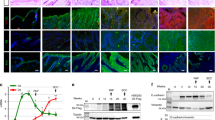
Abstract
There is increasing evidence that mast cells (MCs) and their mediators are involved in the remodeling of the tumor microenvironment and promote tumor growth, angiogenesis and metastasis. We have found that an increased density of MCs in thyroid cancer (TC) correlates with enhanced invasiveness. However, the MC-derived factors responsible for this activity and the mechanisms by which they enhance TC invasiveness remain unidentified. Here, we report that MCs, when activated by TC cells, produce soluble factors that induce epithelial-to-mesenchymal transition (EMT) and stemness features of TC cells. We identified CXCL8/interleukin (IL)-8 as the main mediator contained in activated MC conditioned media (CM) capable of inducing both EMT and stemness of TC cells. Mechanistically, MC CM or exogenous IL-8 stimulated Akt phosphorylation and Slug expression in TC cells. The inhibition of the Akt pathway or depletion of the Slug transcription factor by RNA interference, reverted EMT and stemness responses. TC cells stably transfected with exogenous IL-8 underwent EMT, displayed increased stemness and enhanced tumorigenicity with respect to control cells. The analysis of TC surgical specimens by immunohistochemical analysis demonstrated a positive correlation between MC density (Tryptase+ cells) and stemness features (OCT4 staining). Taken together, our data identify an MC-dependent IL-8–Akt–Slug pathway that sustains EMT/stemness of TC cells. The blockade of this circuit might be exploited for the therapy of advanced TC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Carling T, Udelsman R . Thyroid cancer. Annu Rev Med 2014; 65: 125–137.
Quail DF, Joyce JA . Microenvironmental regulation of tumor progression and metastasis. Nat Med 2013; 19: 1423–1437.
Ryder M, Ghossein RA, Ricarte-Filho JC, Knauf JA, Fagin JA . Increased density of tumor-associated macrophages is associated with decreased survival in advanced thyroid cancer. Endocr Relat Cancer 2008; 15: 1069–1074.
French JD, Kotnis GR, Said S, Raeburn CD, McIntyre RC Jr, Klopper JP et al. Programmed death-1+ T cells and regulatory T cells are enriched in tumor-involved lymph nodes and associated with aggressive features in papillary thyroid cancer. J Clin Endocrinol Metab 2012; 97: E934–E943.
Melillo RM, Guarino V, Avilla E, Galdiero MR, Liotti F, Prevete N et al. Mast cells have a protumorigenic role in human thyroid cancer. Oncogene 2010; 29: 6203–6215.
Kalesnikoff J, Galli SJ . New developments in mast cell biology. Nat Immunol 2008; 9: 1215–1223.
Dalton DK, Noelle RJ . The roles of mast cells in anticancer immunity. Cancer Immunol Immunother 2012; 61: 1511–1520.
Coussens LM, Raymond WW, Bergers G, Laig-Webster M, Behrendtsen O, Werb Z et al. Inflammatory mast cells up-regulate angiogenesis during squamous epithelial carcinogenesis. Genes Dev 1999; 13: 1382–1397.
Detoraki A, Staiano RI, Granata F, Giannattasio G, Prevete N, de Paulis A et al. Vascular endothelial growth factors synthesized by human lung mast cells exert angiogenic effects. J Allergy Clin Immunol 2009; 123: 1142–1149.
Wasiuk A, Dalton DK, Schpero WL, Stan RV, Conejo-Garcia JR, Noelle RJ . Mast cells impair the development of protective anti-tumor immunity. Cancer Immunol Immunother 2012; 61: 2273–2282.
Nowak EC, de Vries VC, Wasiuk A, Ahonen C, Bennett KA, Le Mercier I et al. Tryptophan hydroxylase-1 regulates immune tolerance and inflammation. J Exp Med 2012; 209: 2127–2135.
Kalluri R, Weinberg RA . The basics of epithelial-mesenchymal transition. J Clin Invest 2009; 119: 1420–1428.
Katsuno Y, Lamouille S, Derynck R . TGF-β signaling and epithelial-mesenchymal transition in cancer progression. Curr Opin Oncol 2013; 25: 76–84.
Kwon MJ . Emerging roles of claudins in human cancer. Int J Mol Sci 2013; 14: 18148–18180.
Scheel C, Weinberg RA . Cancer stem cells and epithelial-mesenchymal transition: concepts and molecular links. Semin Cancer Biol 2012; 22: 396–403.
Todaro M, Iovino F, Eterno V, Cammareri P, Gambara G, Espina V et al. Tumorigenic and metastatic activity of human thyroid cancer stem cells. Cancer Res 2010; 70: 8874–8885.
Asiedu MK, Ingle JN, Behrens MD, Radisky DC, Knutson KL . TGFbeta/TNF(alpha)-mediated epithelial-mesenchymal transition generates breast cancer stem cells with a claudin-low phenotype. Cancer Res 2011; 71: 4707–4719.
Cicalese A, Bonizzi G, Pasi CE, Faretta M, Ronzoni S, Giulini B et al. The tumor suppressor p53 regulates polarity of self-renewing divisions in mammary stem cells. Cell 2009; 138: 1083–1095.
Lengner CJ, Welstead GG, Jaenisch R . The pluripotency regulator Oct4: a role in somatic stem cells? Cell Cycle 2008; 7: 725–728.
Shi Q, Xiong Q, Le X, Xie K . Regulation of interleukin-8 expression by tumor-associated stress factors. J Interferon Cytokine Res 2001; 21: 553–566.
Hoffmann E, Dittrich-Breiholz O, Holtmann H, Kracht M . Multiple control of interleukin-8 gene expression. J Leukoc Biol 2002; 72: 847–855.
Waugh DJ, Wilson C . The interleukin-8 pathway in cancer. Clin Cancer Res 2008; 14: 6735–6741.
Campbell LM, Maxwell PJ, Waugh DJ . Rationale and means to target pro-inflammatory interleukin-8 (CXCL8) signaling in cancer. Pharmaceuticals 2013; 6: 929–959.
Palena C, Hamilton DH, Fernando RI . Influence of IL-8 on the epithelial-mesenchymal transition and the tumor microenvironment. Future Oncol 2012; 8: 713–722.
Alves CC, Carneiro F, Hoefler H, Becker KF . Role of the epithelial-mesenchymal transition regulator Slug in primary human cancers. Front Biosci 2009; 14: 3035–3050.
Hardy RG, Vicente-Dueñas C, González-Herrero I, Anderson C, Flores T, Hughes S et al. Snail family transcription factors are implicated in thyroid carcinogenesis. Am J Pathol 2007; 171: 1037–1046.
Buehler D, Hardin H, Shan W, Montemayor-Garcia C, Rush PS, Asioli S et al. Expression of epithelial-mesenchymal transition regulators SNAI2 and TWIST1 in thyroid carcinomas. Mod Pathol 2013; 26: 54–61.
Wang N, Dong CR, Jiang R, Tang C, Yang L, Jiang QF et al. Overexpression of HIF-1α, metallothionein and SLUG is associated with high TNM stage and lymph node metastasis in papillary thyroid carcinoma. Int J Clin Exp Pathol 2013; 7: 322–330.
Wei Q, Fang W, Ye L, Shen L, Zhang X, Fei X et al. Density of tumor associated macrophage correlates with lymph node metastasis in papillary thyroid carcinoma. Thyroid 2012; 22: 905–910.
Fang W, Ye L, Shen L, Cai J, Huang F, Wei Q et al. Tumor-associated macrophages promote the metastatic potential of thyroid papillary cancer by releasing CXCL8. Carcinogenesis 2014; 35: 1780–1787.
Ryder M, Gild M, Hohl TM, Pamer E, Knauf J, Ghossein R et al. Genetic and pharmacological targeting of CSF-1/CSF-1R inhibits tumor-associated macrophages and impairs BRAF-induced thyroid cancer progression. PLoS One 2013; 8: e54302.
Caillou B, Talbot M, Weyemi U, Pioche-Durieu C, Al Ghuzlan A, Bidart JM et al. Tumor-associated macrophages (TAMs) form an interconnected cellular supportive network in anaplastic thyroid carcinoma. PLoS One 2011; 6: e22567.
Phay JE, Ringel MD . Metastatic mechanisms in follicular cell-derived thyroid cancer. Endocr Relat Cancer 2013; 20: R307–R319.
Nehs MA, Nucera C, Nagarkatti SS, Sadow PM, Morales-Garcia D, Hodin RA et al. Late intervention with anti-BRAF(V600E) therapy induces tumor regression in an orthotopic mouse model of human anaplastic thyroid cancer. Endocrinology 2012; 153: 985–994.
Shaik S, Nucera C, Inuzuka H, Gao D, Garnaas M, Frechette G et al. SCF(β-TRCP) suppresses angiogenesis and thyroid cancer cell migration by promoting ubiquitination and destruction of VEGF receptor 2. J Exp Med 2012; 209: 1289–1307.
Ginestier C, Liu S, Diebel ME, Korkaya H, Luo M, Brown M et al. CXCR1 blockade selectively targets human breast cancer stem cells in vitro and in xenografts. J Clin Invest 2010; 120: 485–497.
Singh JK, Farnie G, Bundred NJ, Simões BM, Shergill A, Landberg G et al. Targeting CXCR1/2 significantly reduces breast cancer stem cell activity and increases the efficacy of inhibiting HER2 via HER2-dependent and -independent mechanisms. Clin Cancer Res 2013; 19: 643–656.
Avilla E, Guarino V, Visciano C, Liotti F, Svelto M, Krishnamoorthy G et al. Activation of TYRO3/AXL tyrosine kinase receptors in thyroid cancer. Cancer Res 2011; 71: 1792–1804.
Salerno P, Garcia-Rostan G, Piccinin S, Bencivenga TC, Di Maro G, Doglioni C et al. TWIST1 plays a pleiotropic role in determining the anaplastic thyroid cancer phenotype. J Clin Endocrinol Metab 2011; 96: E772–E781.
Acknowledgements
We thank A Cerrato (IEOS, CNR, Naples) for helpful discussions and technical suggestions. We thank S Sequino for assistance in animal maintenance. We are grateful to PB Gupta (Whitehead Institute for Biomedical Research and Massachusetts Institute of Technology, Cambridge, MA, USA) for shSlug plasmids, to M Kajita (Department of Pathology and Laboratory Medicine, Winship Cancer Institute, Atlanta) for the Slug myc plasmid and to BP Zhou (Department of Molecular and Cellular Oncology, Breast Cancer Basic Research Program, The University of Texas M. D. Anderson Cancer Center, Houston, Texas) for the FLAG Snail wt plasmid. Associazione Italiana per la Ricerca sul Cancro (AIRC) IG grant 11827 ‘Thyroid cancer and inflammation’; Italian Health Ministry grant RF-CAM-353005 ‘Molecular diagnostic and prognostic markers of thyroid neoplasis’; Istituto Superiore di Oncologia grant (MIUR PON01_02782/12); Ministero della Salute (Ricerca Finalizzata 2009–IZSM RF 2009), Regione Campania CISI-Lab Project, CRÈME Project and TIMING Project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Visciano, C., Liotti, F., Prevete, N. et al. Mast cells induce epithelial-to-mesenchymal transition and stem cell features in human thyroid cancer cells through an IL-8–Akt–Slug pathway. Oncogene 34, 5175–5186 (2015). https://doi.org/10.1038/onc.2014.441
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.441
This article is cited by
-
The lncRNA RMST is drastically downregulated in anaplastic thyroid carcinomas where exerts a tumor suppressor activity impairing epithelial-mesenchymal transition and stemness
Cell Death Discovery (2023)
-
NEDD9 links anaplastic thyroid cancer stemness to chromosomal instability through integrated centrosome asymmetry and DNA sensing regulation
Oncogene (2022)
-
Dietary AGEs involvement in colonic inflammation and cancer: insights from an in vitro enterocyte model
Scientific Reports (2020)
-
Hypoxic colorectal cancer cells promote metastasis of normoxic cancer cells depending on IL-8/p65 signaling pathway
Cell Death & Disease (2020)
-
Patterns of immune infiltration in lung adenocarcinoma revealed a prognosis-associated microRNA–mast cells network
Human Cell (2020)