Abstract
Circulatory–respiratory or brain tests are widely accepted for definition and determination of death, but have several controversial issues. Both determinations have been stimulated by organ donation, but must be valid independently of this process. Current controversies in brain death include whether the definition is conceptually coherent, whether the whole-brain or brainstem criterion is correct, whether one neurological examination or two should be required, and when to conduct the examination following therapeutic hypothermia. Controversies about the circulatory determination of death include the minimum duration of asystole that is sufficient for death to be declared, and whether the distinction between permanent and irreversible cessation of circulatory functioning is important. In addition, the goal of organ donation raises issues such as the optimal way to time and conduct the request conversation with family members of the patient, and whether the Dead Donor Rule should be abandoned.
Key Points
-
Death can be determined by circulatory–respiratory or brain tests
-
All brain death practices use the whole-brain or brainstem criterion of death
-
Controversies about brain death focus on the coherence of the concept, the extent of necessary neuronal damage, and the tests required to prove irreversibility
-
Controversies about circulatory death focus on the required duration of asystole before death can be declared, and whether permanent or irreversible cessation should be the standard
-
The option of organ donation should be presented to families of deceased patients, but the optimal timing of and approach to the donation request remain unclear
Similar content being viewed by others
Introduction
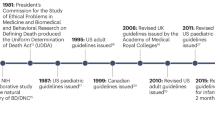
Contemporary controversies about brain and circulatory determinations of death in the intensive care unit (ICU) can be informed by studying the historical impact of life-sustaining therapies (LSTs) on the definition of human death.1 Prior to development of LSTs—particularly tracheal positive-pressure ventilation (TPPV) and cardiopulmonary resuscitation (CPR)—in the 1950s, death was a unitary phenomenon. When illness or injury caused cessation of one of the three 'vital' functions critical to life—circulation, respiration and brain function—both of the others ceased within minutes. This interdependence of vital functions, and the inability of physicians to intervene to restore or support them, made the definition of death straightforward, although its determination was incorrect at times.2
The development of LSTs abolished the unitary nature of death, as the new technologies could support or restore individual vital functions while others remained absent.3 CPR could reverse cardiac arrest and restore circulation, vasopressors could maintain blood pressure in shock, and TPPV could support ventilation in apnoea. These LSTs were valuable interventions when they led to restoration of normal function, but were harmful when they restored circulation and supported ventilation in patients in whom all brain functions had ceased irreversibly. The concept of a brain-based determination of death was proposed, which asserted that the irreversible loss of all clinical brain functions was sufficient grounds to declare death, irrespective of continued technological support of ventilation and circulation.4
Two emerging medical needs stimulated the development of standards of death determination in patients receiving LST: multi-organ transplantation, and the desire to discontinue LST in cases of hopeless brain damage.1 Organ transplantation technology became successful in the 1960s as patients who were 'brain dead' represented ideal multi-organ donors, because their intact circulation preserved organs. The desire to promote organ donation for transplantation encouraged initial acceptance of the concept of brain death.5,6 Additionally, physicians saw the need to discontinue meaningless LSTs for those unfortunate patients who had been rescued by CPR and TPPV but whose brain had been destroyed by lack of blood flow. Prevailing laws at the time prohibited physicians from stopping LSTs before the patient was declared dead.1
The concept that brain function was necessary for life and that its irreversible cessation was sufficient for death arose independently in several countries. In the late 1950s, French neurologists reported cases of complete cessation of brain function that they called coma dépassé (irretrievable coma).7,8 American scholars systematized the criteria for, and popularized, the misleading term 'brain death' in the 1960s,9 and British physicians propounded the concept of brainstem death in the 1970s and 1980s.10,11 By the turn of the 21st century, over 80 countries in the developed and developing world had accepted a brain-based determination of death, and most developed countries enacted this medical practice into public laws.12 Despite the arguments of scholars who reject brain death on scientific, conceptual or religious grounds, the practice of brain-based death determination continues to increase in prevalence throughout the world.13
This Review examines what we mean by the term 'death', and discusses the brain criteria and circulatory–respiratory criteria that can be used to define death. Controversies surrounding the application of these criteria are discussed, and ethical issues arising in the context of organ donation are considered.
A biophilosophical analysis of death
Initially, the brain-based determination of death was accepted as a medical and legal standard in the absence of a rigorous philosophical justification. Throughout the 1970s, many states in the USA incorporated brain death determinations into their death statutes because of its acceptance by physicians, despite the lack of a compelling rationale.14 Early attempts to justify the equivalence of brain death and human death were provided by Korein15 and Pallis,11 as well as a US President's Commission16 and my own research group.3,17 We have refined our original analysis over the past three decades.18,19,20 Most scholars in this field, including some who disagree with our conclusions,21,22,23 regard our approach as the 'standard' analysis of death. A vigorous debate over the definition of and criterion for death continues within academia but, tellingly, this scholarly dispute has had no effect on the medical practice of bedside death determination.
The analysis of death features four sequential stages: first, the context of death states the conditions for the argument, including the nature of death and words we use to describe it; second, the definition of death makes explicit what we mean in our ordinary use of the word 'death'; third, the criterion of death states the general measureable standard that is both necessary and sufficient for death, and that can be included in a death statute; and fourth, the tests of death are devised by physicians to show that the criterion of death has been fulfilled.24 Even scholars who maintain that defining death could be an impossible task agree that conceptual clarity about the nature of death must precede creation of tests for its measurement.25,26
The context conditions include the view that 'death' is a nontechnical word, the meaning of which has been rendered ambiguous by technology. The goal of philosophical analysis is to make explicit the consensual meaning that is implicit in our common use of the word, not to contrive its redefinition. Death, like life, can be considered a fundamentally biological phenomenon that is not primarily a social contrivance or a religious or cultural custom, although many cultures have rich customs, beliefs, practices and laws surrounding death and dying. 'Alive' and 'dead' are the only two possible states of an organism, so the transition from one state to the other is necessarily instantaneous, although determination of this precise moment may only be possible in retrospect. Death is the event that separates the process of dying from the process of bodily disintegration. The transition from alive to dead is unidirectional because death is irreversible.19,24
The most precise definition of death in our technological era is the cessation of the critical functions of the organism as a whole.19 The organism as a whole refers not to the whole organism (the sum of its parts), but to those behaviours that are greater than the sum of its parts and cannot be reduced to individual parts of the organism.27 The phenomenon of consciousness is the most exquisite example of a function of the organism as a whole. These behaviours emerge spontaneously from the normally functioning assembled parts of the organism.28 The concept of the organism as a whole emphasizes the unity and coherent integrity of the organism and the interrelatedness of its parts that, operating in unison, contribute to the overall health of the organism.29 This concept also posits that life in part of an organism is distinct from life of the organism itself. It is possible, therefore, to technologically maintain the life of parts of an organism—such as cells, tissues and organs—despite the cessation of functioning of the organism as a whole.19,24 This is the unique condition of the brain-dead patient: the organism as a whole has ceased to function, although many parts of the organism obviously remain alive by technological support.29
The dual criteria of death present in many death statutes (including those of the USA) are the irreversible cessation of clinical functioning of the brain, and the irreversible cessation of circulatory and respiratory functioning. Physicians apply the circulatory–respiratory death criterion in most patients who are not receiving TPPV. Physicians reserve the brain death criterion for the few ICU patients with apnoea on TPPV, to whom the circulatory–respiratory criterion cannot be applied. The two criteria of death are not independent. Although few data inform the discussion, the circulatory–respiratory criterion is valid because it leads to fulfilment of the brain criterion: circulatory arrest induces a rapid process in which neurons are progressively destroyed by hypoxaemia and ischaemia.3
The brain criterion of death
The brain criterion of death (brain death) is necessary and sufficient for death because the brain enables the critical functions of the organism as a whole, although not all functions of the organism as a whole.30 Debate continues over the extent of the brain functions that must cease in order to satisfy the definition. Three separate brain death criteria, known as the whole-brain, brainstem and higher-brain formulations, have been defended.31 The accepted testing standards are shown in Boxes 1, 2 and 3.
The whole-brain formulation
Death standards in the USA and many other countries employ the whole-brain criterion, which requires that all clinical functions of the brain, including those served by the cerebral hemispheres, diencephalon and brainstem, must have ceased.32,33,34,35 Apnoea, which represents medullary failure, must be present, and all brainstem reflexes such as pupillary light reflexes, corneal reflexes, vestibulo-ocular reflexes, and gag and cough reflexes must be absent, reflecting failure of the midbrain, pons and medulla. Complete unresponsiveness and the deepest possible coma reflect failure of the reticular system in the brainstem and its projections to the thalamus and forebrain. Cerebral hemispheric function is difficult to assess at the bedside, however, once brainstem functioning has ceased. Only clinical brain functions—which, unlike activity of individual neurons, are assessed by bedside neurological examination—must be shown to be absent.16
The pathogenesis of brain death following brain illness and injury accounts for the emphasis on assessment of brainstem functioning in the determination of brain death. Diffuse hypoxic–ischaemic neuronal insults during cardiac arrest, massive traumatic brain injury, intracranial haemorrhage, and meningitis each produce marked increases in intracranial pressure, leading to syndromes of transtentorial cerebral herniation, which cause diffuse infarction of the brainstem.36,37 Once intracranial pressure exceeds mean arterial blood pressure, intracranial circulation ceases, leading to secondary diffuse infarction of cerebral hemispheric neurons that survived the initial brain insult. The whole-brain formulation, therefore, contains a fail-safe mechanism that ensures that the definition encompasses global neuronal damage and loss of all clinical functions.38
The brainstem formulation
In the UK, Pallis championed the concept of brainstem death, pointing out that the accepted clinical tests all assessed brainstem function, that the brainstem was responsible for consciousness, breathing and circulatory regulation, and that it conducted nearly all throughput to and from the brain.11 The whole-brain and brainstem criteria of death are essentially identical, diverging only in the exceedingly rare instance of a massive brainstem infarct or haemorrhage that destroys the midbrain, pons and medulla but spares the cerebral hemispheres.39,40 In such cases, a patient would be diagnosed as dead according to the brainstem criterion but might not be declared dead under the whole-brain criterion. In a modification of this formulation, Machado emphasized the brainstem basis for brain death as the irreversible loss of both dimensions of human consciousness: wakefulness and awareness.41
The higher-brain formulation
The higher-brain formulation was proposed in the 1970s by scholars who argued that because consciousness and cognition are the unique characteristics of humans, irreversible absence of these functions constituted death. According to this formulation, patients in permanent vegetative states with spontaneous breathing should be declared dead, despite the fact that universally they are regarded as alive. The higher-brain formulation fails the context condition of making explicit the ordinary meaning of 'death'. Rather, this formulation is a radical redefinition of death and one that no medical society or country has adopted.18
Controversies about brain death
Two lines of criticism
Critics of the brain criterion of death fall into two camps: those who argue that the concept of brain death should be rejected, and those who accept brain death but disagree with prevailing standards or practices. Most prominent among scholars who argue that the brain criterion of death should be abandoned are Shewmon,21,26,30,42 Miller22 and Truog.22,43
Shewmon agreed with the definition of death as the cessation of critical functions of the organism as a whole, but argued that only the circulatory criterion—and not the brain criterion—satisfies the definition. He posited that the role of the spinal cord in integrating some functions of the organism as a whole undermined the justification for a brain-based concept of death.30 Shewmon reported a series of patients diagnosed with brain death whose circulation and visceral organ function were maintained for many months or longer, which showed both that some functions of the organism as a whole persisted after brain destruction and that prolonged 'survival' was counterintuitive to a concept of death.44 He emphasized the case reported by Repertinger and colleagues of a 4-year-old child who was diagnosed as brain dead after bacterial meningitis and was then supported for 19 years on a ventilator.45 Shewmon argued that the prolonged duration of successful support in this case was inconsistent with the concept of death.
Miller and Truog claimed that use of a brain criterion of death is an anachronism and a legal fiction that was created decades ago to achieve the goals of organ donation and withdrawal of LSTs, which in contemporary medical practice can be more successfully accomplished by other means.22 They argued that although we all know that brain-dead patients are not dead, we accept the legal fiction to permit organ donation.
Are two examinations required?
A current practice controversy concerns whether physicians must conduct two sequential examinations to determine brain death, or whether a single examination is sufficient. Prior practice guidelines throughout the world mandated two sequential examinations separated by a time interval that varied as a function of the patient's age, the nature of the brain injury, and the presence of concomitant ancillary tests. A large study of adults with brain death showed that the second examination added nothing to the first, delayed the declaration of death, and reduced organ donation.46 As a result, the American Academy of Neurology (AAN) revised its long-standing guideline on adult brain death to require only a single examination.32 One critic questioned the methodology, wondering whether some patients had been excluded from the study after the first examination,47 but a later smaller series confirmed the findings.48 The change to a single examination has not been uniformly accepted. One prominent guideline for the determination of brain death in infants and children, published after the AAN guideline update, required two examinations,33 as do some death statutes in the USA. How many future guidelines will adopt the single-examination rule remains unclear.
Ancillary tests
Another practice controversy surrounds the use of ancillary tests to confirm brain death, such as imaging studies that show absent intracranial blood flow, or electrical recording studies that show absent EEG and evoked potentials.49 These tests have been performed since the formulation of the definition of brain death. Wijdicks recently argued that because brain death is a clinical diagnosis and ancillary tests can be inaccurate and misleading, such tests should not be used to support determination of brain death.50 However, ancillary tests remain useful in cases in which clinical tests cannot be performed fully, and to support the diagnosis when inexperienced or careless examiners have performed the brain death examination improperly,38 particularly in light of the known pitfalls of the examination such as the confounding presence of pre-existing neurological deficits and the unexpected presence of potentially reversible metabolic or toxic factors.51
The need for standardization
A survey of brain death protocols in leading neurology departments in the USA disclosed a shocking lack of standardization,52 despite availability of the accepted AAN guidelines for over a decade.32 In one institution, apnoea testing (Box 3), which has been a prerequisite in every published battery of brain death tests since the 1970s was, inexplicably, not required. Other studies have disclosed the inadequacy of documentation of brain death testing.53 The need for greater uniformity in performing and recording the tests is obvious,54 and some have advocated for a national standard.55 Many institutions have developed brain death checklists to ensure that examiners test all appropriate functions and adequately document the test results.32
Confounding effects of induced hypothermia
False-positive brain death examinations result from examination inadequacies. Webb and Samuels reported a well-documented case of spontaneous improvement after brain death declaration in a patient who had received therapeutic hypothermia to protect the brain following cardiac arrest.56 He was rewarmed according to protocol, but just as organ donation was beginning 24 h later, he began to breathe and show corneal and cough reflexes. He improved no further and developed asystole.
This case provoked debate over the mechanism of spontaneous improvement. Some commentators argued reasonably that hypothermia may have slowed the metabolism of depressant or neuromuscular-blocking drugs administered earlier, which continued to exert effects for hours after rewarming.57 Because of this case, some hospitals have altered their therapeutic hypothermia protocols to require a delay of 24–72 h between rewarming and determination of brain death, to allow resolution of a potentially reversible confounding toxic–metabolic encephalopathy. Brain death guidelines require reversible causes of brain dysfunction, including therapeutically induced hypothermia, to be excluded.32
Opposition from family members
Family member opposition to brain death usually results from emotional inability to accept the finality and hopelessness of the diagnosis, particularly when the patient is a previously healthy young person with a traumatic brain injury. For many family members, the counterintuitive nature of death declaration in a person with intact heartbeat and circulation and the desperate hope for recovery makes acceptance of the physician's pronouncement difficult.58,59 Medical and nursing personnel may compound the problem by using misleading terms, such as 'life support' to refer to TPPV, and 'irreversible coma'.60 Thoughtful guidelines have been developed to negotiate and compassionately resolve this conflict.61,62 Inviting family members to observe the patient's complete unresponsiveness during the brain death examination has been advocated as a solution,63 but is difficult to manage in practice.64
Opposition on religious grounds
Religious opposition to brain death is an uncommon issue in most communities.13 Protestantism uniformly accepts brain death as human death. Roman Catholicism endorsed the practice in a statement made by Pope John Paul II in 2000,65 which was reaffirmed in 2006 by the Pontifical Academy of Sciences.66 The situation in Judaism is more complex: Reform, Conservative and many Orthodox rabbis accept the concept of brain death67 but ultra-Orthodox rabbis do not.68 Islam is similarly complex, with general acceptance of brain death but variation among denominations and among Islamic countries.69 Hindu India accepts brain death,70 as does Shinto-Buddhist Japan with some conditions,71 but China does not. Family members may erroneously cite their religion's objection to extubation or organ donation because they are unaware of the actual teachings. Guidelines are available for managing cases of religious exception to brain death.72
The circulatory criterion of death
Determination of death using the circulatory–respiratory criterion is straightforward for most deaths in which respiratory and circulatory support is not administered (Box 4). Use of this determination in the ICU has become controversial, however, owing to increasing use of protocols for organ donation after the circulatory determination of death (DCDD), formerly called non-heart-beating organ donation or donation after cardiac death.73 As with organ donation after brain death, the advent of DCDD has stimulated greater medical exactitude in death determination.
The contentious issue in these protocols is determination of the necessary duration of circulatory cessation. In a 'controlled' DCDD protocol, family members of a severely brain-damaged (but not brain-dead) ventilator-dependent ICU patient decide to withdraw LSTs to allow the patient to die, and request that the patient becomes an organ donor after death, often to follow the patient's wishes. The DCDD protocol coordinates withdrawal of LST with the rapid removal of organs following death determination.74 In an 'uncontrolled' DCDD protocol, practiced in some European countries, patients with unanticipated cardiac arrest who cannot be resuscitated serve as organ donors.75
Identification of the moment of death is necessary because the Dead Donor Rule prohibits transplantation of vital organs until after the donor has been declared dead, so that the donation does not kill the donor.76 Transplant surgeons wish to remove the organs as quickly as possible after death to minimize warm ischaemic time and, thereby, yield healthier organs for transplantation.
Agreement on the exact moment of death remains surprisingly controversial. Since the earliest practice of DCDD, some critics have claimed that the organ donor was not truly dead at the moment death was declared.77 In the pioneering University of Pittsburgh protocol of the early 1990s, donor patients were declared dead after 2 min of asystole and apnoea.78 Critics argued that because some patients with 'do not resuscitate' (DNR) orders nevertheless remained able to be resuscitated at the moment they were declared dead, they did not satisfy the statute of death, which requires the irreversible cessation of circulation.79
The duration of asystole that is required before death can be determined varies widely among protocols.35 Medical societies and expert groups have issued guidelines stipulating for how long the donor patient must have apnoea and asystole to be declared dead. The Ethics Committee of the Society of Critical Care Medicine80 and the National Conference on Donation after Cardiac Death81 both require 2–5 min, whereas the US Institute of Medicine82 and the Canadian Council for Donation and Transplantation83 require 5 min, and some European countries stipulate 10 min.84 In an experimental neonatal DCDD heart transplantation protocol at Denver Children's Hospital, the hospital ethics committee controversially approved the ad hoc reduction of the required interval of asystole from 2 min to 75 s.85
Mechanical versus electrical systole
Attendees at a large US DCDD consensus conference in 2005 agreed that asystole in the context of circulatory death determination requires mechanical but not electrical asystole.81 Because the circulatory criterion addresses blood flow, the absence of circulation—not of cardiac electrical activity—is the relevant criterion. Thus, pulseless electrical activity (PEA) during cardiac arrest86 is fully compatible with the circulatory criterion of death. Electrical asystole, although sufficient to warrant declaration of death, is unnecessary.81
Autoresuscitation
Autoresuscitation initially referred to the spontaneous return of heartbeat after asystole. Early reports did not distinguish between autoresuscitation leading to PEA and autoresuscitation leading to restored circulation.87 Later, it became clear that because the death standard was cessation of circulation and not of cardiac electrical activity, true autoresuscitation required the spontaneous return of circulation.88
The incidence of autoresuscitation is an empirical question, and current data are limited. A recent series of 73 cases of controlled DCDD showed no cases of autoresuscitation.89 In the largest review of all published cases of autoresuscitation, Hornby and colleagues found cases of PEA reported up to 65 s after asystole but no cases of restored circulation after withdrawing LSTs. By contrast, they found many cases of autoresuscitation leading to restored circulation after failed CPR, up to an interval of 7 min after asystole began.88 Autoresuscitation can occur many minutes following unsuccessful CPR owing to delayed effects of administered medications or to the 'auto-positive end-expiratory pressure' phenomenon of reversible PEA resulting from dynamic lung hyperinflation.90
These findings support the prevailing practice of death determination in controlled DCDD, but the occurrence of autoresuscitation after unsuccessful CPR reduces the accuracy of death determination in uncontrolled DCDD. The requirement to wait until after the period in which autoresuscitation can occur could jeopardize organ health in uncontrolled DCDD protocols. Autoresuscitation, therefore, represents a major impediment to uncontrolled DCDD but only a minor one to controlled DCDD.91
Circulatory-criterion controversies
Permanent versus irreversible cessation
Distinguishing between permanent and irreversible cessation of circulation helps to answer the question of whether, in controlled DCDD, the patient is dead at the moment of death declaration. Permanent cessation of a function means that the lost function will not be restored because it will neither recover spontaneously nor will medical attempts be made to restore it. By contrast, irreversible cessation of a function means that the lost function cannot possibly be restored even if medical attempts are made. Many death statutes use the term 'irreversible cessation of circulation', although medical standards for death declaration have always relied on permanent cessation of circulation.92
Following cessation of circulation and breathing in the context of controlled DCDD, the patient can be declared dead after the period during which autoresuscitation could occur, as the DNR order precludes resuscitative attempts. As for terminally ill patients with DNR orders who are not organ donors, once circulation and respiration have ceased permanently, the declaration of death need not be delayed until circulatory–respiratory failure can be shown to be irreversible. Although consensus has not been reached about the importance of this distinction,93,94 an expert panel on death determination recruited by the US department that funds experimental organ transplantation protocols endorsed the distinction as the conceptual basis for prevailing practices of declaring death in DCDD donors.95 The unresolved empirical issue is determination of the exact time during which autoresuscitation can occur. Confidence limits will improve as more cases are studied.
Organ preservation technologies
Another controversy related to the circulatory criterion of death surrounds what types of organ preservation interventions can be performed on the DCDD donor following death determination. Some transplant surgeons have used extracorporeal membrane oxygenation (ECMO) on DCDD donors following death determination to preserve organ function prior to donation.96,97 The expert panel mentioned above criticized this practice on the grounds that, by re-establishing blood flow to the brain, ECMO retroactively negates the prior death determination because the cessation of circulation is not permanent, and ECMO circulation might allow the brain to continue to function.93 Other transplant surgeons amended the ECMO protocol to include inflation of a balloon catheter in the thoracic aorta to restrict the ECMO perfusion circuit to abdominal–pelvic organs and block perfusion to the thorax and head.98 The expert panel acknowledged that this modification eliminated the problem of retroactive invalidation of the prior death determination but criticized it on ethical and legal grounds.95
Recent analyses of death determination
The most recent comprehensive re-examination of the definition of death was published in 2008 by the US President's Council on Bioethics.99 The Council reaffirmed that brain-dead patients were truly dead, and that public laws supporting the practice of brain death determination should be maintained. However, the Council rejected the standard rationale for equating brain death with human death—namely, the loss of the organism's capacity for integration—and replaced it with cessation of the capacity to perform the essential work of a living organism. A prominent critic of the integration rationale subsequently argued that the Council's new explanation contained the same flaw as the one it replaced.42 The Council also endorsed DCDD and the importance of maintaining the Dead Donor Rule.
An international research group supported by the Canadian Blood Services is currently working in collaboration with the WHO to produce international guidelines on the determination of death. The group met in Geneva in 2010 and Montreal in 2012. To date, they have produced a framework for discussing medical and scientific aspects of death determination, as well as clinical standards, definitions and a research agenda. Next, they will grade evidence to confirm the clinical standards and to develop strategies to implement the guidelines globally.100
Death and organ donation
ICU patients declared dead by either brain or circulatory criteria are potential organ donors. Because the demand for organs to transplant far exceeds the supply of donor organs,101 many countries have developed programmes that aim to increase the donor organ supply. Governmental and professional organizations support the laudable goal of increasing organ donation because of its obvious life-saving benefits to the organ recipients. Donation also confers benefits to family members of the deceased patient by giving profound meaning to a tragic death, through knowledge that the loved one has contributed to the life and health of others.102
Several scholars have expressed concern that the way in which the organ donation programmes are implemented may unintentionally harm end-of-life care.103,104,105 Once organ donation becomes a goal, medical and nursing staff might consider—or be perceived to consider—the dying patient as a means to the end of organ donation.106 This problem is more serious in DCDD than in organ donation after brain death, because organ donation discussions with family members in DCDD always occur before death is declared. Some ICU physicians expressed concern that awareness that their patients could serve as organ donors may subconsciously influence their treatment of patients while they are alive.107 Two recent surveys of the opinions of critical care physicians and nurses about DCDD have confirmed the existence of these concerns.108,109
In the USA, the regulations of the Centers for Medicare and Medicaid Services require hospitals to notify the local organ procurement organization (OPO) “of individuals whose death is imminent or who have died in the hospital,” to allow a 'designated requestor' (usually an OPO representative) to present the option of donation to the family and obtain consent.105 Several studies have shown that OPO representatives, because of their knowledge and training, are more successful than are physicians and nurses in securing family consent.110 Furthermore, most OPOs lack a policy whereby physicians can obtain consent.111
Some OPOs adopt a 'presumptive approach strategy' to organ donation that frames the consent discussion in a leading way.112 Defenders of this consent technique cite the concept of dual advocacy, in which the interests of the donor family and of patients awaiting transplantation are simultaneously valued because of the presumption that “given the opportunity, most people will chose to help others.”113 One instance in which the dual advocacy concept becomes strained is in the decision to maintain TPPV and other LSTs in an unsalvageable patient with massive brain injury solely for the purpose of declaring brain death to permit organ donation.114
Another controversy in deceased organ donation is whether the Dead Donor Rule is necessary. This rule is an informal but widely accepted requirement that the organ donor must first be declared dead so that donation does not cause the death of the donor.76 The organ transplantation community uniformly supports this rule as a condition necessary to maintain public confidence in physicians and the transplantation enterprise. Miller and Truog22 called for abandonment of the rule, however, arguing that the present or prior consent of the dying patient, who is beyond harm, or of the lawful surrogate validly consenting on behalf of the patient, should be sufficient to allow donation before death is declared. The benefits and drawbacks of adopting this approach have been debated,93,115 but most scholars endorse retention of the Dead Donor Rule.
Conclusions and future directions
Death determination in the ICU using circulatory–respiratory or brain tests are well-established practices with rigorous conceptual foundations, and are not contrived to facilitate organ donation. Controversies about practical issues of death determination can be resolved by reasoned arguments citing physiology and public policy, leading to consensus among practitioners. International consensus remains an important goal, but is challenged by differences in culture, religion, accepted practices, and laws. Conceptual disagreements over the definition of death will not be resolved in the foreseeable future, but might not impede development of consensus on practices of death determination. Controversies about organ donation may be mitigated by optimizing conversations among family members, physicians and organ donation personnel to reassure family members and physicians that the dual goals of excellent end-of-life care and organ donation need not conflict.
Review criteria
The PubMed MEDLINE database was searched for articles published between 2000 and 2012, using the keywords: “definition of death”, “brain death”, “donation after cardiac death”, and “ethics/organ transplantation”. Full-length articles published in or translated into English were retrieved. Reference lists of review articles, and selected articles and books from the author's collection, were consulted.
References
Giacomini, M. A change of heart and a change of mind? Technology and the redefinition of death in 1968. Soc. Sci. Med. 44, 1465–1482 (1997).
Powner, D. J., Ackerman, B. M. & Grenvik, A. Medical diagnosis of death in adults: historical contributions to current controversies. Lancet 348, 1219–1223 (1996).
Bernat, J. L., Culver, C. M. & Gert, B. On the definition and criterion of death. Ann. Intern. Med. 94, 389–394 (1981).
Wijdicks, E. F. The neurologist and Harvard criteria for brain death. Neurology 61, 970–976 (2003).
Belkin, G. S. Brain death and the historical understanding of bioethics. J. Hist. Med. Allied Sci. 58, 325–361 (2003).
Pernick, M. S. in The Definition of Death: Contemporary Controversies (eds Youngner, S. J. et al.) 3–33 (Johns Hopkins University Press, Baltimore, 1999).
Mollaret, P. & Goulon, M. Le coma dépassé (mémoire préliminaire) [French]. Rev. Neurol. (Paris) 101, 3–15 (1959).
Wertheimer, P., Jouvet, M. & Descotes, J. A propos du diagnostic de la morte du système nerveux dans les comas avec arrêt respiratoire traités par respiration artificielle [French]. Presse Méd. 67, 87–88 (1959).
[No authors listed]. A definition of irreversible coma. Report of the Ad Hoc Committee of the Harvard Medical School to Examine the Definition of Brain Death. JAMA 205, 337–340 (1968).
[No authors listed] Diagnosis of brain death. Lancet 2, 1069–1070 (1976).
Pallis, C. ABC of Brainstem Death (British Medical Journal Publishers, London, 1983).
Wijdicks, E. F. Brain death worldwide: accepted fact but no global consensus in diagnostic criteria. Neurology 58, 20–25 (2002).
Wijdicks, E. F. Brain Death 2nd edn (Oxford University Press, New York, 2011).
Beresford, H. R. Brain death. Neurol. Clin. 17, 295–306 (1999).
Korein, J. The problem of brain death: development and history. Ann. NY Acad. Sci. 315, 19–38 (1978).
President's Commission for the Study of Ethical Problems in Medicine and Biomedical and Behavioral Research. Defining Death: Medical, Ethical, and Legal Issues in the Determination of Death (US Government Printing Office, Washington, DC, 1981).
Bernat, J. L., Culver, C. M. & Gert, B. Defining death in theory and practice. Hastings Cent. Rep. 12 (1), 5–8 (1982).
Bernat, J. L. A defense of the whole-brain concept of death. Hastings Cent. Rep. 28 (2), 14–23 (1998).
Bernat, J. L. The biophilosophical basis of whole-brain death. Soc. Philos. Policy 19, 324–342 (2002).
Bernat, J. L. The whole-brain concept of death remains optimum public policy. J. Law Med. Ethics 34, 35–43 (2006).
Shewmon, D. A. Constructing the death elephant: a synthetic context shift for the definition, criteria, and tests for death. J. Med. Philos. 35, 256–298 (2010).
Miller, F. A. & Truog, R. D. Death, Dying and Organ Transplantation: Reconstructing Medical Ethics at the End of Life (Oxford University Press, New York, 2011).
Halevy, A. & Brody, B. Brain death: reconciling definitions, criteria, and tests. Ann. Intern. Med. 119, 519–525 (1993).
Bernat, J. L. Contemporary controversies in the definition of death. Prog. Brain Res. 177, 21–31 (2009).
Chiong, W. Brain death without definitions. Hastings Cent. Rep. 35 (6), 20–30 (2005).
Shewmon, D. A. The brain and somatic integration: insights into the standard biological rationale for equating “brain death” with death. J. Med. Philos. 26, 457–478 (2001).
Loeb, J. The Organism as a Whole: From a Physicochemical Viewpoint (G. P. Putnam's Sons, New York, 1916).
Clayton, P. & Kauffman, S. A. On emergence, agency, and organization. Biol. Philos. 21, 501–521 (2006).
Bonelli, R. M., Prat, E. H. & Bonelli, J. Philosophical considerations on brain death and the concept of the organism as a whole. Psychiatr. Danub. 21, 3–8 (2009).
Shewmon, D. A. The “critical organ” for the organism as a whole: lessons from the lowly spinal cord. Adv. Exp. Med. Biol. 550, 23–42 (2004).
Bernat, J. L. How much of the brain must die in brain death? J. Clin. Ethics 3, 21–26 (1992).
Wijdicks, E. F., Varelas, P. N., Gronseth, G. S. & Greer, D. M. American Academy of Neurology. Evidence-based guideline update: determining brain death in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 74, 1911–1918 (2010).
Nakagawa, T. A. et al. Guidelines for the determination of brain death in infants and children: an update of the 1987 Task Force recommendations. Crit. Care Med. 39, 2139–2155 (2011).
Shemie, S. D. et al. Severe brain injury to neurological determination of death: Canadian forum recommendations. CMAJ 174, S1–S13 (2006).
Gardiner, D., Shemie, S., Manara, A. & Opdam, H. International perspective on the diagnosis of death. Br. J. Anaesth. 108 (Suppl. 1), i14–i28 (2012).
Posner, J. B., Saper, C. B., Schiff, N. D. & Plum, F. Plum and Posner's Diagnosis of Stupor and Coma 4th edn 95–114 (Oxford University Press, New York, 2007).
Ropper, A. H. Lateral displacement of the brain and level of consciousness in patients with an acute hemispheral mass. N. Engl. J. Med. 314, 953–958 (1986).
Bernat, J. L. On irreversibility as a prerequisite for brain death determination. Adv. Exper. Med. Biol. 550, 161–167 (2004).
Ogata, J., Imakita, M., Yutani, C., Miyamoto, S. & Kikuchi, H. Primary brainstem death: a clinico-pathological study. J. Neurol. Neurosurg. Psychiatry 51, 646–650 (1988).
Kosteljanitz, M., Øhrstrøm, J. K., Skjøt, S. & Teglbjærg, P. S. Clinical brain death with preserved arterial circulation. Acta Neurol. Scand. 78, 418–421 (1988).
Machado, C. Consciousness as a definition of death: its appeal and complexity. Clin. Electroencephalogr. 30, 156–164 (1999).
Shewmon, D. A. Brain death: can it be resuscitated? Hastings Cent. Rep. 39 (2), 18–24 (2009).
Truog, R. D. Is it time to abandon brain death? Hastings Cent. Rep. 27 (1), 29–37 (1997).
Shewmon, D. A. Chronic “brain death:” meta-analysis and conceptual consequences. Neurology 51, 1538–1545 (1998).
Repertinger, S., Fitzgibbons, W. P., Omojola, M. F. & Brumback, R. A. Long survival following bacterial meningitis-associated brain destruction. J. Child Neurol. 21, 591–595 (2006).
Lustbader, D. et al. Second brain death examination may negatively affect organ donation. Neurology 76, 119–124 (2011).
Gelb, D. J. Second brain death examination may negatively affect organ donation. Neurology 77, 1314–1315 (2011).
Varelas, P. N. et al. Single brain death examination is equivalent to dual brain death examinations. Neurocrit. Care 15, 547–553 (2011).
Heran, M. K., Heran. N. S. & Shemie, S. D. A review of ancillary tests in evaluating brain death. Can. J. Neurol. Sci. 35, 409–419 (2008).
Wijdicks, E. F. The case against confirmatory tests for determining brain death in adults. Neurology 75, 77–83 (2010).
Busl, K. M. & Greer, D. M. Pitfalls in the diagnosis of brain death. Neurocrit. Care 11, 276–287 (2009).
Greer. D. M., Varelas, P. N., Haque, S. & Wijdicks, E. F. Variability of brain death determination guidelines in leading US neurologic institutions. Neurology 70, 284–289 (2008).
Wang, M. Y., Wallace, P. & Gruen, J. P. Brain death documentation: analysis and issues. Neurosurgery 51, 731–736 (2002).
Bernat, J. L. How can we achieve uniformity in brain death determinations? Neurology 70, 252–253 (2008).
Choi, E. K. et al. Brain death revisited: the case for a national standard. J. Law Med. Ethics 36, 824–836 (2008).
Webb, A. C. & Samuels, O. B. Reversible brain death after cardiopulmonary arrest and induced hypothermia. Crit. Care Med. 39, 1538–1542 (2011).
Streat, S. 'Reversible brain death'—is it true, confounded, or 'not proven'? Crit. Care Med. 39, 1601–1602 (2011).
Miedema, F. Medical treatment after brain death: a case report and ethical analysis. J. Clin. Ethics 2, 50–52 (1991).
Hardwig, J. Treating the brain dead for the benefit of the family. J. Clin. Ethics 2, 53–56 (1991).
Molinari, G. F. Brain death, irreversible coma, and words doctors use. Neurology 32, 400–402 (1982).
Cranford, R. E. Discontinuation of ventilation after brain death: policy should be balanced with concern for the family. BMJ 318, 1754–1755 (1999).
Shaner, D. M., Orr, R. D., Drought, T., Miller, R. B. & Siegel, M. Really, most SINCERELY dead: policy and procedure in the diagnosis of death by neurologic criteria. Neurology 62, 1683–1686 (2004).
Ormrod, J. A., Ryder, T., Chadwick, R. J. & Bonner, S. M. Experiences of families when a relative is diagnosed brain stem dead: understanding of death, observation of brain stem death testing, and attitudes to organ donation. Anaesthesia 60, 1002–1008 (2005).
Kompanje, E. J., de Groot, Y. J., Bakker, J. & Ijzermans, J. N. A national multicenter trial on family presence during brain death determination: the FABRA study. Neurocrit. Care 17, 301–308 (2012).
Furton, E. J. Brain death, the soul, and organic life. Natl Cathol. Bioeth. Q. 2, 455–470 (2002).
Pontifical Academy of Sciences. The Signs of Death. Scripta Varia 110 (Pontifical Academy of Sciences, Vatican City, 2007).
Halachic Organ Donor Society. Halachic Issues. Halachic Organ Donor Society [online].
Rosner, F. in The Definition of Death: Contemporary Controversies (eds Youngner, S. J. et al.) 210–221 (Johns Hopkins University Press, Baltimore, 1999).
Padela A. I., Arozullah, A. & Moosa, E. Brain death in Islamic ethico-legal deliberation: challenges for applied Islamic bioethics. Bioethics http://dx.doi.org/10.1111/j.1467–85192011.01935.x.
Jain, S. & Maheshawari, M. C. in Brain Death (ed. Machado, C.) 261–263 (Elsevier, Amsterdam, 1995).
Lock, M. Contesting the natural in Japan: moral dilemmas and technologies of dying. Cult. Med. Psychiatry 19, 1–38 (1995).
Olick, R. S., Braun, E. A. & Potasj, J. Accommodating religious and moral objections to neurological death. J. Clin. Ethics 20, 183–191 (2009).
Steinbrook, R. Organ donation after cardiac death. N. Engl. J. Med. 357, 209–213 (2007).
Manara, A. R., Murphy, P. G. & O'Callaghan, G. Donation after circulatory death. Br. J. Anaesth. 108 (Suppl. 1), i108–i121 (2012).
Wall, S. P. et al. Derivation of the uncontrolled donation after circulatory death protocol for New York City. Am. J. Transplant. 11, 1417–1426 (2011).
Robertson, J. A. The dead donor rule. Hastings Cent. Rep. 29 (6), 6–14 (1999).
Lynn, J. & Cranford, R. E. in The Definition of Death: Contemporary Controversies (eds Youngner, S. J. et al.) 101–114 (Johns Hopkins University Press, Baltimore, 1999).
DeVita, M. A. & Snyder, J. V. Development of the University of Pittsburgh Medical Center policy for the care of terminally ill patients who may become organ donors after death following the removal of life support. Kennedy Inst. Ethics J. 3, 131–143 (1993).
Bernat, J. L. Are donors after cardiac death really dead? J. Clin. Ethics 17, 122–132 (2006).
Ethics Committee, American College of Critical Care Medicine & Society of Critical Care Medicine. Recommendations for non-heartbeating organ donation. A position paper by the Ethics Committee, American College of Critical Care Medicine, Society of Critical Care Medicine. Crit. Care Med. 29, 1826–1831 (2001).
Bernat, J. L. et al. Report of a national conference on donation after cardiac death. Am. J. Transplant. 6, 281–291 (2006).
Institute of Medicine. Non-Heart-Beating Organ Transplantation: Practice and Protocols (National Academy Press, Washington, DC, 2000).
Shemie, S. D. et al. National recommendations for donation after cardiocirculatory death in Canada. CMAJ 175 (8 Suppl.), S1–S24 (2006).
Dhanini, S., Hornby, L., Ward, R. & Shemie, S. Variability in the determination of death after cardiac arrest: a review of guidelines and statements. J. Intensive Care Med. 27, 238–252 (2012).
Boucek, M. M. et al. Pediatric heart transplantation after declaration of cardiocirculatory death. N. Engl. J. Med. 359, 709–714 (2008).
Wijdicks, E. F. & Diringer, M. N. Electrocardiographic activity after terminal cardiac arrest in neurocatastrophes. Neurology 62, 673–674 (2004).
DeVita, M. A., Snyder, J. V., Arnold, R. M. & Siminoff, L. A. Observations of withdrawal of life-sustaining treatment from patients who became non-heart-beating organ donors. Crit. Care Med. 28, 1709–1712 (2000).
Hornby, K., Hornby, L. & Shemie, S. A systematic review of autoresuscitation after cardiac arrest. Crit. Care Med. 38, 1246–1253 (2010).
Sheth, K. N., Nutter, T., Stein, D. M., Scalea, T. M. & Bernat, J. L. Auto-resuscitation after asystole in patients being considered for organ donation. Crit. Care Med. 40, 158–161 (2012).
Rogers, P. L., Schlicktig, R., Miro, A. & Pinsky, M. Auto-PEEP during CPR: an “occult” cause of electromechanical dissociation. Chest 99, 492–493 (1991).
Bernat, J. L. How auto-resuscitation impacts death determinations in organ donors. Crit. Care Med. 38, 1377–1378 (2010).
Bernat, J. L. How the distinction between “irreversible” and “permanent” illuminates circulatory–respiratory death determination. J. Med. Philos. 35, 242–255 (2010).
Truog, R. D. & Miller, F. G. Counterpoint: are donors after circulatory death really dead and does it matter? No and not really. Chest 138, 16–19 (2010).
Marquis, D. Are DCD donors dead? Hastings Cent. Rep. 40 (3), 24–31 (2010).
Bernat, J. L. et al. The circulatory–respiratory determination of death in organ donation. Crit. Care Med. 38, 972–979 (2010).
Ko, W. J., Chen, Y. S., Tsai, P. R. & Lee, P. H. Extracorporeal membrane oxygenation support of donor abdominal organs in non-heart-beating donors. Clin. Transplant. 14, 152–156 (2000).
Gravel, M. T. et al. Kidney transplantation from organ donors following cardiopulmonary death using extracorporeal membrane oxygenation support. Ann. Transplant. 9, 57–58 (2004).
Magliocca, J. F. et al. Extracorporeal support for organ donation after cardiac death effectively expands the donor pool. J. Trauma 58, 1095–1102 (2005).
President's Council on Bioethics. Controversies in the Determination of Death: A White Paper by the President's Council on Bioethics. The President's Council on Bioethics [online].
Shemie, S. D. International Guidelines for the Determination of Death. Critical Care Canada Forum [online].
Klein, A. S. et al. Organ donation and utilization in the United States: 1998–2008. Am. J. Transplant. 10, 973–986 (2010).
Merchant, S. J. et al. Exploring the psychological effects of deceased organ donation on the families of organ donors. Clin. Transplant. 22, 341–347 (2008).
DeVita, M. A. & Caplan, A. L. Caring for organs or patients? Ethical concerns about the Uniform Anatomical Gift Act (2006). Ann. Intern. Med. 147, 876–879 (2007).
Iltis, A. S., Rie, M. A. & Wall, A. Organ donation, patients' rights, and medical responsibilities at the end of life. Crit. Care Med. 37, 310–315 (2009).
Truog, R. D. Consent for organ donation—balancing conflicting ethical obligations. N. Engl. J. Med. 358, 1209–1211 (2008).
Arnold, R. M., Siminoff, L. A. & Frader, J. E. Ethical issues in organ procurement: a review for intensivists. Crit. Care Clin. 12, 29–48 (1996).
Mandell, M. S. et al. National evaluation of healthcare provider attitudes toward organ donation after cardiac death. Crit. Care Med. 34, 2952–2958 (2006).
Hart, J. L., Kohn, R. & Halpern, S. D. Perceptions of organ donation after circulatory determination of death among critical care physicians and nurses: a national survey. Crit. Care Med. 40, 2595–2600 (2012).
Bastami, S., Krones, T. & Biller-Adorno, N. Systematic review of attitudes toward donation after cardiac death among health care providers and the general public. Crit. Care Med. (in press).
Simpkin, A. L., Robertson, L. C., Barber, V. S. & Young, J. D. Modifiable factors influencing relatives' decision to offer organ donation: systematic review. BMJ 338, b991 (2009).
Kalkbrenner, K. J. & Hardart, G. E. Consent for donation after cardiac death: a survey of procurement organizations. J. Intensive Care Med. 27, 253–263 (2012).
Zink, S. & Wertleib, S. A study of the presumptive approach to consent for organ donation: a new solution to an old problem. Crit. Care Nurse 26, 129–136 (2006).
Luskin, R. S., Glazier, A. K. & Delmonico, F. L. Organ donation and dual advocacy. N. Engl. J. Med. 358, 1207–1208 (2008).
Browne, A., Gillett, G. & Tweedale, M. The ethics of elective (non-therapeutic) ventilation. Bioethics 14, 42–57 (2000).
Bernat, J. L. Point: are donors after circulatory death really dead and does it matter? Yes and Yes. Chest 138, 13–16 (2010).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Bernat, J. Controversies in defining and determining death in critical care. Nat Rev Neurol 9, 164–173 (2013). https://doi.org/10.1038/nrneurol.2013.12
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2013.12
This article is cited by
-
The History of Donation after Circulatory Death (DCD): Backlash and Boomerang
Neurocritical Care (2024)
-
Rationale for revisions to the definition of death and criteria for its determination in Canada
Canadian Journal of Anesthesia/Journal canadien d'anesthésie (2023)
-
The ethical obligation of the dead donor rule
Medicine, Health Care and Philosophy (2020)
-
Medicolegal Complications of Apnoea Testing for Determination of Brain Death
Journal of Bioethical Inquiry (2018)
-
Donation after brain circulation determination of death
BMC Medical Ethics (2017)