Abstract
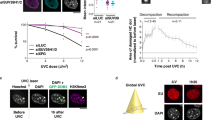
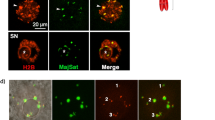
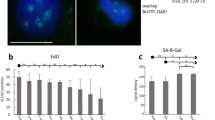
Eucaryotic cell nuclei contain a number of different organelles that are highly dynamic structures and respond to a variety of stimuli. Here we investigated the effect of UV irradiation on a recently identified group of organelles, Histone Locus Bodies. Histone Locus Bodies contain at least two main proteins, FLASH and NPAT, and have been shown to be involved in replication-dependent histone gene transcription. We show that these organelles are disrupted after sublethal irradiation and both FLASH and NPAT are degraded, which in turn results in cell-cycle arrest at the S/G2 transition. The effect on the cell cycle is due to reduced transcription of histone genes and restoring normal histone protein levels by stabilizing histone mRNA allows cells to progress through the cell cycle. This provides a novel mechanism of S-phase arrest in response to DNA damage that potentially allows DNA repair before cells continue into mitosis, and thus prevents transmission of genomic alterations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Abraham RT . (2001). Cell cycle checkpoint signaling through the ATM and ATR kinases. Genes Dev 15: 2177–2196.
Barcaroli D, Bongiorno-Borbone L, Terrinoni A, Hofmann TG, Rossi M, Knight RA et al. (2006a). FLASH is required for histone transcription and S-phase progression. Proc Natl Acad Sci USA 103: 14808–14812.
Barcaroli D, Dinsdale D, Neale MH, Bongiorno-Borbone L, Ranalli M, Munarriz E et al. (2006b). FLASH is an essential component of Cajal bodies. Proc Natl Acad Sci USA 103: 14802–14807.
Bartek J, Lukas C, Lukas J . (2004). Checking on DNA damage in S phase. Nat Rev Mol Cell Biol 5: 792–804.
Bongiorno-Borbone L, De Cola A, Vernole P, Finos L, Barcaroli D, Knight RA et al. (2008). FLASH and NPAT positive but not Coilin positive Cajal Bodies correlate with cell ploidy. Cell Cycle 7: 2357–2367.
Callegari AJ, Kelly TJ . (2007). Shedding light on the DNA damage checkpoint. Cell Cycle 6: 660–666.
Cioce M, Boulon S, Matera AG, Lamond AI . (2006). UV-induced fragmentation of Cajal bodies. J Cell Biol 175: 401–413.
Dellaire G, Bazett-Jones DP . (2007). Beyond repair foci: subnuclear domains and the cellular response to DNA damage. Cell Cycle 6: 1864–1872.
Galluzzi L, Aaronson SA, Abrams J, Alnemri ES, Andrews DW, Baehrecke EH et al. (2009). Guidelines for the use and interpretation of assays for monitoring cell death in higher eukaryotes. Cell Death Differ 16: 1093–1107.
Ghule PN, Dominski Z, Yang XC, Marzluff WF, Becker KA, Harper JW et al. (2008). Staged assembly of histone gene expression machinery at subnuclear foci in the abbreviated cell cycle of human embryonic stem cells. Proc Natl Acad Sci USA 105: 16964–16969.
Hall C, Nelson DM, Ye X, Baker K, DeCaprio JA, Seeholzer S et al. (2001). HIRA, the human homologue of yeast Hir1p and Hir2p, is a novel cyclin-cdk2 substrate whose expression blocks S-phase progression. Mol Cell Biol 21: 1854–1865.
Liu JL, Murphy C, Buszczak M, Clatterbuck S, Goodman R, Gall JG . (2006). The Drosophila melanogaster Cajal body. J Cell Biol 172: 875–884.
Ma T, Van Tine BA, Wei Y, Garrett MD, Nelson D, Adams PD et al. (2000). Cell cycle-regulated phosphorylation of p220(NPAT) by cyclin E/Cdk2 in Cajal bodies promotes histone gene transcription. Genes Dev 14: 2298–2313.
Marti TM, Hefner E, Feeney L, Natale V, Cleaver JE . (2006). H2AX phosphorylation within the G1 phase after UV irradiation depends on nucleotide excision repair and not DNA double-strand breaks. Proc Natl Acad Sci USA 103: 9891–9896.
Matera AG . (2006). Drosophila Cajal bodies: accessories not included. J Cell Biol 172: 791–793.
Munarriz E, Barcaroli D, Stephanou A, Townsend PA, Maisse C, Terrinoni A et al. (2004). PIAS-1 is a checkpoint regulator which affects exit from G1 and G2 by sumoylation of p73. Mol Cell Biol 24: 10593–10610.
Nelson DM, Ye X, Hall C, Santos H, Ma T, Kao GD et al. (2002). Coupling of DNA synthesis and histone synthesis in S phase independent of cyclin/cdk2 activity. Mol Cell Biol 22: 7459–7472.
Nicoletti I, Migliorati G, Pagliacci MC, Grignani F, Riccardi C . (1991). A rapid and simple method for measuring thymocyte apoptosis by propidium iodide staining and flow cytometry. J Immunol Methods 139: 271–279.
Nyberg KA, Michelson RJ, Putnam CW, Weinert TA . (2002). Toward maintaining the genome: DNA damage and replication checkpoints. Annu Rev Genet 36: 617–656.
Su C, Gao G, Schneider S, Helt C, Weiss C, O’Reilly MA et al. (2004). DNA damage induces downregulation of histone gene expression through the G1 checkpoint pathway. EMBO J 23: 1133–1143.
Wang A, Ikura T, Eto K, Ota MS . (2004). Dynamic interaction of p220(NPAT) and CBP/p300 promotes S-phase entry. Biochem Biophys Res Commun 325: 1509–1516.
Wei Y, Jin J, Harper JW . (2003). The cyclin E/Cdk2 substrate and Cajal body component p220(NPAT) activates histone transcription through a novel LisH-like domain. Mol Cell Biol 23: 3669–3680.
Ye X, Adams PD . (2003). Coordination of S-phase events and genome stability. Cell Cycle 2: 185–187.
Ye X, Wei Y, Nalepa G, Harper JW . (2003). The cyclin E/Cdk2 substrate p220(NPAT) is required for S-phase entry, histone gene expression, and Cajal body maintenance in human somatic cells. Mol Cell Biol 23: 8586–8600.
Zhao J . (2004). Coordination of DNA synthesis and histone gene expression during normal cell cycle progression and after DNA damage. Cell Cycle 3: 695–697.
Zhao J, Kennedy BK, Lawrence BD, Barbie DA, Matera AG, Fletcher JA et al. (2000). NPAT links cyclin E-Cdk2 to the regulation of replication-dependent histone gene transcription. Genes Dev 14: 2283–2297.
Acknowledgements
We thank Peter D Adams for the SLBP expression plasmid. The work was supported by grants from AIRC, MIUR to VDL, AIRC, EU-EPISTEM, FIRB, MIUR, MinSan, Telethon and Alleanza Contro il Cancro to GM. DB was supported by an FIRC scholarship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Rights and permissions
About this article
Cite this article
Bongiorno-Borbone, L., De Cola, A., Barcaroli, D. et al. FLASH degradation in response to UV-C results in histone locus bodies disruption and cell-cycle arrest. Oncogene 29, 802–810 (2010). https://doi.org/10.1038/onc.2009.388
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.388
Keywords
This article is cited by
-
Systems-wide RNAi analysis of CASP8AP2/FLASH shows transcriptional deregulation of the replication-dependent histone genes and extensive effects on the transcriptome of colorectal cancer cells
Molecular Cancer (2012)
-
FLASH is essential during early embryogenesis and cooperates with p73 to regulate histone gene transcription
Oncogene (2012)
-
PIAS1 interacts with FLASH and enhances its co-activation of c-Myb
Molecular Cancer (2011)