S. & Pavoine, C. M2 Kupffer cells promote hepatocyte senescence: an IL-6-dependent protective mechanism against alcoholic liver disease. Am. J. Pathol. 184, 1763–1772 (2014).
Petrasek, J. et al. IL-1 receptor antagonist ameliorates inflammasome-dependent alcoholic steatohepatitis in mice. J. Clin. Invest. 122, 3476–3489 (2012).
Bala, S. et al. The pro-inflammatory effects of miR-155 promote liver fibrosis and alcohol-induced steatohepatitis. J. Hepatol. 64, 1378–1387 (2016).
Navarro, L. A. et al. Arginase 2 deficiency results in spontaneous steatohepatitis: a novel link between innate immune activation and hepatic de novo lipogenesis. J. Hepatol. 62, 412–420 (2015).
Mandrekar, P., Ambade, A., Lim, A., Szabo, G. & Catalano, D. An essential role for monocyte chemoattractant protein-1 in alcoholic liver injury: regulation of proinflammatory cytokines and hepatic steatosis in mice. Hepatology 54, 2185–2197 (2011).
Martinez-Micaelo, N., Gonzalez-Abuin, N., Pinent, M., Ardevol, A. & Blay, M. Dietary fatty acid composition is sensed by the NLRP3 inflammasome: omega-3 fatty acid (DHA) prevents NLRP3 activation in human macrophages. Food Funct. 7, 3480–3487 (2016).
Vats, D. et al. Oxidative metabolism and PGC-1β attenuate macrophage-mediated inflammation. Cell Metab. 4, 13–24 (2006).
Huang, S. C. et al. Cell-intrinsic lysosomal lipolysis is essential for alternative activation of macrophages. Nat. Immunol. 15, 846–855 (2014).
Nomura, M. et al. Fatty acid oxidation in macrophage polarization. Nat. Immunol. 17, 216–217 (2016).
York, A. G. et al. Limiting cholesterol biosynthetic flux spontaneously engages type I IFN signaling. Cell 163, 1716–1729 (2015).
Wiktor, S. Z. & Hutin, Y. J. The global burden of viral hepatitis: better estimates to guide hepatitis elimination efforts. Lancet 388, 1030–1031 (2016).
Boltjes, A., Movita, D., Boonstra, A. & Woltman, A. M. The role of Kupffer cells in hepatitis B and hepatitis C virus infections. J. Hepatol. 61, 660–671 (2014).
Shrivastava, S., Mukherjee, A., Ray, R. & Ray, R. B. Hepatitis C virus induces interleukin-1β (IL-1β)/IL-18 in circulatory and resident liver macrophages. J. Virol. 87, 12284–12290 (2013).
Broering, R. et al. Toll-like receptor-stimulated non-parenchymal liver cells can regulate hepatitis C virus replication. J. Hepatol. 48, 914–922 (2008).
Boltjes, A. et al. Kupffer cells interact with hepatitis B surface antigen in vivo and in vitro, leading to proinflammatory cytokine production and natural killer cell function. J. Infect. Dis. 211, 1268–1278 (2015).
Huang, L. R. et al. Intrahepatic myeloid-cell aggregates enable local proliferation of CD8+ T cells and successful immunotherapy against chronic viral liver infection. Nat. Immunol. 14, 574–583 (2013).
Preisser, L. et al. IL-34 and macrophage colony-stimulating factor are overexpressed in hepatitis C virus fibrosis and induce profibrotic macrophages that promote collagen synthesis by hepatic stellate cells. Hepatology 60, 1879–1890 (2014).
Li, H. et al. Hepatitis B virus particles preferably induce Kupffer cells to produce TGF-β1 over pro-inflammatory cytokines. Dig. Liver Dis. 44, 328–333 (2012).
Rehermann, B. Pathogenesis of chronic viral hepatitis: differential roles of T cells and NK cells. Nat. Med. 19, 859–868 (2013).
Xu, L., Yin, W., Sun, R., Wei, H. & Tian, Z. Kupffer cell-derived IL-10 plays a key role in maintaining humoral immune tolerance in hepatitis B virus-persistent mice. Hepatology 59, 443–452 (2014).
Li, M. et al. Kupffer cells support hepatitis B virus-mediated CD8+ T cell exhaustion via hepatitis B core antigen-TLR2 interactions in mice. J. Immunol. 195, 3100–3109 (2015).
Tian, Y., Kuo, C. F., Akbari, O. & Ou, J. H. Maternal-derived hepatitis B virus e antigen alters macrophage function in offspring to drive viral persistence after vertical transmission. Immunity 44, 1204–1214 (2016).
Angulo, P. et al. Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology 149, 389–397.e10 (2015).
Duffield, J. S. et al. Selective depletion of macrophages reveals distinct, opposing roles during liver injury and repair. J. Clin. Invest. 115, 56–65 (2005).
Pradere, J. P. et al. Hepatic macrophages but not dendritic cells contribute to liver fibrosis by promoting the survival of activated hepatic stellate cells in mice. Hepatology 58, 1461–1473 (2013).
Baeck, C. et al. Pharmacological inhibition of the chemokine CCL2 (MCP-1) diminishes liver macrophage infiltration and steatohepatitis in chronic hepatic injury. Gut 61, 416–426 (2012).
Tosello-Trampont, A. C. et al. NKp46+ natural killer cells attenuate metabolism-induced hepatic fibrosis by regulating macrophage activation in mice. Hepatology 63, 799–812 (2016).
Seki, E. et al. TLR4 enhances TGF-β signaling and hepatic fibrosis. Nat. Med. 13, 1324–1332 (2007).
Hackstein, C. P. et al. Gut microbial translocation corrupts myeloid cell function to control bacterial infection during liver cirrhosis. Gut 66, 507–518 (2016).
Ramachandran, P. et al. Differential Ly-6C expression identifies the recruited macrophage phenotype, which orchestrates the regression of murine liver fibrosis. Proc. Natl Acad. Sci. USA 109, E3186–E3195 (2012). This is the first paper to demonstrate that infiltrating monocytes mature into LY6C− macrophages, which promote the resolution of liver fibrosis.
Pellicoro, A., Ramachandran, P., Iredale, J. P. & Fallowfield, J. A. Liver fibrosis and repair: immune regulation of wound healing in a solid organ. Nat. Rev. Immunol. 14, 181–194 (2014).
Tacke, F. & Trautwein, C. Mechanisms of liver fibrosis resolution. J. Hepatol. 63, 1038–1039 (2015).
Ju, C. & Tacke, F. Hepatic macrophages in homeostasis and liver diseases: from pathogenesis to novel therapeutic strategies. Cell. Mol. Immunol. 13, 316–327 (2016).
Thomas, J. A. et al. Macrophage therapy for murine liver fibrosis recruits host effector cells improving fibrosis, regeneration, and function. Hepatology 53, 2003–2015 (2011).
King, A. et al. REpeated AutoLogous Infusions of STem cells In Cirrhosis (REALISTIC): a multicentre, phase II, open-label, randomised controlled trial of repeated autologous infusions of granulocyte colony-stimulating factor (GCSF) mobilised CD133+ bone marrow stem cells in patients with cirrhosis. A study protocol for a randomised controlled trial. BMJ Open 5, e007700 (2015).
Bartneck, M., Warzecha, K. T. & Tacke, F. Therapeutic targeting of liver inflammation and fibrosis by nanomedicine. Hepatobiliary Surg. Nutr. 3, 364–376 (2014).
Bartneck, M. et al. Fluorescent cell-traceable dexamethasone-loaded liposomes for the treatment of inflammatory liver diseases. Biomaterials 37, 367–382 (2015).
Traber, P. G. & Zomer, E. Therapy of experimental NASH and fibrosis with galectin inhibitors. PLoS ONE 8, e83481 (2013).
Baeck, C. et al. Pharmacological inhibition of the chemokine C-C motif chemokine ligand 2 (monocyte chemoattractant protein 1) accelerates liver fibrosis regression by suppressing Ly-6C+ macrophage infiltration in mice. Hepatology 59, 1060–1072 (2014).
Oberthur, D. et al. Crystal structure of a mirror-image L-RNA aptamer (Spiegelmer) in complex with the natural L-protein target CCL2. Nat. Commun. 6, 6923 (2015).
Friedman, S. et al. Efficacy and safety study of cenicriviroc for the treatment of non-alcoholic steatohepatitis in adult subjects with liver fibrosis: CENTAUR phase 2b study design. Contemp. Clin. Trials 47, 356–365 (2016).
Sanyal, A. J. et al. Cenicriviroc placebo for the treatment of non-alcoholic steatohepatitis with liver fibrosis: results from the year 1 primary analysis of the phase 2b CENTAUR study. Hepatology 64, 1118A–1119A (2016).
Loomba, R. et al. GS-4997, an inhibitor of apoptosis signal-regulating kinase (ASK1), alone or in combination with simtuzumab for the treatment of nonalcoholic steatohepatitis (NASH): a randomized, phase 2 trial. Hepatology 64, 1119A–1120A (2016).
Capece, D. et al. The inflammatory microenvironment in hepatocellular carcinoma: a pivotal role for tumor-associated macrophages. Biomed. Res. Int. 2013, 187204 (2013).
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2016. CA Cancer J. Clin. 66, 7–30 (2016).
Bruix, J., Reig, M. & Sherman, M. Evidence-based diagnosis, staging, and treatment of patients with hepatocellular carcinoma. Gastroenterology 150, 835–853 (2016).
Mossanen, J. C. & Tacke, F. Role of lymphocytes in liver cancer. Oncoimmunology 2, e26468 (2013).
Medina-Echeverz, J., Eggert, T., Han, M. & Greten, T. F. Hepatic myeloid-derived suppressor cells in cancer. Cancer Immunol. Immunother. 64, 931–940 (2015).
Wan, S., Kuo, N., Kryczek, I., Zou, W. & Welling, T. H. Myeloid cells in hepatocellular carcinoma. Hepatology 62, 1304–1312 (2015).
Yeung, O. W. et al. Alternatively activated (M2) macrophages promote tumour growth and invasiveness in hepatocellular carcinoma. J. Hepatol. 62, 607–616 (2015).
Li, X. et al. Targeting of tumour-infiltrating macrophages via CCL2/CCR2 signalling as a therapeutic strategy against hepatocellular carcinoma. Gut 66, 157–167 (2017).
Ehling, J. et al. CCL2-dependent infiltrating macrophages promote angiogenesis in progressive liver fibrosis. Gut 63, 1960–1971 (2014).
Wu, J. et al. The proinflammatory myeloid cell receptor TREM-1 controls Kupffer cell activation and development of hepatocellular carcinoma. Cancer Res. 72, 3977–3986 (2012).
Zhou, S. L. et al. Tumor-associated neutrophils recruit macrophages and T-regulatory cells to promote progression of hepatocellular carcinoma and resistance to sorafenib. Gastroenterology 150, 1646–1658.e17 (2016).
Ding, T. et al. High tumor-infiltrating macrophage density predicts poor prognosis in patients with primary hepatocellular carcinoma after resection. Hum. Pathol. 40, 381–389 (2009).
Kuang, D. M. et al. Activated monocytes in peritumoral stroma of hepatocellular carcinoma foster immune privilege and disease progression through PD-L1. J. Exp. Med. 206, 1327–1337 (2009).
Lim, S. Y., Yuzhalin, A. E., Gordon-Weeks, A. N. & Muschel, R. J. Tumor-infiltrating monocytes/macrophages promote tumor invasion and migration by upregulating S100A8 and S100A9 expression in cancer cells. Oncogene 35, 5735–5745 (2016).
Elsegood, C. L. et al. Kupffer cell-monocyte communication is essential for initiating murine liver progenitor cell-mediated liver regeneration. Hepatology 62, 1272–1284 (2015).
Eggert, T. et al. Distinct functions of senescence-associated immune responses in liver tumor surveillance and tumor progression. Cancer Cell 30, 533–547 (2016). This study shows that while CCR2+ monocytes clear premalignant senescent cells in early hepatocarcinogenesis, they have a tumour-promoting effect — mediated by NK cell inhibition — in models of established HCC.
Afik, R. et al. Tumor macrophages are pivotal constructors of tumor collagenous matrix. J. Exp. Med. 213, 2315–2331 (2016).
Schneider, C. et al. Adaptive immunity suppresses formation and progression of diethylnitrosamine-induced liver cancer. Gut 61, 1733–1743 (2012).
Kang, T. W. et al. Senescence surveillance of pre-malignant hepatocytes limits liver cancer development. Nature 479, 547–551 (2011).
Ma, C. et al. NAFLD causes selective CD4+ T lymphocyte loss and promotes hepatocarcinogenesis. Nature 531, 253–257 (2016).
Makarova-Rusher, O. V., Medina-Echeverz, J., Duffy, A. G. & Greten, T. F. The yin and yang of evasion and immune activation in HCC. J. Hepatol. 62, 1420–1429 (2015).
Suk, K. T. et al. Opposite roles of cannabinoid receptors 1 and 2 in hepatocarcinogenesis. Gut 65, 1721–1732 (2016).
Halama, N. et al. Tumoral immune cell exploitation in colorectal cancer metastases can be targeted effectively by anti-CCR5 therapy in cancer patients. Cancer Cell 29, 587–601 (2016).
Murray, P. J. et al. Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity 41, 14–20 (2014).
Acknowledgements
This work was supported by the German Research Foundation (DFG; grants Ta434/3-1, Ta434/5-1 and SFB/TRR57) and by the Interdisciplinary Center for Clinical Research (IZKF), Aachen, Germany.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Work in the laboratory of F.T. has received research funding from Tobira Therapeutics (California, USA), Galapagos (Belgium) and Noxxon (Germany).
Glossary
- Portal triad
-
The anatomical structure within the liver in which branches of the hepatic artery, portal vein and bile ducts align. The portal triad is the typical region of pro-inflammatory and fibrotic responses in liver injury.
- Alarmins
-
Signal molecules released by damaged, diseased or dead cells that activate immune cells.
- Necroptosis
-
A specific variation of programmed necrosis, which is an inflammatory cell death process. Necroptosis is dependent on the activity of receptor-interacting protein kinases.
- Liver decompensation
-
A deterioration of liver function due to a precipitating event such as bleeding or infections. It is typically characterized by clinical symptoms (for example, ascites and hepatic encephalopathy), complications of portal hypertension (for example, variceal bleeding), and single organ or multi-organ failure.
- Hepatic stellate cells
-
Vitamin A-storing, star-shaped cells that have radially extending filaments. These cells are located in the subendothelial space in the liver and can trans-differentiate into extracellular matrix-producing myofibroblasts that promote liver fibrosis.
- Nonalcoholic fatty liver disease
-
(NAFLD). The most common type of liver disease in the world. It is characterized by an excessive accumulation of hepatic fat (steatosis in > 5% of hepatocytes), and is unrelated to alcohol consumption or other secondary causes, but associated with metabolic comorbidities such as obesity and insulin resistance.
- Nonalcoholic steatohepatitis
-
(NASH). The inflammatory stage of nonalcoholic fatty liver disease in which there is a high risk of progressing to cirrhosis or liver cancer. It is characterized by steatosis, hepatocyte ballooning and lobular inflammation, which are observed by liver histology.
- Myeloid-derived suppressor cells
-
(MDSCs). A heterogeneous population of myeloid cells that have potent immune-suppressive properties (for example, they produce anti-inflammatory cytokines, direct T cell suppression and induce the development of regulatory T cells). They arise in conditions of chronic inflammation, infections and tumours.
Rights and permissions
About this article
Cite this article
Krenkel, O., Tacke, F. Liver macrophages in tissue homeostasis and disease. Nat Rev Immunol 17, 306–321 (2017). https://doi.org/10.1038/nri.2017.11
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri.2017.11
This article is cited by
-
The roles of tissue resident macrophages in health and cancer
Experimental Hematology & Oncology (2024)
-
Reexamining the effects of drug loading on the in vivo performance of PEGylated liposomal doxorubicin
Acta Pharmacologica Sinica (2024)
-
Progress of immune checkpoint inhibitors therapy for non-small cell lung cancer with liver metastases
British Journal of Cancer (2024)
-
METTL16 promotes liver cancer stem cell self-renewal via controlling ribosome biogenesis and mRNA translation
Journal of Hematology & Oncology (2024)
-
Dietary chitosan reversed the toxic effects of polystyrene microplastics on Nile tilapia (Oreochromis niloticus) liver by inhibiting mitochondrial damage
Reviews in Fish Biology and Fisheries (2024)