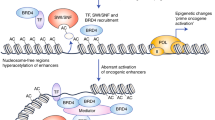
Abstract
Bromodomain and extraterminal (BET) domain containing protein (BRD)-4 modulates the expression of oncogenes such as c-myc, and is a promising therapeutic target in diverse cancer types. We performed pre-clinical studies in myeloma models with bi-functional protein-targeting chimeric molecules (PROTACs) which target BRD4 and other BET family members for ubiquitination and proteasomal degradation. PROTACs potently reduced the viability of myeloma cell lines in a time-dependent and concentration-dependent manner associated with G0/G1 arrest, reduced levels of CDKs 4 and 6, increased p21 levels, and induction of apoptosis. These agents specifically decreased cellular levels of downstream BRD4 targets, including c-MYC and N-MYC, and a Cereblon-targeting PROTAC showed downstream effects similar to those of an immunomodulatory agent. Notably, PROTACs overcame bortezomib, dexamethasone, lenalidomide, and pomalidomide resistance, and their activity was maintained in otherwise isogenic myeloma cells with wild-type or deleted TP53. Combination studies showed synergistic interactions with dexamethasone, BH3 mimetics, and Akt pathway inhibitors. BET-specific PROTACs induced a rapid loss of viability of primary cells from myeloma patients, and delayed growth of MM1.S-based xenografts. Our data demonstrate that BET degraders have promising activity against pre-clinical models of multiple myeloma, and support their translation to the clinic for patients with relapsed and/or refractory disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Ott G. Impact of MYC on malignant behavior. Hematology. 2014;2014:100–6.
Horiuchi D, Anderton B, Goga A. Taking on challenging targets: making MYC druggable. Am Soc Clin Oncol Meet. 2014;e497–502.
Nishida K, Tamura A, Nakazawa N, Ueda Y, Abe T, Matsuda F, et al. The Ig heavy chain gene is frequently involved in chromosomal translocations in multiple myeloma and plasma cell leukemia as detected by in situ hybridization. Blood. 1997;90:526–34.
Shou Y, Martelli ML, Gabrea A, Qi Y, Brents LA, Roschke A, et al. Diverse karyotypic abnormalities of the c-myc locus associated with c-myc dysregulation and tumor progression in multiple myeloma. Proc Natl Acad Sci USA. 2000;97:228–33.
Affer M, Chesi M, Chen WD, Keats JJ, Demchenko YN, Tamizhmani K, et al. Promiscuous MYC locus rearrangements hijack enhancers but mostly super-enhancers to dysregulate MYC expression in multiple myeloma. Leukemia. 2014;28:1725–35.
Walker BA, Wardell CP, Brioli A, Boyle E, Kaiser MF, Begum DB, et al. Translocations at 8q24 juxtapose MYC with genes that harbor superenhancers resulting in overexpression and poor prognosis in myeloma patients. Blood Cancer J. 2014;4:e191.
Szabo AG, Gang AO, Pedersen MO, Poulsen TS, Klausen TW, Norgaard P. Overexpression of c-myc is associated with adverse clinical features and worse overall survival in multiple myeloma. Leuk Lymphoma. 2016;57:2526–34.
Sekiguchi N, Ootsubo K, Wagatsuma M, Midorikawa K, Nagata A, Noto S, et al. Impact of C-Myc gene-related aberrations in newly diagnosed myeloma with bortezomib/dexamethasone therapy. Int J Hematol. 2014;99:288–95.
Glitza IC, Lu G, Shah R, Bashir Q, Shah N, Champlin RE, et al. Chromosome 8q24.1/c-MYC abnormality: a marker for high-risk myeloma. Leuk Lymphoma. 2015;56:602–7.
Greco C, D’Agnano I, Vitelli G, Vona R, Marino M, Mottolese M, et al. c-MYC deregulation is involved in melphalan resistance of multiple myeloma: role of PDGF-BB. Int J Immunopathol Pharmacol. 2006;19:67–79.
Sonneveld P, Avet-Loiseau H, Lonial S, Usmani S, Siegel D, Anderson KC, et al. Treatment of multiple myeloma with high-risk cytogenetics: a consensus of the International Myeloma Working Group. Blood. 2016;127:2955–62.
Filippakopoulos P, Qi J, Picaud S, Shen Y, Smith WB, Fedorov O, et al. Selective inhibition of BET bromodomains. Nature. 2010;468:1067–73.
Delmore JE, Issa GC, Lemieux ME, Rahl PB, Shi J, Jacobs HM, et al. BET bromodomain inhibition as a therapeutic strategy to target c-Myc. Cell. 2011;146:904–17.
Fu LL, Tian M, Li X, Li JJ, Huang J, Ouyang L, et al. Inhibition of BET bromodomains as a therapeutic strategy for cancer drug discovery. Oncotarget. 2015;6:5501–16.
Sakamoto KM, Kim KB, Kumagai A, Mercurio F, Crews CM, Deshaies RJ. Protacs: chimeric molecules that target proteins to the Skp1-Cullin-F box complex for ubiquitination and degradation. Proc Natl Acad Sci USA. 2001;98:8554–9.
Sakamoto KM, Kim KB, Verma R, Ransick A, Stein B, Crews CM, et al. Development of Protacs to target cancer-promoting proteins for ubiquitination and degradation. Mol Cell Proteom. 2003;2:1350–8.
Ito T, Ando H, Suzuki T, Ogura T, Hotta K, Imamura Y, et al. Identification of a primary target of thalidomide teratogenicity. Science. 2010;327:1345–50.
Zhu YX, Braggio E, Shi CX, Bruins LA, Schmidt JE, Van Wier S, et al. Cereblon expression is required for the antimyeloma activity of lenalidomide and pomalidomide. Blood. 2012;118:4771–9.
Winter GE, Buckley DL, Paulk J, Roberts JM, Souza A, Dhe-Paganon S, et al. Drug development. Phthalimide conjugation as a strategy for in vivo target protein degradation. Science. 2015;348:1376–81.
Lu J, Qian Y, Altieri M, Dong H, Wang J, Raina K, et al. Hijacking the E3 ubiquitin ligase cereblon to efficiently target BRD4. Chem Biol. 2015;22:755–63.
Raina K, Lu J, Qian Y, Altieri M, Gordon D, Rossi AM, et al. PROTAC-induced BET protein degradation as a therapy for castration-resistant prostate cancer. Proc Natl Acad Sci USA. 2016;113:7124–9.
Lee HC, Wang H, Baladandayuthapani V, Lin H, He J, Jones RJ, et al. RNA polymerase I inhibition with CX-5461 as a novel therapeutic strategy to target MYC in multiple myeloma. Br J Haematol. 2017;177:80–94.
Li B, Fu J, Chen P, Ge X, Li Y, Kuiatse I, et al. The nuclear factor (erythroid-derived 2)-like 2 and proteasome maturation protein axis mediate bortezomib resistance in multiple myeloma. J Biol Chem. 2015;290:29854–68.
Kuhn DJ, Berkova Z, Jones RJ, Woessner R, Bjorklund CC, Ma W, et al. Targeting the insulin-like growth factor-1 receptor to overcome bortezomib resistance in preclinical models of multiple myeloma. Blood. 2012;120:3260–70.
Bjorklund CC, Ma W, Wang ZQ, Davis RE, Kuhn DJ, Kornblau SM, et al. Evidence of a role for activation of Wnt/beta-catenin signaling in the resistance of plasma cells to lenalidomide. J Biol Chem. 2011;286:11009–20.
Tibes R, Qiu Y, Lu Y, Hennessy B, Andreeff M, Mills GB, et al. Reverse phase protein array: validation of a novel proteomic technology and utility for analysis of primary leukemia specimens and hematopoietic stem cells. Mol Cancer Ther. 2006;5:2512–21.
Woolson RF, Clarke WR. Statistical methods for the analysis of biomedical data. 2nd edn. New York, NY: Wiley; 2002.
Dib A, Gabrea A, Glebov OK, Bergsagel PL, Kuehl WM. Characterization of MYC translocations in multiple myeloma cell lines. J Natl Cancer Inst Monogr. 2008;39:25–31.
Wyce A, Ganji G, Smitheman KN, Chung CW, Korenchuk S, Bai Y, et al. BET inhibition silences expression of MYCN and BCL2 and induces cytotoxicity in neuroblastoma tumor models. PLoS One. 2013;8:e72967.
Ezoe S, Matsumura I, Nakata S, Gale K, Ishihara K, Minegishi N, et al. GATA-2/estrogen receptor chimera regulates cytokine-dependent growth of hematopoietic cells through accumulation ofp21(WAF1) and p27(Kip1) proteins. Blood. 2002;100:3512–20.
Zhang Y, Wang Z, Li X, Magnuson NS. Pim kinase-dependent inhibition of c-Myc degradation. Oncogene. 2008;27:4809–19.
Manasanch EE, Orlowski RZ. Proteasome inhibitors in cancer therapy. Nat Rev Clin Oncol. 2017;14:417–33.
Chang H, Liu Y, Xue M, Liu H, Du S, Zhang L, et al. Synergistic action of master transcription factors controls epithelial-to-mesenchymal transition. Nucleic Acids Res. 2016;44:2514–27.
Qi J. Bromodomain and extraterminal domain inhibitors (BETi) for cancer therapy: chemical modulation of chromatin structure. Cold Spring Harb Perspect Biol. 2014;6:a018663.
Stratikopoulos EE, Dendy M, Szabolcs M, Khaykin AJ, Lefebvre C, Zhou MM, et al. Kinase and BET inhibitors together clamp inhibition of PI3K signaling and overcome resistance to therapy. Cancer Cell. 2015;27:837–51.
Gunther A, Baumann P, Burger R, Kellner C, Klapper W, Schmidmaier R, et al. Activity of everolimus (RAD001) in relapsed and/or refractory multiple myeloma: a phase I study. Haematologica. 2015;100:541–7.
Spencer A, Yoon SS, Harrison SJ, Morris SR, Smith DA, Brigandi RA, et al. The novel AKT inhibitor afuresertib shows favorable safety, pharmacokinetics, and clinical activity in multiple myeloma. Blood. 2014;124:2190–5.
Wu SY, Lee AY, Lai HT, Zhang H, Chiang CM. Phospho switch triggers Brd4 chromatin binding and activator recruitment for gene-specific targeting. Mol Cell. 2013;49:843–57.
Lee HC, Wang H, Baladandayuthapani V, Lin H, He J, Jones RJ, et al. RNA polymerase I inhibition with CX-5461 as a novel therapeutic strategy to target MYC in multiple myeloma. Br J Haematol. 2016;177:80–94.
Kortum KM, Zhu YX, Shi CX, Jedlowski P, Stewart AK. Cereblon binding molecules in multiple myeloma. Blood Rev. 2015;29:329–34.
Bellamy WT, Dalton WS, Kailey JM, Gleason MC, McCloskey TM, Dorr RT, et al. Verapamil reversal of doxorubicin resistance in multidrug-resistant human myeloma cells and association with drug accumulation and DNA damage. Cancer Res. 1988;48:6365–70.
Ito T, Handa H. Cereblon and its downstream substrates as molecular targets of immunomodulatory drugs. Int J Hematol. 2016;104:293–9.
Hao F, Itoh T, Morita E, Shirahama-Noda K, Yoshimori T, Noda T. The PtdIns3-phosphatase MTMR3 interacts with mTORC1 and suppresses its activity. FEBS Lett. 2016;590:161–73.
Kakumoto K, Ikeda J, Okada M, Morii E, Oneyama C. mLST8 promotes mTOR-mediated tumor progression. PLoS One. 2015;10:e0119015.
Zhu J, Blenis J, Yuan J. Activation of PI3K/Akt and MAPK pathways regulates Myc-mediated transcription by phosphorylating and promoting the degradation of Mad1. Proc Natl Acad Sci USA. 2008;105:6584–9.
Han K, Xu X, Xu Z, Chen G, Zeng Y, Zhang Z, et al. SC06, a novel small molecule compound, displays preclinical activity against multiple myeloma by disrupting the mTOR signaling pathway. Sci Rep. 2015;5:12809.
Bendell JC, Kelley RK, Shih KC, Grabowsky JA, Bergsland E, Jones S, et al. A phase I dose-escalation study to assess safety, tolerability, pharmacokinetics, and preliminary efficacy of the dual mTORC1/mTORC2 kinase inhibitor CC-223 in patients with advanced solid tumors or multiple myeloma. Cancer. 2015;121:3481–90.
Arcaro A. Targeting PI3K/mTOR signaling in cancer. Front Oncol. 2014;4:84.
Hutter G, Zimmermann Y, Rieken M, Hartmann E, Rosenwald A, Hiddemann W, et al. Proteasome inhibition leads to dephosphorylation and downregulation of protein expression of members of the Akt/mTOR pathway in MCL. Leukemia. 2012;26:2442–4.
Younes H, Leleu X, Hatjiharissi E, Moreau AS, Hideshima T, Richardson P, et al. Targeting the phosphatidylinositol 3-kinase pathway in multiple myeloma. Clin Cancer Res. 2007;13:3771–5.
Pourdehnad M, Truitt ML, Siddiqi IN, Ducker GS, Shokat KM, Ruggero D. Myc and mTOR converge on a common node in protein synthesis control that confers synthetic lethality in Myc-driven cancers. Proc Natl Acad Sci USA. 2013;110:11988–93.
Laplante M, Sabatini DM. mTOR signaling in growth control and disease. Cell. 2012;149:274–93.
Wang L, Harris TE, Roth RA, Lawrence JC Jr. PRAS40 regulates mTORC1 kinase activity by functioning as a direct inhibitor of substrate binding. J Biol Chem. 2007;282:20036–44.
Wiza C, Nascimento EB, Ouwens DM. Role of PRAS40 in Akt and mTOR signaling in health and disease. Am J Physiol Endocrinol Metab. 2012;302:E1453–1460.
Lu J, Zavorotinskaya T, Dai Y, Niu XH, Castillo J, Sim J, et al. Pim2 is required for maintaining multiple myeloma cell growth through modulating TSC2 phosphorylation. Blood. 2013;122:1610–20.
Sonneveld P, Lokhorst HM, Vossebeld P. Drug resistance in multiple myeloma. Semin Hematol. 1997;34:34–39.
Buda G, Ricci D, Huang CC, Favis R, Cohen N, Zhuang SH, et al. Polymorphisms in the multiple drug resistance protein 1 and in P-glycoprotein 1 are associated with time to event outcomes in patients with advanced multiple myeloma treated with bortezomib and pegylated liposomal doxorubicin. Ann Hematol. 2010;89:1133–40.
Hawley TS, Riz I, Yang W, Wakabayashi Y, Depalma L, Chang YT, et al. Identification of an ABCB1 (P-glycoprotein)-positive carfilzomib-resistant myeloma subpopulation by the pluripotent stem cell fluorescent dye CDy1. Am J Hematol. 2013;88:265–72.
Zhou W, Yang Y, Xia J, Wang H, Salama ME, Xiong W, et al. NEK2 induces drug resistance mainly through activation of efflux drug pumps and is associated with poor prognosis in myeloma and other cancers. Cancer Cell. 2013;23:48–62.
Amorim S, Stathis A, Gleeson M, Iyengar S, Magarotto V, Leleu X, et al. Bromodomain inhibitor OTX015 in patients with lymphoma or multiple myeloma: a dose-escalation, open-label, pharmacokinetic, phase 1 study. Lancet Haematol. 2016;3:e196–204.
Acknowledgements
This work was supported by funding from The MD Anderson Cancer Center Knowledge Gaps program, The MD Anderson Cancer Center Moon Shot in High Risk Multiple Myeloma, the National Cancer Institute (The MD Anderson Cancer Center SPORE in Multiple Myeloma (P50 CA142509) and R01s CA184464 and CA194264), the Leukemia & Lymphoma Society (New Idea Award 8994-12 and Specialized Center of Research SCOR-12206-17), the National Natural Science Foundation of China (Grant 81600170), and a Jiangsu government scholarship for overseas studies (JS-2014-195). The authors would like to thank the MD Anderson Flow Cytometry and Cellular Imaging Core Facility and the Characterized Cell Line Core Facility, which are supported by the Cancer Center Support Grant (P30 CA16672). R.Z.O. would also like to acknowledge support from the Florence Maude Thomas Cancer Research Professorship, the Brock Family Myeloma Research Fund, and the Jean Clarke High-risk Myeloma Research Fund.
Author information
Authors and Affiliations
Contributions
XZ planned and conducted most of the experiments, analyzed the results, and drafted the manuscript. HCL planned and conducted experiments, analyzed results, and revised the manuscript. FS planned and conducted experiments and analyzed results. VB and HL performed statistical analyses and interpreted the data. IK, FS and YH performed animal experiments, while RJJ, JL, and YQ participated in the design and analysis of experiments. SKT and HCL provided primary samples, BL and HW with HCL provided cell lines, while RED performed gene expression analysis. RKS performed immunohistochemistry and immunofluorescence studies. ZB helped analyze the data and draft the manuscript. KR and KGC reviewed the data and the manuscript. CMC helped develop the PROTACs. RZO conceived of the line of investigation, designed the experiments, analyzed the results, and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
RZO has served on advisory boards for Amgen, which manufactures and distributes carfilzomib, Takeda Pharmaceuticals USA, Inc., which manufactures and distributes bortezomib, and for Celgene Corporation, which manufactures and distributes lenalidomide and pomalidomide, but there was no commercial support for this research. JL, YQ, KR, and KGC are employees of Arvinas, LLC, which manufactures the PROTACs studied in this work. CMC is founder, consultant and shareholder of Arvinas, LLC. In addition, his lab receives research funding from Arvinas, LLC. The remaining authors have no conflicts of interest to declare.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Zhang, X., Lee, H.C., Shirazi, F. et al. Protein targeting chimeric molecules specific for bromodomain and extra-terminal motif family proteins are active against pre-clinical models of multiple myeloma. Leukemia 32, 2224–2239 (2018). https://doi.org/10.1038/s41375-018-0044-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41375-018-0044-x
This article is cited by
-
Enhancer rewiring in tumors: an opportunity for therapeutic intervention
Oncogene (2021)
-
The Role of the Ubiquitin Proteasome System in Glioma: Analysis Emphasizing the Main Molecular Players and Therapeutic Strategies Identified in Glioblastoma Multiforme
Molecular Neurobiology (2021)
-
Proteolysis-targeting chimera (PROTAC) for targeted protein degradation and cancer therapy
Journal of Hematology & Oncology (2020)
-
Proteolysis targeting chimeras (PROTACs) are emerging therapeutics for hematologic malignancies
Journal of Hematology & Oncology (2020)
-
Discovering and validating cancer genetic dependencies: approaches and pitfalls
Nature Reviews Genetics (2020)