Key Points
-
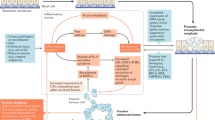
Male fertility is controlled by a Zn2+-dependent short circuit of the Krebs cycle within prostate epithelial cells
-
Homeostasis of the prostate epithelium is reliant on the intracellular androgen-dependent accumulation of Zn2+ and citrate
-
Sperm motility requires the coordinated action of the components of the two main fluids in the human seminal plasma: the prostatic fluid, which is enriched with Zn2+, citrate and kallikreins, and the semenogelin-enriched seminal vesicle secretion
-
The prostate is the direct target for a number of benign and malignant diseases that are potentially linked to impaired fertility status
-
Prostatitis might be directly linked with changes in fertility
Abstract
Ejaculation is a synchronized cascade of events that has the ultimate goal of activating sperm and enabling them to reach an egg for fertilization. The seminal plasma contains a complex mixture of fluids that is secreted from the testes, epididymis and male accessory glands. The prostate gland has a pivotal role in this process, as prostatic fluid enriched in Zn2+, citrate and kallikreins is crucial for the molecular synchronization of the functional cascade triggered by ejaculatory stimuli. The prostate is the target of a number of common diseases that can affect male fertility at different ages. In both young and aged men, prostatic diseases or an unhealthy prostate can affect spermatozoa functioning and, therefore, male fertility. Consideration of prostate physiology emphasizes a number of points: the central role of Zn2+ and citrate in the regulation of prostate epithelium homeostasis and in ejaculation; the influence of bacteria-related prostatic inflammation on male fertility; and the potential role of prostatic inflammation in promoting the development of prostatic hyperplastic growth and carcinogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Gilany, K., Minai-Tehrani, A., Savadi-Shiraz, E., Rezadoost, H. & Lakpour, N. Exploring the human seminal plasma proteome: an unexplored gold mine of biomarker for male infertility and male reproduction disorder. J. Reprod. Infertil. 16, 61–71 (2015).
Tena-Sempere, M. Ghrelin, the gonadal axis and the onset of puberty. Endocr. Dev. 25, 69–82 (2013).
Nargund, V. H. Effects of psychological stress on male fertility. Nat. Rev. Urol. 12, 373–382 (2015).
Rey, R. A. et al. Male hypogonadism: an extended classification based on a developmental, endocrine physiology-based approach. Andrology 1, 3–16 (2013).
Nieto, C. M., Rider, L. C. & Cramer, S. D. Influence of stromal–epithelial interactions on androgen action. Endocr. Relat. Cancer 21, T147–T160 (2014).
Wagenlehner, F. et al. Prostatitis and andrological implications. Minerva Urol. Nefrol. 65, 117–123 (2013).
Ficarra, V. et al. The role of prostatic inflammation in low urinary tract symptoms (LUTS) due to benign prostatic hyperplasia (BPH) and its potential impact on medical therapy. Curr. Urol. Rep. 15, 463–469 (2014).
Wagenlehner, F. M. E. et al. The role of inflammation and infection in the pathogenesis of prostate carcinoma. BJU Int. 100, 733–737 (2007).
Roberts, R. O., Lieber, M. M., Bostwick, D. G. & Jacobsen, S. J. A review of clinical and pathological prostatitis syndromes. Urology 49, 809–821 (1997).
Kalinska, M., Meyer-Hoffert, U., Kantyka, T., Potempa, J. Kallikreins. The melting pot of activity and function. Biochimie 122, 270–282 (2016).
Medrano, A. et al. Utilization of citrate and lactate through a lactate dehydrogenase and ATP-regulated pathway in boar spermatozoa. Mol. Reprod. Dev. 73, 369–378 (2006).
Franz, M. C. et al. Zinc transporters in prostate cancer. Mol. Aspects Med. 34, 735–741 (2013).
Franklin, R. B., Milon, B., Feng, P. & Costello, L. C. Zinc and zinc transporters in normal prostate and the pathogenesis of prostate cancer. Front. Biosci. 10, 2230–2239 (2005).
Lorenzetti, S. & Narciso, L. in Computational Approaches To Nuclear Receptors (eds Cozzini, P. & Kellogg, G. E.) 1–22 (RSC Publishing, 2012).
Takayama, K. & Inoue, S. Transcriptional network of androgen receptor in prostate cancer progression. Int. J. Urol. 20, 756–768 (2013).
Horie-Inoue, K. & Inoue, S. Genome-wide integrated analyses of androgen receptor signaling in prostate cancer based on high-throughput technology. Curr. Drug Targets 14, 472–480 (2013).
Lamont, K. R. & Tindall, D. J. Androgen regulation of gene expression. Adv. Cancer Res. 107, 137–162 (2010).
Johnson, L. A., Kanak, M. A., Kajdacsy-Balla, A., Pestaner, J. P. & Bagasra, O. Differential zinc accumulation and expression of human zinc transporter 1 (hZIP1) in prostate glands. Methods 52, 316–321 (2010).
Costello, L. C. & Franklin, R. B. The clinical relevance of the metabolism of prostate cancer; zinc and tumor suppression: connecting the dots. Mol. Cancer 5, 17 (2006).
Franklin, R. B. et al. hZIP1 zinc uptake transporter down regulation and zinc depletion in prostate cancer. Mol. Cancer 4, 32 (2005).
Desouki, M. M., Geradts, J., Milon, B., Franklin, R. B. & Costello, L. C. hZip2 and hZip3 zinc transporters are down regulated in human prostate adenocarcinomatous glands. Mol. Cancer 6, 37 (2007).
Kolenko, V., Teper, E., Kutikov, A. & Uzzo, R. Zinc and zinc transporters in prostate carcinogenesis. Nat. Rev. Urol. 10, 219–226 (2013).
Tepaamorndech, S., Huang, L. & Kirschke, C. P. A null-mutation in the Znt7 gene accelerates prostate tumor formation in a transgenic adenocarcinoma mouse prostate model. Cancer Lett. 308, 33–42 (2011).
Thorek, D. L., Evans, M. J., Carlsson, S. V., Ulmert, D. & Lilja, H. Prostate-specific kallikrein-related peptidases and their relation to prostate cancer biology and detection. Thromb. Haemost. 110, 484–492 (2013).
Bartoletti, R. et al. Prevalence, incidence estimation, risk factors and characterization of chronic prostatitis/chronic pelvic pain syndrome in urological hospital outpatients in Italy: results of a multicenter case-control observational study. J. Urol. 178, 2411–2415 (2007).
Domes, T. et al. The incidence and effect of bacteriospermia and elevated seminal leukocytes on semen parameters. Fertil. Steril. 97, 1050–1055 (2012).
Mändar, R., Raukas, E., Tu¨rk, S., Korrovits, P. & Punab, M. Mycoplasmas in semen of chronic prostatitis patients. Scand. J. Urol. Nephrol. 39, 479–482 (2005).
Fraczek, M. et al. Membrane stability and mitochondrial activity of human-ejaculated spermatozoa during in vitro experimental infection with Escherichia coli, Staphylococcus haemolyticus and Bacteroides ureolyticus. Andrologia 44, 315–329 (2012).
Schulz, M., Sánchez, R., Soto, L., Risopatrón, J. & Villegas, J. Effect of Escherichia coli and its soluble factors on mitochondrial membrane potential, phosphatidylserine translocation, viability, and motility of human spermatozoa. Fertil. Steril. 94, 619–623 (2010).
Diemer, T. et al. Escherichia coli-induced alterations of human spermatozoa. An electron microscopy analysis. Int. J. Androl. 23, 178–186 (2000).
Marconi, M., Pilatz, A., Wagenlehner, F., Diemer, T. & Weidner, W. Impact of infection on the secretory capacity of the male accessory glands. Int. Braz. J. Urol. 35, 299–309 (2000).
Dobrindt, U., Hochhut, B., Hentschel, U. & Hacker, J. Genomic islands in pathogenic and environmental microorganisms. Nat. Rev. Microbiol. 2, 414–424 (2004).
Marrs, C. F., Zhang, L. & Foxman, B. Escherichia coli mediated urinary tract infections: are there distinct uropathogenic E. coli (UPEC) pathotypes? FEMS Microbiol. Lett. 252, 183–190 (2005).
Dhakal, B. K. & Mulvey, M. A. The UPEC pore-forming toxin α-hemolysin triggers proteolysis of host proteins to disrupt cell adhesion, inflammatory, and survival pathways. Cell Host Microbe 11, 58–69 (2012).
Diemer, T., Ludwig, M., Huwe, P., Hales, B. & Weidner, W. Influence of urogenital infection on sperm function. Curr. Opin. Urol. 10, 39–44 (2000).
Kaur, K. & Prabha, V. Impairment by sperm agglutinating factor isolated from Escherichia coli: receptor specific interactions. Biomed. Res. Int. 54, 84–97 (2013).
Ludwig, M. et al. Experimental Escherichia coli epididymitis in rats: a model to assess the outcome of antibiotic treatment. BJU Int. 90, 933–938 (2002).
Lu, Y. et al. Necrosis is the dominant cell death pathway in uropathogenic Escherichia coli elicited epididymo-orchitis and is responsible for damage of rat testis. PLoS ONE 8, e52919 (2013).
Bhushan, S. et al. Uropathogenic Escherichia coli block MyD88-dependent and activate MyD88-independent signaling pathways in rat testicular cells. J. Immunol. 180, 5537–5547 (2008).
Dohle, G. R. Inflammatory-associated obstructions of the male reproductive tract. Andrologia 35, 321–324 (2003).
Cai, T., Mazzoli, S., Mondaini, N., Malossini, G. & Bartoletti, R. Chlamydia trachomatis infection: a challenge for the urologist. Microbiol. Res. 2, e14 (2011).
Krishnan, R. & Heal, M. R. Study of the seminal vesicles in acute epididymitis. Br. J. Urol. 67, 632–637 (1991).
Alwaal, A., Breyer, B. N. & Lue, T. F. Normal male sexual function: emphasis on orgasm and ejaculation. Fertil. Steril. 104, 1051–1060 (2015).
Eley, A., Pacey, A. A., Galdiero, M., Galdiero, M. & Galdiero, F. Can Chlamydia trachomatis directly damage your sperm? Lancet Infect. Dis. 5, 53–57 (2005).
Erbengi, T. Ultrastructural observations on the entry of Chlamydia trachomatis into human spermatozoa. Hum. Reprod. 8, 416–421 (1993).
Munoz, G., Posnett, D. N. & Witkin, S. S. Enrichment of γδ T lymphocytes in human semen: relation between γδ T cell concentration and antisperm antibody status. J. Reprod. Immunol. 22, 47–57 (1992).
Karinen, L. et al. Antibodies to Chlamydia trachomatis heat shock proteins Hsp60 and Hsp10 and subfertility in general population at age 31. Am. J. Reprod. Immunol. 52, 291–297 (2004).
Domeika, M., Domeika, K., Paavonen, J., Mardh, P. A. & Witkin, S. S. Humoral immune response to conserved epitopes of Chlamydia trachomatis and human 60-kDa heat-shock protein in women with pelvic inflammatory disease. J. Infect. Dis. 177, 714–719 (1998).
Mazzoli, S. Chlamydia trachomatis infection is related to poor semen quality in young prostatitis patients. Eur. Urol. 57, 708–714 (2010).
Leib, Z., Bartoov, B., Eltes, F. & Servadio, C. Reduced semen quality caused by chronic abacterial prostatitis: an enigma or reality? Fertil. Steril. 61, 1109–1116 (1994).
Potts, J. M. & Pasqualotto, F. F. Seminal oxidative stress in patients with chronic prostatitis. Andrologia 35, 304–308 (2003).
Showell, M. G. et al. Antioxidants for male subfertility. Cochrane Database Syst. Rev. 12, CD007411 (2014).
Hochreiter, W. W., Duncan, J. L. & Schaeffer, A. J. Evaluation of the bacterial flora of the prostate using a 16S rRNA gene based polymerase chain reaction. J. Urol. 163, 127–130 (2000).
Kaur, K. & Prabha, V. Spermagglutinating Escherichia coli and its role in infertility: in vivo study. Microb. Pathog. 69–70, 33–38 (2014).
Ochsendorf, F. R. et al. Chlamydia trachomatis and male infertility: Chlamydia-IgA antibodies in seminal plasma are C. trachomatis specific and associated with an inflammatory response. J. Eur. Acad. Dermatol. Venereol. 12, 143–152 (1999).
Cai, T. et al. Semen quality in patients with Chlamydia trachomatis genital infection treated concurrently with prulifloxacin and a phytotherapeutic agent. J. Androl. 33, 615–623 (2012).
Steiner, G. et al. Phenotype and function of peripheral and prostatic lymphocytes in patients with benign prostatic hyperplasia. J. Urol. 151, 480–484 (1994).
Gandaglia, G. et al. The role of chronic prostatic inflammation in the pathogenesis and progression of benign prostatic hyperplasia (BPH). BJU Int. 112, 432–441 (2013).
Shah, R., Mucci, N. R., Amin, A., Macoska, J. A. & Rubin, M. A. Postatrophic hyperplasia of the prostate gland: neoplastic precursor or innocent bystander? Am. J. Pathol. 158, 1767–1773 (2001).
Elkahwaji, J. E., Zhong, W., Hopkins, W. J. & Bushman, W. Chronic bacterial infection and inflammation incite reactive hyperplasia in a mouse model of chronic prostatitis. Prostate 67, 14–21 (2007).
Simons, B. W. et al. A human prostatic bacterial isolate alters the prostatic microenvironment and accelerates prostate cancer progression. J. Pathol. 235, 478–489 (2015).
Sampson, N. et al. The ageing male reproductive tract. J. Pathol. 211, 206–218 (2007).
Untergasser, G. et al. Proliferative disorders of the aging human prostate: involvement of protein hormones and their receptors. Exp. Gerontol. 34, 275–287 (1999).
Hoover, P. & Naz, R. K. Do men with prostate abnormalities (prostatitis/benign prostatic hyperplasia/prostate cancer) develop immunity to spermatozoa or seminal plasma? Int. J. Androl. 35, 608–615 (2012).
Author information
Authors and Affiliations
Contributions
P.V., T.C. and S.L. equally contributed to researching data for the article, discussion of content, writing and reviewing/editing of the manuscript under the coordination of the corresponding author.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Verze, P., Cai, T. & Lorenzetti, S. The role of the prostate in male fertility, health and disease. Nat Rev Urol 13, 379–386 (2016). https://doi.org/10.1038/nrurol.2016.89
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2016.89
This article is cited by
-
Long- and short-read RNA sequencing from five reproductive organs of boar
Scientific Data (2023)
-
SARS-CoV-2 and male infertility: from short- to long-term impacts
Journal of Endocrinological Investigation (2023)
-
Extracellular vesicles as a source of prostate cancer biomarkers in liquid biopsies: a decade of research
British Journal of Cancer (2022)
-
Developments in proton MR spectroscopic imaging of prostate cancer
Magnetic Resonance Materials in Physics, Biology and Medicine (2022)
-
Semen as a rich source of diagnostic biomarkers for prostate cancer: latest evidence and implications
Molecular and Cellular Biochemistry (2022)