Abstract
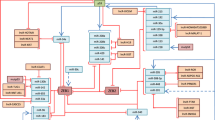
The epithelial–mesenchymal transition (EMT) is the pivotal mechanism underlying the initiation of cancer invasion and metastasis. Although Mel-18 has been implicated in several biological processes in cancer, its function in the EMT of human cancers has not yet been studied. Here, we demonstrate that Mel-18 negatively regulates the EMT by epigenetically modulating miR-205. We identified miR-205 as a novel target of Mel-18 using a microRNA microarray analysis and found that Mel-18 increased miR-205 transcription by the inhibition of DNA methyltransferase-mediated DNA methylation of the miR-205 promoter, thereby downregulating its target genes, ZEB1 and ZEB2. Furthermore, the loss of Mel-18 promoted ZEB1- and ZEB2-mediated downregulation of E-cadherin transcription and also enhanced the expression of mesenchymal markers, leading to increased migration and invasion in MCF-7 cells. In MDA-MB-231 cells, Mel-18 overexpression restored E-cadherin expression, resulting in reduced migration and invasion. These effects were reversed by miR-205 overexpression or inhibition. A tumor xenograft with Mel-18 knockdown MCF-7 cells consistently showed increased ZEB1 and ZEB2 expression and decreased E-cadherin expression. Taken together, these results suggest that Mel-18 functions as a tumor suppressor by its novel negative control of the EMT, achieved through regulating the expression of miR-205 and its target genes, ZEB1 and ZEB2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Kalluri R, Weinberg RA . The basics of epithelial-mesenchymal transition. J Clin Invest 2009; 119: 1420–1428.
Gupta PB, Chaffer CL, Weinberg RA . Cancer stem cells: mirage or reality? Nat Med 2009; 15: 1010–1012.
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 2008; 133: 704–715.
Scheel C, Weinberg RA . Cancer stem cells and epithelial-mesenchymal transition: concepts and molecular links. Semin Cancer Biol 2012; 22: 396–403.
Song LB, Li J, Liao WT, Feng Y, Yu CP, Hu LJ et al. The polycomb group protein Bmi-1 represses the tumor suppressor PTEN and induces epithelial-mesenchymal transition in human nasopharyngeal epithelial cells. J Clin Invest 2009; 119: 3626–3636.
McDonald OG, Wu H, Timp W, Doi A, Feinberg AP . Genome-scale epigenetic reprogramming during epithelial-to-mesenchymal transition. Nat Struct Mol Biol 2011; 18: 867–874.
Blumenberg M, Gao S, Dickman K, Grollman AP, Bottinger EP, Zavadil J . Chromatin structure regulation in transforming growth factor-beta-directed epithelial-mesenchymal transition. Cells Tissues Organs 2007; 185: 162–174.
Ke XS, Qu Y, Cheng Y, Li WC, Rotter V, Oyan AM et al. Global profiling of histone and DNA methylation reveals epigenetic-based regulation of gene expression during epithelial to mesenchymal transition in prostate cells. BMC Genomics 2010; 11: 669.
Korpal M, Lee ES, Hu G, Kang Y . The miR-200 family inhibits epithelial-mesenchymal transition and cancer cell migration by direct targeting of E-cadherin transcriptional repressors ZEB1 and ZEB2. J Biol Chem 2008; 283: 14910–14914.
Huang Q, Gumireddy K, Schrier M, le Sage C, Nagel R, Nair S et al. The microRNAs miR-373 and miR-520c promote tumour invasion and metastasis. Nat Cell Biol 2008; 10: 202–210.
Ma L, Teruya-Feldstein J, Weinberg RA . Tumour invasion and metastasis initiated by microRNA-10b in breast cancer. Nature 2007; 449: 682–688.
Tavazoie SF, Alarcon C, Oskarsson T, Padua D, Wang Q, Bos PD et al. Endogenous human microRNAs that suppress breast cancer metastasis. Nature 2008; 451: 147–152.
Tellez CS, Juri DE, Do K, Bernauer AM, Thomas CL, Damiani LA et al. EMT and stem cell-like properties associated with miR-205 and miR-200 epigenetic silencing are early manifestations during carcinogen-induced transformation of human lung epithelial cells. Cancer Res 2011; 71: 3087–3097.
Iorio MV, Piovan C, Croce CM . Interplay between microRNAs and the epigenetic machinery: an intricate network. Biochim Biophys Acta 2010; 1799: 694–701.
Valk-Lingbeek ME, Bruggeman SW, van Lohuizen M . Stem cells and cancer; the polycomb connection. Cell 2004; 118: 409–418.
Glinsky GV, Berezovska O, Glinskii AB . Microarray analysis identifies a death-from-cancer signature predicting therapy failure in patients with multiple types of cancer. J Clin Invest 2005; 115: 1503–1521.
Kim JH, Yoon SY, Jeong SH, Kim SY, Moon SK, Joo JH et al. Overexpression of Bmi-1 oncoprotein correlates with axillary lymph node metastases in invasive ductal breast cancer. Breast 2004; 13: 383–388.
Yang MH, Hsu DS, Wang HW, Wang HJ, Lan HY, Yang WH et al. Bmi1 is essential in Twist1-induced epithelial-mesenchymal transition. Nat Cell Biol 2010; 12: 982–992.
Kashyap V, Rezende NC, Scotland KB, Shaffer SM, Persson JL, Gudas LJ et al. Regulation of stem cell pluripotency and differentiation involves a mutual regulatory circuit of the NANOG, OCT4, and SOX2 pluripotency transcription factors with polycomb repressive complexes and stem cell microRNAs. Stem Cells Dev 2009; 18: 1093–1108.
Alkema MJ, Bronk M, Verhoeven E, Otte A, van ‘t Veer LJ, Berns A et al. Identification of Bmi1-interacting proteins as constituents of a multimeric mammalian polycomb complex. Genes Dev 1997; 11: 226–240.
Ringrose L, Paro R . Epigenetic regulation of cellular memory by the Polycomb and Trithorax group proteins. Annu Rev Genet 2004; 38: 413–443.
Guo WJ, Datta S, Band V, Dimri GP . Mel-18, a polycomb group protein, regulates cell proliferation and senescence via transcriptional repression of Bmi-1 and c-Myc oncoproteins. Mol Biol Cell 2007; 18: 536–546.
Lee JY, Jang KS, Shin DH, Oh MY, Kim HJ, Kim Y et al. Mel-18 negatively regulates INK4a/ARF-independent cell cycle progression via Akt inactivation in breast cancer. Cancer Res 2008; 68: 4201–4209.
Park JH, Lee JY, Shin DH, Jang KS, Kim HJ, Kong G . Loss of Mel-18 induces tumor angiogenesis through enhancing the activity and expression of HIF-1alpha mediated by the PTEN/PI3K/Akt pathway. Oncogene 2011; 30: 4578–4589.
Cao Q, Mani RS, Ateeq B, Dhanasekaran SM, Asangani IA, Prensner JR et al. Coordinated regulation of polycomb group complexes through microRNAs in cancer. Cancer Cell 2011; 20: 187–199.
Gregory PA, Bert AG, Paterson EL, Barry SC, Tsykin A, Farshid G et al. The miR-200 family and miR-205 regulate epithelial to mesenchymal transition by targeting ZEB1 and SIP1. Nat Cell Biol 2008; 10: 593–601.
Wiklund ED, Bramsen JB, Hulf T, Dyrskjot L, Ramanathan R, Hansen TB et al. Coordinated epigenetic repression of the miR-200 family and miR-205 in invasive bladder cancer. Int J Cancer 2011; 128: 1327–1334.
Mak P, Leav I, Pursell B, Bae D, Yang X, Taglienti CA et al. ERbeta impedes prostate cancer EMT by destabilizing HIF-1alpha and inhibiting VEGF-mediated snail nuclear localization: implications for Gleason grading. Cancer Cell 2010; 17: 319–332.
Wanami LS, Chen HY, Peiro S, Garcia de Herreros A, Bachelder RE . Vascular endothelial growth factor-A stimulates Snail expression in breast tumor cells: implications for tumor progression. Exp Cell Res 2008; 314: 2448–2453.
Yang AD, Camp ER, Fan F, Shen L, Gray MJ, Liu W et al. Vascular endothelial growth factor receptor-1 activation mediates epithelial to mesenchymal transition in human pancreatic carcinoma cells. Cancer Res 2006; 66: 46–51.
Blobe GC, Schiemann WP, Lodish HF . Role of transforming growth factor beta in human disease. N Engl J Med 2000; 342: 1350–1358.
Liu YN, Abou-Kheir W, Yin JJ, Fang L, Hynes P, Casey O et al. Critical and reciprocal regulation of KLF4 and SLUG in transforming growth factor beta-initiated prostate cancer epithelial-mesenchymal transition. Mol Cell Biol 2012; 32: 941–953.
Scheel C, Eaton EN, Li SH, Chaffer CL, Reinhardt F, Kah KJ et al. Paracrine and autocrine signals induce and maintain mesenchymal and stem cell states in the breast. Cell 2011; 145: 926–940.
Leung-Kuen AuS, Chak-Lui Wong C, Man-Fong Lee J, Ngo-Yin Fan D, Hoching Tsang F, Oi-Lin NgI et al. Enhancer of zeste homolog 2 (EZH2) epigenetically silences multiple tumor suppressor miRNAs to promote liver cancer metastasis. Hepatology 2012; 56: 622–631.
Yamagishi M, Nakano K, Miyake A, Yamochi T, Kagami Y, Tsutsumi A et al. Polycomb-mediated loss of miR-31 activates NIK-dependent NF-kappaB pathway in adult T cell leukemia and other cancers. Cancer Cell 2012; 21: 121–135.
Sempere LF, Christensen M, Silahtaroglu A, Bak M, Heath CV, Schwartz G et al. Altered microRNA expression confined to specific epithelial cell subpopulations in breast cancer. Cancer Res 2007; 67: 11612–11620.
Sato F, Tsuchiya S, Meltzer SJ, Shimizu K . MicroRNAs and epigenetics. FEBS J 2011; 278: 1598–1609.
Rush M, Appanah R, Lee S, Lam LL, Goyal P, Lorincz MC . Targeting of EZH2 to a defined genomic site is sufficient for recruitment of Dnmt3a but not de novo DNA methylation. Epigenetics 2009; 4: 404–414.
Vire E, Brenner C, Deplus R, Blanchon L, Fraga M, Didelot C et al. The Polycomb group protein EZH2 directly controls DNA methylation. Nature 2006; 439: 871–874.
Mohammad HP, Cai Y, McGarvey KM, Easwaran H, Van Neste L, Ohm JE et al. Polycomb CBX7 promotes initiation of heritable repression of genes frequently silenced with cancer-specific DNA hypermethylation. Cancer Res 2009; 69: 6322–6330.
Negishi M, Saraya A, Miyagi S, Nagao K, Inagaki Y, Nishikawa M et al. Bmi1 cooperates with Dnmt1-associated protein 1 in gene silencing. Biochem Biophys Res Commun 2007; 353: 992–998.
Guo WJ, Zeng MS, Yadav A, Song LB, Guo BH, Band V et al. Mel-18 acts as a tumor suppressor by repressing Bmi-1 expression and down-regulating Akt activity in breast cancer cells. Cancer Res 2007; 67: 5083–5089.
Qian T, Lee JY, Park JH, Kim HJ, Kong G . Id1 enhances RING1b E3 ubiquitin ligase activity through the Mel-18/Bmi-1 polycomb group complex. Oncogene 2010; 29: 5818–5827.
Shin DH, Jang SH, Kang BC, Kim HJ, Oh SH, Kong G . Constitutive overexpression of Id-1 in mammary glands of transgenic mice results in precocious and increased formation of terminal end buds, enhanced alveologenesis, delayed involution. J Cell Physiol 2011; 226: 1340–1352.
Acknowledgements
This study was supported by grants from the Korea Healthcare Technology R&D Project (A101836), the Ministry of Health Welfare and Family Affairs and the Research Program for New Drug Target Discovery (2011-0030173).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Lee, JY., Park, M., Park, JH. et al. Loss of the polycomb protein Mel-18 enhances the epithelial–mesenchymal transition by ZEB1 and ZEB2 expression through the downregulation of miR-205 in breast cancer. Oncogene 33, 1325–1335 (2014). https://doi.org/10.1038/onc.2013.53
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.53
Keywords
This article is cited by
-
microRNA-205 represses breast cancer metastasis by perturbing the rab coupling protein [RCP]-mediated integrin β1 recycling on the membrane
Apoptosis (2024)
-
Mechanisms of Polycomb group protein function in cancer
Cell Research (2022)
-
Interplay between p53 and non-coding RNAs in the regulation of EMT in breast cancer
Cell Death & Disease (2021)
-
The Eupentacta fraudatrix transcriptome provides insights into regulation of cell transdifferentiation
Scientific Reports (2020)
-
SNHG16 regulates invasion and migration of bladder cancer through induction of epithelial-to-mesenchymal transition
Human Cell (2020)