Abstract
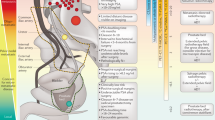
High-risk prostate cancer can be defined by the assessment of pretreatment prognostic factors such as clinical stage, Gleason score, and PSA level. High-risk features include PSA >20 ng/ml, Gleason score 8–10, and stage T3 tumors. Patients with adverse prognostic factors have historically fared poorly with monotherapeutic approaches. Multimodal treatment utilizing combined androgen suppression and radiotherapy has improved survival rates for patients with high-risk prostate cancer. In addition, multiple randomized trials in patients treated with primary radical prostatectomy have demonstrated improved outcomes with the addition of adjuvant radiotherapy. Improved radiotherapy techniques that allow for dose escalation, and new systemic therapy approaches such as adjuvant chemotherapy, present promising future therapeutic alternatives for patients with high-risk prostate cancer.
Key Points
-
Risk stratification using prognostic factors such as clinical stage, Gleason score, and PSA level is essential for determining the appropriate treatment for patients with prostate cancer
-
High-risk patients include those with Gleason score 8–10, PSA >20 ng/ml, and clinical stage T3 disease
-
The combination of radiotherapy and long-term androgen suppression improves disease control and survival, compared with either treatment alone, for patients with high-risk disease
-
Adjuvant radiotherapy following radical prostatectomy improves disease control and survival for patients with stage pT3 disease
-
Improved local treatments, such as dose-escalated radiotherapy, adjuvant radiotherapy following prostatectomy, and novel systemic treatments such as adjuvant chemotherapy have the potential to improve disease control in the future
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Jemal, A. et al. Cancer Statistics, 2009. CA Cancer J. Clin. 59, 225–249 (2009).
Albertsen, P. C., Fryback, D. G., Storer, B. E., Kolon, T. F. & Fine, J. Long-term survival among men with conservatively treated prostate cancer. JAMA 274, 626–631 (1995).
Albertsen, P. C. A challenge to contemporary management of prostate cancer. Nat. Clin. Pract. Urol. 6, 12–13 (2009).
Ryan, C. J., Elkin, E. P., Cowan, J. & Carroll, P. R. Initial treatment patterns and outcome of contemporary prostate cancer patients with bone metastases at initial presentation: data from CaPSURE. Cancer 110, 81–86 (2007).
American Joint Committee on Cancer. AJCC Staging Manual, 6th edn (Springer, New York, 2002).
Gleason, D. F. & Mellinger, G. T. Prediction of prognosis for prostatic adenocarcinoma by combined histological grading and clinical staging. J. Urol. 111, 58–64 (1974).
Zagars, G. K., Ayala, A. G., von Eschenbach, A. C. & Pollack, A. The prognostic importance of Gleason grade in prostatic adenocarcinoma: a long term-follow-up study of 648 patients treated with radiation therapy. Int. J. Radiat. Oncol. Biol. Phys. 31, 237–245 (1995).
Rioux-Leclercq, N. C., Chan, D. Y. & Epstein, J. I. Prediction of outcome after radical prostatectomy in men with organ-confined Gleason score 8–10 adenocarcinoma. Urology 60, 666–669 (2002).
Bastian, P. J. et al. Clinical and pathologic outcome after radical prostatectomy for prostate cancer patients with a preoperative Gleason Sum of 8 to 10. Cancer 107, 1265–1272 (2006).
Zagars, G. K., Pollock, A. & Eschenbach, A. C. Prognostic factors for clinically localized prostate carcinoma: analysis of 938 patients irradiated in the prostate specific antigen era. Cancer 79, 1370–1380 (1997).
Mitchell, R. E. et al. Preoperative serum prostate specific antigen remains a significant prognostic variable in predicting biochemical failure after radical prostatectomy. J. Urol. 175, 1663–1667 (2006).
Inman, B. A. et al. Long-term outcomes of radical prostatectomy with multimodal adjuvant therapy in men with a preoperative serum prostate-specific antigen level of > or = 50 ng/ml. Cancer 113, 1544–1551 (2008).
Pisansky, T. M., Kahn, M. J. & Bostwick, D. G. An enhanced prognostic system for clinically localized carcinoma of the prostate. Cancer 79, 2154–2161 (1997).
Soloway, M. & Roach, M. Prostate cancer progression after therapy of primary curative intent: a review of data from prostate-specific antigen era. Cancer 104, 2310–2322 (2005).
Partin, A. W. et al. Combination of prostate-specific antigen, clinical stage, and Gleason score to predict pathological stage of localized prostate cancer. A multi-institutional update. JAMA 277, 1445–1451 (1997).
Kattan, M. W., Eastham, J. A., Stapleton, A. M., Wheeler, T. M. & Scardino, P. T. A preoperative nomogram for disease recurrence following radical prostatectomy for prostate cancer. J. Natl Cancer Inst. 90, 766–771 (1998).
Kattan, M. W. et al. Pretreatment nomogram for predicting the outcome of three-dimensional conformal radiotherapy in prostate cancer. J. Clin. Oncol. 18, 3352–3359 (2000).
D'Amico, A. V. et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA 280, 969–974 (1998).
Greene, K. L. et al. Who is the average patient presenting with prostate cancer? Urology 66, 76–82 (2005).
D'Amico, A. V., Chen, M. H., Roehl, K. A. & Catalona, W. J. Preoperative PSA velocity and the risk of death from prostate cancer after radical prostatectomy. N. Engl. J. Med. 351, 125–135 (2004).
D'Amico, A. V., Renshaw, A. A., Sussman, B. & Chen, M. H. Preoperative PSA velocity and risk of death from prostate cancer following external beam radiation therapy. JAMA 294, 440–447 (2005).
Hanks, G. E., Hanlon, A. L., Lee, W. R., Slivjak, A. & Schultheiss, T. E. Pretreatment prostate-specific antigen doubling times: clinical utility of this predictor of prostate cancer behavior. Int. J. Radiat. Oncol. Biol. Phys. 34, 549–553 (1996).
Roberts, S. G., Blute, M. L., Bergstralh, E. J., Slezak, J. M. & Zincke, H. PSA doubling time as a predictor of clinical progression after biochemical failure following radical prostatectomy for prostate cancer. Mayo Clin. Proc. 76, 576–581 (2001).
Grossfeld, G. D. et al. Predicting disease recurrence in intermediate and high-risk patients undergoing radical prostatectomy using percent positive biopsies: results from CaPSURE. Urology 59, 560–565 (2002).
Dong, F. et al. Prostate cancer volume at biopsy predicts clinically significant upgrading. J. Urol. 179, 896–900 (2008).
Whittemore, D. E. et al. Significance of tertiary Gleason pattern 5 in Gleason Score 7 radical prostatectomy specimens. J. Urol. 179, 516–522 (2008).
Nanda, A., Chen, M. H., Renshaw, A. A. & D'Amico, A. V. Gleason pattern 5 prostate cancer: further stratification of patients with high-risk disease and implications for future randomized trials. Int. J. Radiat. Oncol. Biol. Phys. 74, 1419–1423 (2009).
Roach, M., Waldman, F. & Pollack, A. Predictive models in external beam radiotherapy for clinically localized prostate cancer. Cancer 115, 3112–3120 (2009).
Pilepich, M. V. et al. Phase III trial of androgen suppression using goserelin in unfavorable-prognosis carcinoma of the prostate treated with definitive radiotherapy: report of Radiation Therapy Oncology Group Protocol 85–31. J. Clin. Oncol. 15, 1013–1021 (1997).
Lawton, C. A. et al. Updated results of the phase III Radiation Therapy Oncology Group (RTOG) trial 85–31 evaluating the potential benefit of androgen suppression following standard radiation therapy for unfavorable prognosis carcinoma of the prostate. Int. J. Radiat. Oncol. Biol. Phys. 49, 937–946 (2001).
Pilepich, M. V. et al. Androgen suppression adjuvant to definitive radiotherapy in prostate adenocarcinoma—long-term results of phase III RTOG 85–31. Int. J. Radiat. Oncol. Biol. Phys. 61, 1285–1290 (2005).
Pilepich, M. V. et al. Androgen deprivation with radiation therapy compared with radiation therapy alone for locally advanced prostatic carcinoma: a randomized comparative trial of the Radiation Therapy Oncology Group. Urology 45, 616–623 (1995).
Pilepich, M. V. et al. Phase III radiation therapy oncology group (RTOG) trial 86–10 of androgen deprivation adjuvant to definitive radiotherapy in locally advanced carcinoma of the prostate. Int. J. Radiat. Oncol. Biol. Phys. 50, 1243–1252 (2001).
Roach, M. et al. Short-term neoadjuvant androgen deprivation therapy and external-beam radiotherapy for locally advanced prostate cancer: long-term results of RTOG 8610. J. Clin. Oncol. 26, 585–591 (2008).
D'Amico, A. V. et al. 6-month androgen suppression plus radiation therapy vs radiation therapy alone for patients with clinically localized prostate cancer: a randomized controlled trial. JAMA 292, 821–827 (2004).
Denham, J. W. et al. Short-term androgen deprivation and radiotherapy for locally advanced prostate cancer: results form the Trans-Tasman Radiation Oncology group 96.01 randomised controlled trial. Lancet Oncol. 6, 841–850 (2005).
Bolla, M. et al. Improved survival in patients with locally advanced prostate cancer treated with radiotherapy and goserelin. N. Engl. J. Med. 337, 295–300 (1997).
Bolla, M. et al. Long-term results with immediate androgen suppression and external irradiation in patients with locally advanced prostate cancer (an EORTC study): a phase III randomised trial. Lancet 360, 103–106 (2002).
Bolla, M., Descotes, J. L., Artignan, X. & Fourneret, P. Adjuvant treatment to radiation: combined hormone therapy and external radiotherapy for locally advanced prostate cancer. BJU Int. 100, 44–47 (2007).
Hanks, G. E. et al. Phase III trial of long-term adjuvant androgen deprivation after neoadjuvant hormonal cytoreduction and radiotherapy in locally advanced carcinoma of the prostate: the Radiation Therapy Oncology Group Trial 92–02. J. Clin. Oncol. 21, 3972–3978 (2003).
Horwitz, E. M. et al. Ten-year follow-up of radiation therapy oncology group protocol 92–02: a phase III trial of the duration of elective androgen deprivation in locally advanced prostate cancer. J. Clin. Oncol. 26, 2497–2504 (2008).
Bolla, M. et al. Duration of androgen suppression in the treatment of prostate cancer. N. Engl. J. Med. 360, 2516–2527 (2009).
Widmark, A. et al. Endocrine treatment, with or without radiotherapy, in locally advanced prostate cancer (SPCG-7/SFUO-3): an open randomised phase III trial. Lancet 373, 301–308 (2009).
Rosenthal, S. A. Prostate cancer: local control and radiotherapy matter in prostate cancer. Nat. Rev. Urol. 6, 250–251 (2009).
Nanda, A. et al. Total androgen blockade versus a luteinizing hormone-releasing hormone agonist alone in men with high-risk prostate cancer treated with radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. doi:10.1016/j.ijrobp.2009.03.034.
Crook, J. et al. Report of a multicenter Canadian phase III randomized trial of 3 months vs. 8 months neoadjuvant androgen deprivation before standard-dose radiotherapy for clinically localized prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 60, 15–23 (2004).
Roach, M. et al. Predicting the risk lymph node involvement using the pre-treatment prostate specific antigen and Gleason score in men with clinically localized prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 28, 33–37 (1994).
Roach, M. et al. Phase III trial comparing whole-pelvic versus prostate-only radiotherapy and neoadjuvant versus adjuvant combined androgen suppression: Radiation Therapy Oncology Group 9413. J. Clin. Oncol. 21, 1904–1911 (2004).
Lawton, C. A. et al. An update of the phase III trial comparing whole pelvic to prostate only radiotherapy and neoadjuvant to adjuvant total androgen suppression: updated analysis of RTOG 94–13, with emphasis on unexpected hormone/radiation interactions. Int. J. Radiat. Oncol. Biol. Phys. 69, 646–655 (2007).
Pommier, P. et al. Is there a role for pelvic irradiation in localized prostate adenocarcinoma? Preliminary Results of GETUG-01. J. Clin. Oncol. 25, 5366–5373 (2007).
Nguyen, P. L. & D'Amico, A. V. Targeting pelvic lymph nodes in men with intermediate- and high-risk prostate cancer despite two negative randomized trials. J. Clin. Oncol. 26, 2055–2056 (2008).
Shahinian, V. B., Kuo, Y. F., Freeman, J. L. & Goodwin, J. S. Risk of fracture after androgen deprivation for prostate cancer. N. Engl. J. Med. 352, 154–164 (2005).
Feigenberg, S. J. et al. Long-term androgen deprivation increases Grade 2 and higher late morbidity in prostate cancer patients treated with three-dimensional conformal radiation therapy. Int. J. Radiat. Oncol. Biol. Phys. 62, 397–405 (2005).
D'Amico, A. V. et al. Influence of androgen suppression therapy for prostate cancer on the frequency and timing of fatal myocardial infarction. J. Clin. Oncol. 25, 2420–2425 (2007).
Saigal, C. S. et al. Androgen deprivation therapy increases cardiovascular morbidity in men with prostate cancer. Cancer 110, 1493–1500 (2007).
Tsai, H. K., D'Amico, A. V., Sadetsky, N., Chen, M. H. & Carroll, P. R. Androgen deprivation therapy for localized prostate cancer and the risk of cardiovascular mortality. J. Natl Cancer Inst. 99, 1516–1524 (2007).
Lawton, C. A., Bae, K., Pilepich, M., Hanks, G. & Shipley, W. Long-term treatment sequelae after external beam irradiation with or without hormonal manipulation for adenocarcinoma of the prostate: analysis of radiation therapy oncology group studies 85–31, 86–10, and 92–02. Int. J. Radiat. Oncol. Biol. Phys. 70, 437–441 (2008).
Israeli, R. S., Ryan, C. W. & Jung, L. L. Managing bone loss in men with locally advanced prostate cancer receiving androgen deprivation therapy. J. Urol. 179, 414–423 (2008).
Smith, M. R. et al. Denosumab in men receiving androgen-deprivation therapy for prostate cancer. N. Engl. J. Med. 361, 745–755 (2009).
Rosenthal, S. A. et al. Phase III multi-institutional trial of adjuvant chemotherapy with paclitaxel, estramustine, and oral etoposide combined with long-term androgen suppression therapy and radiotherapy versus long-term androgen suppression plus radiotherapy alone for high-risk prostate cancer: preliminary toxicity analysis of RTOG 99–02. Int. J. Radiat. Oncol. Biol. Phys. 73, 672–678 (2009).
Tannock, I. F. et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N. Engl. J. Med. 351, 1502–1512 (2004).
James, N. D. et al. Systemic therapy for advancing or metastatic prostate cancer (STAMPEDE): a multi-arm, multistage randomized controlled trial. BJU Int. 103, 464–469 (2008).
Oh, W. K. An overview of chemotherapy trials in localized and recurrent nonmetastatic prostate cancer. J. Urol. 172, S34–S37 (2004).
Eastham, J. A., Kelly, W. K., Grossfeld, G. D. & Small, E. J. Cancer and Leukemia Group B (CALGB) 90203: a randomized phase 3 study of radical prostatectomy alone versus estramustine and docetaxel before radical prostatectomy for patients with high-risk localized disease. Urology 62, 55–62 (2003).
Flaig, T. W. et al. Randomization reveals unexpected acute leukemias in Southwest Oncology Group prostate cancer trial. J. Clin. Oncol. 26, 1532–1536 (2008).
Pollack, A. et al. Prostate cancer radiation dose response: results of the M. D. Anderson phase III randomized trial. Int. J. Radiat. Oncol. Biol. Phys. 53, 1097–1105 (2002).
Dearnaley, D. P. et al. Phase III pilot study of dose escalation using conformal radiotherapy in prostate cancer: PSA control and side effects. Br. J. Cancer 92, 488–498 (2005).
Zeitman, A. L. et al. Comparison of conventional-dose vs high-dose conformal radiation therapy in clinically localized adenocarcinoma of the prostate: a randomized controlled trial. JAMA 294, 1233–1239 (2005).
Dearnaley, D. P. et al. Escalated-dose versus standard-dose conformal radiotherapy in prostate cancer: first results from the MRC RT01 randomised controlled trial. Lancet Oncol. 8, 475–487 (2007).
Al-Mamgani, A. et al. Update of Dutch multicenter dose-escalation trial of radiotherapy for localized prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 72, 980–988 (2008).
Kuban, D. A. et al. Long-term results of the M. D. Anderson randomized dose-escalation trial for prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 70, 67–74 (2008).
Martinez, A. A. et al. Dose escalation using conformal high-dose-rate brachytherapy improves outcome in unfavorable prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 53, 316–327 (2002).
Vicini, F. A., Vargas, C., Edmunson, G., Kestin, L. & Martinez, A. The role of high-dose rate brachytherapy in locally advanced prostate cancer. Semin. Radiat. Oncol. 13, 98–108 (2003).
Demanes, D. J., Rodriguez, R. R., Schour, L., Brandt, D. & Altieri, G. High-dose-rate intensity-modulated brachytherapy with external beam radiotherapy for prostate cancer: California endocurietherapy's 10-year results. Int. J. Radiat. Oncol. Biol. Phys. 61, 1306–1316 (2005).
Lee, W. R. The role of androgen deprivation therapy combined with prostate brachytherapy. Urology 60, 39–44 (2002).
Stock, R. G., Ho, A., Cesaretti, J. A. & Stone, N. N. Changing the patterns of failure for high-risk prostate cancer patients by optimizing local control. Int. J. Radiat. Oncol. Biol. Phys. 66, 389–394 (2006).
Stock, R. G., Cesaretti, J. A., Hall, S. J. & Stone, N. N. Outcomes for patients with high-grade prostate cancer treated with a combination of brachytherapy, external beam radiotherapy and hormonal therapy. BJU Int. 104, 1631–1636 (2009).
Stone, N. N. et al. Multicenter analysis of effect of high biologic effective dose on biochemical failure and survival outcomes in patients with Gleason score 7–10 prostate cancer treated with permanent prostate brachytherapy. Int. J. Radiat. Oncol. Biol. Phys. 73, 341–346 (2009).
Mohler, J. L. Updating the prostate cancer guidelines. J. Natl Compr. Canc. Netw. 5, 647–648 (2007).
Thompson, I. M. et al. Adjuvant radiotherapy for pathologically advanced prostate cancer: a randomized clinical trial. JAMA 296, 2329–2335 (2006).
Bolla, M. et al. Postoperative radiotherapy after radical prostatectomy: a randomized controlled trial (EORTC trial 22911). Lancet 366, 572–576 (2005).
Weigel, T. et al. Phase III postoperative adjuvant radiotherapy after radical prostatectomy compared with radical prostatectomy alone in pT3 prostate cancer with postoperative undetectable prostate-specific antigen: ARO 96–02/AUO AP 09/95. J. Clin. Oncol. 27, 2924–2930 (2009).
Thompson, I. M. et al. Adjuvant radiotherapy for pathological T3N0M0 prostate cancer significantly reduces risk of metastases and improves survival: long-term followup of a randomized clinical trial. J. Urol. 181, 956–962 (2009).
Aragon-Ching, J. B. & Dahut, W. L. The role of angiogenesis inhibitors in prostate cancer. Cancer J. 14, 20–25 (2008).
Aragon-Ching, J. B. & Dahut, W. L. VEGF inhibitors and prostate cancer therapy. Curr. Mol. Pharmacol. 2, 161–168 (2009).
Attard, G. et al. Phase I clinical trial of a selective inhibitor of CYP17, abiraterone acetate, confirms that castration-resistant prostate cancer commonly remains hormone driven. J. Clin. Oncol. 26, 4563–4571 (2008).
Tran, C. et al. Development of a second-generation antiandrogen for treatment of advanced prostate cancer. Science 324, 787–790 (2009).
Nilsson, S. et al. Bone-targeted radium-223 in symptomatic, hormone-refractory prostate cancer: a randomised, multicentre, placebo-controlled phase II study. Lancet Oncol. 8, 587–594 (2007).
Tu, S. M. & Lin, S. H. Current trials using bone-targeting agents in prostate cancer. Cancer J. 14, 35–39 (2008).
Acknowledgements
The authors would like to thank Heidi Mortensen, Medical Librarian, for her great help in assembling the references. Charles P. Vega, University of California, Irvine, CA is the author of and is solely responsible for the content of the learning objectives, questions and answers of the MedscapeCME-accredited continuing medical education activity associated with this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Howard M. Sandler declares he has acted as a consultant for Sanofi-Aventis. S. A. Rosenthal, the Journal Editor S. Farley and the CME questions author C. P. Vega declare no competing interests.
Rights and permissions
About this article
Cite this article
Rosenthal, S., Sandler, H. Treatment strategies for high-risk locally advanced prostate cancer. Nat Rev Urol 7, 31–38 (2010). https://doi.org/10.1038/nrurol.2009.237
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2009.237
This article is cited by
-
Association of radiotherapy for prostate cancer and second primary colorectal cancer: a US population-based analysis
Techniques in Coloproctology (2024)
-
Is it essential to use fiducial markers during cone-beam CT-based radiotherapy for prostate cancer patients?
Japanese Journal of Radiology (2017)
-
Menthol Inhibits the Proliferation and Motility of Prostate Cancer DU145 Cells
Pathology & Oncology Research (2012)