Key Points
-
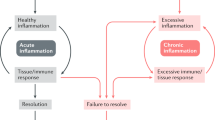
Autoinflammatory diseases are marked by uncontrolled inflammation in the absence of autoantibodies or autoreactive T cell responses.
-
The genetic basis of familial autoinflammatory diseases involves mutations in the genes encoding regulatory proteins with key roles in innate immune responses.
-
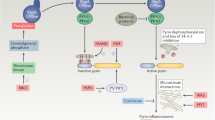
Autoinflammatory diseases involving increased production of interleukin-1β (IL-1β) result from enhanced activation of the NOD-, LRR- and pyrin domain-containing 3 (NLRP3) inflammasome, owing to activating NLRP3 mutations, or from loss-of-function mutations in interleukin-1 receptor antagonist.
-
Spondyloarthropathies and related conditions can arise from abnormal innate immune responses that are caused by misfolding and accumulation of the HLA-B27 protein.
-
The discovery of the genetic basis of autoinflammatory diseases has led to a new era of targeted therapies for these conditions.
Abstract
Autoinflammatory diseases are characterized by seemingly unprovoked pathological activation of the innate immune system in the absence of autoantibodies or autoreactive T cells. Discovery of the causative mutations underlying several monogenic autoinflammatory diseases has identified key regulators of innate immune responses. Recent studies have highlighted the role of misfolding, oligomerization and abnormal trafficking of pathogenic mutant proteins in triggering autoinflammation, and suggest that more common rheumatic diseases may have an autoinflammatory component. This coincides with recent discoveries of new links between endoplasmic reticulum stress and inflammatory signalling pathways, which support the emerging view that autoinflammatory diseases may be due to pathological dysregulation of stress-sensing pathways that normally function in host defence.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
McGonagle, D. & McDermott, M. F. A proposed classification of the immunological diseases. PLoS Med. 3, e297 (2006).
Masters, S. L., Simon, A., Aksentijevich, I. & Kastner, D. L. Horror autoinflammaticus: the molecular pathophysiology of autoinflammatory disease. Annu. Rev. Immunol. 27, 621–668 (2009).
Martinon, F., Petrilli, V., Mayor, A., Tardivel, A. & Tschopp, J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440, 237–241 (2006).
Schroder, M. & Kaufman, R. J. ER stress and the unfolded protein response. Mutat. Res. 569, 29–63 (2005).
Pipe, S. W. & Kaufman, R. J. Factor VIII C2 domain missense mutations exhibit defective trafficking of biologically functional proteins. J. Biol. Chem. 271, 25671–25676 (1996).
Kopito, R. R. Biosynthesis and degradation of CFTR. Physiol. Rev. 79, S167–S173 (1999).
Oyadomari, S. et al. Targeted disruption of the Chop gene delays endoplasmic reticulum stress-mediated diabetes. J. Clin. Invest. 109, 525–532 (2002).
Wang, J. et al. A mutation in the insulin 2 gene induces diabetes with severe pancreatic β-cell dysfunction in the Mody mouse. J. Clin. Invest. 103, 27–37 (1999).
Datta, R., Waheed, A., Bonapace, G., Shah, G. N. & Sly, W. S. Pathogenesis of retinitis pigmentosa associated with apoptosis-inducing mutations in carbonic anhydrase IV. Proc. Natl Acad. Sci. USA 106, 3437–3442 (2009).
Harraz, M. M. et al. SOD1 mutations disrupt redox-sensitive Rac regulation of NADPH oxidase in a familial ALS model. J. Clin. Invest. 118, 659–670 (2008).
Urano, F. et al. Coupling of stress in the ER to activation of JNK protein kinases by transmembrane protein kinase IRE1. Science 287, 664–666 (2000).
Zhang, K. et al. Endoplasmic reticulum stress activates cleavage of CREBH to induce a systemic inflammatory response. Cell 124, 587–599 (2006).
Martinon, F., Chen, X., Lee, A. H. & Glimcher, L. H. TLR activation of the transcription factor XBP1 regulates innate immune responses in macrophages. Nature Immunol. 11, 411–418 (2010).
Masters, S. L. et al. Activation of the NLRP3 inflammasome by islet amyloid polypeptide provides a mechanism for enhanced IL-1β in type 2 diabetes. Nature Immunol. 11, 897–904 (2010).
Halle, A. et al. The NALP3 inflammasome is involved in the innate immune response to amyloid-β. Nature Immunol. 9, 857–865 (2008).
Kastner, D., Aksentijevich, I. & Goldbach-Mansky, R. Autoinflammatory disease reloaded: a clinical perspective. Cell 140, 784–790 (2010). A concise review that highlights recent advances in the pathogenesis and treatment of autoinflammatory diseases.
Jordan, C. T. et al. PSORS2 is due to mutations in CARD14. Am. J. Hum. Genet. 90, 784–795 (2012).
Jordan, C. T. et al. Rare and common variants in CARD14, encoding an epidermal regulator of NF-κB, in psoriasis. Am. J. Hum. Genet. 90, 796–808 (2012).
Agarwal, A. K. et al. PSMB8 encoding the β5i proteasome subunit is mutated in joint contractures, muscle atrophy, microcytic anemia, and panniculitis-induced lipodystrophy syndrome. Am. J. Hum. Genet. 87, 866–872 (2010).
Arima, K. et al. Proteasome assembly defect due to a proteasome subunit β type 8 (PSMB8) mutation causes the autoinflammatory disorder, Nakajo-Nishimura syndrome. Proc. Natl Acad. Sci. USA 108, 14914–14919 (2011).
Kitamura, A., et al. A mutation in the immunoproteasome subunit PSMB8 causes autoinflammation and lipodystrophy in humans. J. Clin. Invest. 121, 4150–4160 (2011).
Liu, Y. et al. Mutations in PSMB8 cause CANDLE syndrome with evidence of genetic and phenotypic heterogeneity. Arthritis Rheum. (in the press). References 20, 21 and 22 describe recessive mutations in the PSMB8 immunoproteasome subunit that cause autoinflammatory syndromes.
Aksentijevich, I. et al. The clinical continuum of cryopyrinopathies: novel CIAS1 mutations in North American patients and a new cryopyrin model. Arthritis Rheum. 56, 1273–1285 (2007).
Strowig, T., Henao-Mejia, J., Elinav, E. & Flavell, R. Inflammasomes in health and disease. Nature 481, 278–286 (2012).
Henao-Mejia, J. et al. Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature 482, 179–185 (2012).
Elinav, E. et al. NLRP6 inflammasome regulates colonic microbial ecology and risk for colitis. Cell 145, 745–757 (2011).
Chen, G. Y., Liu, M., Wang, F., Bertin, J. & Nunez, G. A functional role for Nlrp6 in intestinal inflammation and tumorigenesis. J. Immunol. 186, 7187–7194 (2011).
Eisenbarth, S. C. & Flavell, R. A. Innate instruction of adaptive immunity revisited: the inflammasome. EMBO Mol. Med. 1, 92–98 (2009).
Jeru, I., Hayrapetyan, H., Duquesnoy, P., Sarkisian, T. & Amselem, S. PYPAF1 nonsense mutation in a patient with an unusual autoinflammatory syndrome: role of PYPAF1 in inflammation. Arthritis Rheum. 54, 508–514 (2006).
Tanaka, N. et al. High incidence of NLRP3 somatic mosaicism in patients with chronic infantile neurologic, cutaneous, articular syndrome: results of an International Multicenter Collaborative Study. Arthritis Rheum. 63, 3625–3632 (2011).
Kanneganti, T. D., Lamkanfi, M. & Nunez, G. Intracellular NOD-like receptors in host defense and disease. Immunity 27, 549–559 (2007).
Pelegrin, P. & Surprenant, A. Pannexin-1 mediates large pore formation and interleukin-1β release by the ATP-gated P2X7 receptor. EMBO J. 25, 5071–5082 (2006).
Marina-Garcia, N. et al. Pannexin-1-mediated intracellular delivery of muramyl dipeptide induces caspase-1 activation via cryopyrin/NLRP3 independently of Nod2. J. Immunol. 180, 4050–4057 (2008).
Sharp, F. A. et al. Uptake of particulate vaccine adjuvants by dendritic cells activates the NALP3 inflammasome. Proc. Natl Acad. Sci. USA 106, 870–875 (2009).
Hornung, V. et al. Silica crystals and aluminum salts activate the NALP3 inflammasome through phagosomal destabilization. Nature Immunol. 9, 847–856 (2008).
Dostert, C. et al. Innate immune activation through Nalp3 inflammasome sensing of asbestos and silica. Science 320, 674–677 (2008). References 34–36 demonstrate activation of the NLRP3 inflammasome through environmental particulate stimuli and the adjuvant aluminium hydroxide (alum).
Parikh, H. et al. TXNIP regulates peripheral glucose metabolism in humans. PLoS Med. 4, e158 (2007).
Zhou, R., Tardivel, A., Thorens, B., Choi, I. & Tschopp, J. Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nature Immunol. 11, 136–140 (2009).
Menu, P. et al. ER stress activates the NLRP3 inflammasome via an UPR-independent pathway. Cell Death Dis. 3, e261 (2012).
Petrilli, V. et al. Activation of the NALP3 inflammasome is triggered by low intracellular potassium concentration. Cell Death Differ. 14, 1583–1589 (2007).
Cruz, C. M. et al. ATP activates a reactive oxygen species-dependent oxidative stress response and secretion of proinflammatory cytokines in macrophages. J. Biol. Chem. 282, 2871–2879 (2007).
Tassi, S. et al. Altered redox state of monocytes from cryopyrin-associated periodic syndromes causes accelerated IL-1β secretion. Proc. Natl Acad. Sci. USA 107, 9789–9794 (2010).
Bulua, A. C. et al. Mitochondrial reactive oxygen species promote production of proinflammatory cytokines and are elevated in TNFR1-associated periodic syndrome (TRAPS). J. Exp. Med. 208, 519–533 (2011). This study demonstrates a mitochondrial source for the ROS that are observed in TRAPS and suggests that enhanced pro-inflammatory cytokine production results from ROS-triggered MAPK signalling.
Pollock, J. D. et al. Mouse model of X-linked chronic granulomatous disease, an inherited defect in phagocyte superoxide production. Nature Genet. 9, 202–209 (1995).
van de Veerdonk, F. L. et al. Reactive oxygen species-independent activation of the IL-1β inflammasome in cells from patients with chronic granulomatous disease. Proc. Natl Acad. Sci. USA 107, 3030–3033 (2010).
Meissner, F. et al. Inflammasome activation in NADPH oxidase defective mononuclear phagocytes from patients with chronic granulomatous disease. Blood 116, 1570–1573 (2010).
Zhou, R., Yazdi, A. S., Menu, P. & Tschopp, J. A role for mitochondria in NLRP3 inflammasome activation. Nature 469, 221–225 (2010).
Saitoh, T. et al. Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1β production. Nature 456, 264–268 (2008).
Shimada, K. et al. Oxidized mitochondrial DNA activates the NLRP3 inflammasome during apoptosis. Immunity 36, 401–414 (2012).
Bauernfeind, F. et al. Cutting edge: reactive oxygen species inhibitors block priming, but not activation, of the NLRP3 inflammasome. J. Immunol. 187, 613–617 (2011). Reference 47 was the first study to suggest that dysfunctional mitochondria may activate the NLRP3 inflammasome, although references 45 and 50 suggest that ROS also have a role in this process through increasing the transcription of inflammasome component proteins.
Touitou, I. (ed.) Infevers: the registry of hereditary auto-inflammatory disorders mutations [online], (2012).
Duncan, J. A. et al. Cryopyrin/NALP3 binds ATP/dATP, is an ATPase, and requires ATP binding to mediate inflammatory signaling. Proc. Natl Acad. Sci. USA 104, 8041–8046 (2007).
McDermott, M. F. et al. Germline mutations in the extracellular domains of the 55 kDa TNF receptor, TNFR1, define a family of dominantly inherited autoinflammatory syndromes. Cell 97, 133–144 (1999).
Hull, K. M. et al. The TNF receptor-associated periodic syndrome (TRAPS): emerging concepts of an autoinflammatory disorder. Medicine 81, 349–368 (2002).
Kimberley, F. C., Lobito, A. A., Siegel, R. M. & Screaton, G. R. Falling into TRAPS — receptor misfolding in the TNF receptor 1-associated periodic fever syndrome. Arthritis Res. Ther. 9, 217 (2007).
Stojanov, S. et al. Clinical and functional characterisation of a novel TNFRSF1A c.605T>A/V173D cleavage site mutation associated with tumour necrosis factor receptor-associated periodic fever syndrome (TRAPS), cardiovascular complications and excellent response to etanercept treatment. Ann. Rheum. Dis. 67, 1292–1298 (2008).
Bulua A. C. et al. Efficacy of etanercept in the tumor necrosis factor receptor-associated periodic syndrome (TRAPS). Arthritis Rheum. 64, 908–913 (2011).
Lobito, A. A. et al. Abnormal disulfide-linked oligomerization results in ER retention and altered signaling by TNFR1 mutants in TNFR1-associated periodic fever syndrome (TRAPS). Blood 108, 1320–1327 (2006).
Todd, I. et al. Mutant tumor necrosis factor receptor associated with tumor necrosis factor receptor-associated periodic syndrome is altered antigenically and is retained within patients' leukocytes. Arthritis Rheum. 56, 2765–2773 (2007).
Rebelo, S. L. et al. Modeling of tumor necrosis factor receptor superfamily 1A mutants associated with tumor necrosis factor receptor-associated periodic syndrome indicates misfolding consistent with abnormal function. Arthritis Rheum. 54, 2674–2687 (2006).
Simon, A. et al. Concerted action of wild-type and mutant TNF receptors enhances inflammation in TNF receptor 1-associated periodic fever syndrome. Proc. Natl Acad. Sci. USA 107, 9801–9806 (2010). A study describing cooperativity between functional wild-type TNFR1 and TRAPS-associated mutant TNFR1 that leads to enhanced inflammatory responses through MAPK activation.
Dickie, L. J. et al. Involvement of X-box binding protein 1 and reactive oxygen species pathways in the pathogenesis of tumour necrosis factor receptor-associated periodic syndrome. Ann. Rheum. Dis. 7 Jun 2012 (doi:10.1136/annrheumdis-2011-201197).
The International FMF Consortium. Ancient missense mutations in a new member of the RoRet gene family are likely to cause familial Mediterranean fever. Cell 90, 797–807 (1997). This paper describes the cloning of the gene now known as MEFV , which encodes the protein pyrin, and the first discovery of the genetic cause of a familial fever syndrome.
Chae, J. J. et al. Isolation, genomic organization, and expression analysis of the mouse and rat homologs of MEFV, the gene for familial mediterranean fever. Mamm. Genome 11, 428–435 (2000).
Yu, J.-W. et al. Cryopyrin and pyrin activate caspase-1, but not NF-κB, via ASC oligomerization. Cell Death Differ. 13, 236–249 (2006).
Seshadri, S., Duncan, M. D., Hart, J. M., Gavrilin, M. A. & Wewers, M. D. Pyrin levels in human monocytes and monocyte-derived macrophages regulate IL-1β processing and release. J. Immunol. 179, 1274–1281 (2007).
Chae, J. J. et al. The B30.2 domain of pyrin, the familial Mediterranean fever protein, interacts directly with caspase-1 to modulate IL-1β production. Proc. Natl Acad. Sci. USA 103, 9982–9987 (2006).
Papin, S. et al. The SPRY domain of Pyrin, mutated in familial Mediterranean fever patients, interacts with inflammasome components and inhibits proIL-1β processing. Cell Death Differ. 14, 1457–1466 (2007).
Chae, J. J. et al. Gain-of-function Pyrin mutations induce NLRP3 protein-independent interleukin-1β activation and severe autoinflammation in mice. Immunity 34, 755–768 (2011). Using a knock-in mouse model, this study demonstrates that the human-specific B30.2 domain of pyrin that is the focus of mutations in FMF contributes to caspase 1- and IL-1β-dependent inflammation through a mechanism independent of NLRP3.
Booty, M. G. et al. Familial Mediterranean fever with a single MEFV mutation: where is the second hit? Arthritis Rheum. 60, 1851–1861 (2009).
Braun, J. & Sieper, J. Ankylosing spondylitis. Lancet 369, 1379–1390 (2007).
Ambarus, C., Yeremenko, N., Tak, P. P. & Baeten, D. Pathogenesis of spondyloarthritis: autoimmune or autoinflammatory? Curr. Opin. Rheumatol. 24, 351–358 (2012).
Smith, J. A., Marker-Hermann, E. & Colbert, R. A. Pathogenesis of ankylosing spondylitis: current concepts. Best Pract. Res. Clin. Rheumatol. 20, 571–591 (2006).
Turner, M. J., Delay, M. L., Bai, S., Klenk, E. & Colbert, R. A. HLA-B27 up-regulation causes accumulation of misfolded heavy chains and correlates with the magnitude of the unfolded protein response in transgenic rats: implications for the pathogenesis of spondylarthritis-like disease. Arthritis Rheum. 56, 215–223 (2007).
Turner, M. J. et al. HLA-B27 misfolding in transgenic rats is associated with activation of the unfolded protein response. J. Immunol. 175, 2438–2448 (2005).
Smith, J. A. et al. Endoplasmic reticulum stress and the unfolded protein response are linked to synergistic IFN-β induction via X-box binding protein 1. Eur. J. Immunol. 38, 1194–1203 (2008).
DeLay, M. L. et al. HLA-B27 misfolding and the unfolded protein response augment interleukin-23 production and are associated with Th17 activation in transgenic rats. Arthritis Rheum. 60, 2633–2643 (2009).
Goodall, J. C. et al. Endoplasmic reticulum stress-induced transcription factor, CHOP, is crucial for dendritic cell IL-23 expression. Proc. Natl Acad. Sci. USA 107, 17698–17703 (2010).
Burton, P. R. et al. Genome-wide association study of 14,000 cases of seven common diseases and 3,000 shared controls. Nature 447, 661–678 (2007).
Griffin, T. A. & Reed, A. M. Pathogenesis of myositis in children. Curr. Opin. Rheumatol. 19, 487–491 (2007).
Liao, A. P. et al. Interferon β is associated with type 1 interferon-inducible gene expression in dermatomyositis. Ann. Rheum. Dis. 70, 831–836 (2011).
Tournadre, A., Lenief, V., Eljaafari, A. & Miossec, P. Immature muscle precursors are a source of interferon-β in myositis: role of Toll-like receptor 3 activation and contribution to HLA class I up-regulation. Arthritis Rheum. 64, 533–541 (2012).
Nagaraju, K. et al. Conditional up-regulation of MHC class I in skeletal muscle leads to self-sustaining autoimmune myositis and myositis-specific autoantibodies. Proc. Natl Acad. Sci. USA 97, 9209–9214 (2000).
Todd, D. J., Lee, A. H. & Glimcher, L. H. The endoplasmic reticulum stress response in immunity and autoimmunity. Nature Rev. Immunol. 8, 663–674 (2008).
Kaser, A. et al. XBP1 links ER stress to intestinal inflammation and confers genetic risk for human inflammatory bowel disease. Cell 134, 743–756 (2008).
Iwakoshi, N. N. et al. Plasma cell differentiation and the unfolded protein response intersect at the transcription factor XBP-1. Nature Immunol. 4, 321–329 (2003).
Seifert, U. et al. Immunoproteasomes preserve protein homeostasis upon interferon-induced oxidative stress. Cell 142, 613–624 (2010).
Goldbach-Mansky, R. et al. Neonatal-onset multisystem inflammatory disease responsive to interleukin-1β inhibition. N. Engl. J. Med. 355, 581–592 (2006).
Hoffman, H. M. et al. Prevention of cold-associated acute inflammation in familial cold autoinflammatory syndrome by interleukin-1 receptor antagonist. Lancet 364, 1779–1785 (2004). References 88 and 89 describe the efficacy of using IL-1 blockade to treat the inflammatory syndromes linked to dominant NLRP3 mutations.
Sibley, C. H. et al. Sustained response and prevention of damage progression in patients with neonatal-onset multisystem inflammatory disease treated with anakinra: a cohort study to determine three- and five-year outcomes. Arthritis Rheum. 64, 2375–2386 (2012).
Dinarello, C. A. Interleukin-1 in the pathogenesis and treatment of inflammatory diseases. Blood 117, 3720–3732 (2011).
Schumacher, H. R. Jr et al. Rilonacept (Interleukin-1 Trap) for prevention of gout flares during initiation of uric acid-lowering therapy: results of the presurge-1 trial. Arthritis Care Res. 1 May 2012 (doi:10.1002/acr.21690).
So, A. et al. Canakinumab for the treatment of acute flares in difficult-to-treat gouty arthritis: results of a multicenter, phase II, dose-ranging study. Arthritis Rheum. 62, 3064–3076 (2010).
Larsen, C. M. et al. Interleukin-1-receptor antagonist in type 2 diabetes mellitus. N. Engl. J. Med. 356, 1517–1526 (2007).
Larsen, C. M. et al. Sustained effects of interleukin-1 receptor antagonist treatment in type 2 diabetes. Diabetes Care 32, 1663–1668 (2009).
Pascual, V., Allantaz, F., Arce, E., Punaro, M. & Banchereau, J. Role of interleukin-1 (IL-1) in the pathogenesis of systemic onset juvenile idiopathic arthritis and clinical response to IL-1 blockade. J. Exp. Med. 201, 1479–1486 (2005).
Lequerre, T. et al. Interleukin-1 receptor antagonist (anakinra) treatment in patients with systemic-onset juvenile idiopathic arthritis or adult onset Still disease: preliminary experience in France. Ann. Rheum. Dis. 67, 302–308 (2008).
Drenth, J. P. et al. Mutations in the gene encoding mevalonate kinase cause hyper-IgD and periodic fever syndrome. Nature Genet. 22, 178 (1999).
Feldmann, J. et al. Chronic infantile neurological cutaneous and articular syndrome is caused by mutations in CIAS1, a gene highly expressed in polymorphonuclear cells and chondrocytes. Am. J. Hum. Genet. 71, 198–203 (2002).
Aksentijevich, I. et al. The clinical continuum of cryopyrinopathies: novel CIAS1 mutations in North American patients and a new cryopyrin model. Arthritis Rheum. 56, 1273–1285 (2007). References 99 and 100 describe the inflammatory syndromes linked to dominant mutations in the NLRP3 gene.
Rose, C., Martin, T. & Wouters, C. Blau syndrome revisited. Curr. Opin. Rheumatol. 23, 411–418 (2011).
Miceli-Richard, C. et al. CARD15 mutations in Blau syndrome. Nature Genet. 29, 19–20 (2001).
Shoham, N. G. et al. Pyrin binds the PSTPIP1/CD2BP1 protein, defining familial Mediterranean fever and PAPA syndrome as disorders in the same pathway. Proc. Natl Acad. Sci. USA 100, 13501–13506 (2003).
Aksentijevich, I. et al. An autoinflammatory disease with deficiency of the interleukin-1-receptor antagonist. N. Engl. J. Med. 360, 2426–2437 (2009). A report describing the familial syndrome DIRA, which is linked to a deficiency of IL-1 receptor antagonist and leads to severe systemic autoinflammation. This study confirms a crucial role of IL-1 receptor antagonist in dampening the hyperproduction of IL-1β during the inflammatory response in human disease.
Marrakchi, S. et al. Interleukin-36-receptor antagonist deficiency and generalized pustular psoriasis. N. Engl. J. Med. 365, 620–628 (2011).
Acknowledgements
We would like to thank M. Pelletier, I. Aksentijevich and R. Goldbach-Mansky for critical reading and helpful comments on the manuscript. This work was supported by the Intramural Research Program of the US National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Gout
-
An autoinflammatory disease triggered by crystalline uric acid and characterized by episodic flares of arthritis that can progress to destructive joint damage.
- Spondyloarthropathies
-
A group of immune-mediated inflammatory disorders that affect the vertebral column. Spondyloarthropathies also frequently affect the entheses (the areas of tendon and ligament insertion into bone).
- ER stress
-
Stress caused by the perturbation of endoplasmic reticulum (ER) functions that are necessary for cellular homeostasis. The unfolded protein response is the collective outcome of ER stress and must be tightly regulated, as otherwise it is directly linked to the pathogenesis of several diseases.
- Inflammasome
-
A large multiprotein complex usually comprising a NOD-like receptor (NLR), the adaptor protein ASC and pro-caspase 1. The assembly of the inflammasome leads to the activation of caspase 1, which cleaves pro-interleukin-1β (pro-IL-1β) and pro-IL-18 to generate the active pro-inflammatory cytokines.
- Immunoproteasome
-
The standard proteasome is composed of 14 α- and β-subunits, of which three (β1, β2 and β5) are involved in peptide-bond cleavage. Interferon-γ induces the expression of the immunosubunits β1i, β2i and β5i, which can replace the catalytic subunits of the standard proteasome to generate the immunoproteasome, which has distinct cleavage-site preferences.
- NADPH oxidases
-
Plasma membrane- and phagosomal membrane-bound enzyme complexes that transfer electrons from NADPH to molecular oxygen, promoting the generation of the reactive oxygen species superoxide.
- Mitophagy
-
Selective removal of mitochondria under conditions of nutrient starvation or mitochondrial stress.
- Autophagy
-
An evolutionarily conserved process in which acidic double-membrane vacuoles sequester intracellular contents (such as damaged organelles and macromolecules) and target them for degradation through fusion to secondary lysosomes.
- Colchicine
-
An inhibitor of microtubule polymerization that has anti-inflammatory properties, possibly through blockade of neutrophil chemotaxis. Colchicine is effective in treating acute attacks of gout and protecting against recurrent flares. It is also an effective treatment for familial Mediterranean fever, but not for other genetic autoinflammatory syndromes.
Rights and permissions
About this article
Cite this article
Park, H., Bourla, A., Kastner, D. et al. Lighting the fires within: the cell biology of autoinflammatory diseases. Nat Rev Immunol 12, 570–580 (2012). https://doi.org/10.1038/nri3261
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3261
This article is cited by
-
RBP–RNA interactions in the control of autoimmunity and autoinflammation
Cell Research (2023)
-
Paradigm Shift in Diagnosis and Targeted Therapy in Recurrent Pericarditis
Current Cardiology Reports (2023)
-
Air pollution exposure and auto-inflammatory and autoimmune diseases of the musculoskeletal system: a review of epidemiologic and mechanistic evidence
Environmental Geochemistry and Health (2023)
-
Inflammatory Diseases of the Eye, Bowel, and Bone in Children
Indian Journal of Pediatrics (2023)
-
Peripheral T Cell Populations are Differentially Affected in Familial Mediterranean Fever, Chronic Granulomatous Disease, and Gout
Journal of Clinical Immunology (2023)