Abstract
Background/objectives
Retinopathy of prematurity (ROP) is a severe neonatal complication potentially leading to visual impairment and blindness. Known risk factors include preterm birth, low birth weight and respiratory support. Limited and contradictory data exist on the risk of maternal smoking during pregnancy on the development of ROP. This study aims to investigate smoking as an independent risk factor for the development of severe ROP (≥stage 3).
Subjects/methods
This is a single centre retrospective case-control study of prospectively collected clinical data of infants born before 32 weeks of gestation between 2001 and 2012 at a tertiary care university hospital. The association between maternal smoking during pregnancy and the development of severe ROP was analyzed by multivariate logistic regression.
Results
In total, n = 751 infants born < 32 weeks of gestation were included in this study. In total, 52.9% (n = 397) were diagnosed with ROP and 10.8% (n = 81) developed ROP ≥ stage 3. In total, 8.4% (n = 63) mothers presented with a history of smoking during pregnancy, which was associated to a higher rate of ROP (OR 2.59, 95% CI 1.10–6.12). Low gestational age, low birth weight and prolonged respiratory support were confirmed as independent risk factors for the development of severe ROP.
Conclusions
To date, this is the largest study evaluating the effect of maternal smoking on the development of ROP. Maternal smoking during pregnancy is identified as an independent risk factor for the development of severe ROP in preterm infants born < 32 weeks of gestation.
Similar content being viewed by others
Introduction
Preterm birth is the leading determinant of neonatal morbidity and mortality and affects about 11% of births worldwide corresponding to 15 million babies each year [1, 2]. Despite a global decline in neonatal mortality (from 93 deaths per 1000 live births in 1990 to 41 deaths per 1000 live births in 2016), preterm birth is still responsible for 16% of all deaths of children under the age of 5 years [1]. Further, very preterm and extremely preterm birth in particular are highly associated not only with short term (e.g., respiratory distress syndrome, necrotizing enterocolitis, intraventricular haemorrhage and sepsis) but especially long-term infant morbidities such as cognitive developmental delays, hearing and visual impairment [2].
Retinopathy of prematurity (ROP) is one of these severe neonatal complications with dramatic long-term health consequences and a risk of visual impairment and blindness [3, 4]. With advancing neonatal care the number of infants surviving prematurity increases and with this the absolute number of neonates at risk for ROP [5]. In 2010 alone, about 20,000 of preterm infants became blind or severely visually impaired as a consequence of ROP [6]. Established risk factors for the development of ROP include preterm birth and low birth weight [5, 7]. Further, the use of supplemental oxygen, oxygen concentration, duration and prolonged mechanical ventilation were associated with severe ROP [5]. Potential risk factors with conflicting results in clinical trials include maternal age [8,9,10], mode of delivery [11,12,13,14], gender [7, 12], multiple pregnancies [5, 7] and Apgar Score [5, 12]. In addition, there is no consensus, whether maternal smoking during pregnancy leads to higher rates of severe ROP [5, 15,16,17]. Exposure to tobacco smoke during pregnancy has been associated with stillbirth, preterm birth and adverse perinatal outcome such as low birth weight, placenta abruption, congenital anomalies, bronchopulmonary dysplasia and severe intraventricular haemorrhage [18,19,20,21,22]. The pathogenesis of ROP is characterized by two successive steps: Initial high oxygen supply suppresses VEGF synthesis and blocks retinal vascularization [23]. During phase two, the impaired vascularization is compensated by excessive VEGF synthesis which results in uncontrolled neovascularization of physiologically avascular areas of the retina [24]. In vitro studies indicate an upregulation of vascular endothelial growth factor (VEGF) by nicotine, which might promote endothelial cellular proliferation und contribute to neovascularization, as seen in ROP [25, 26].
The aim of the current study was to investigate maternal smoking during pregnancy as an independent risk factor for the development of severe ROP in infants born < 32 weeks of gestation.
Material and methods
Study design
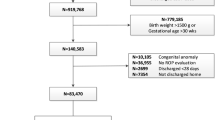
All infants born before 32 weeks of gestation (≤31 + 6/7) between 2001 and 2012 at Heidelberg University Hospital were assessed for eligibility in this retrospective case-control study of prospectively collected clinical data. Smoking during pregnancy was recorded at admission to the Department of Obstetrics. Maternal age, number of pregnancies (gravida) and deliveries (para), maternal body mass index (BMI) before pregnancy, antenatal treatment with glucocorticoids, smoking during pregnancy and the average number of cigarettes per day, multiple gestation, mode of delivery, gestational age, birth weight, gender, APGAR score (5 min) and respiratory support (mechanical ventilation and continuous airway pressure, CPAP, in hours) were assessed as potential risk factors for the development of ROP.
ROP screening
ROP screening was performed by successive dilated eye exams until the retina was fully vascularized and classified per the 2005 International Classification of Retinopathy of Prematurity [27]. ROP stages in this study are defined as the maximal stage in either eye and retinal zone. ROP stage 3 and greater was considered severe.
Statistics
SAS Version 9.4 was used for statistical analysis. Findings were reported using the mean and standard deviation for continuous data and absolute and relative frequencies for categorical variables. Student’s t test and Chi-square test were applied for univariate analysis depending on underlying distribution. Multivariate regression analysis was performed by multivariate binary logistic regression. For multivariate analyses, maximum likelihood estimates, odd ratios and Wald confidence intervals are presented. A p value < 0.05 was considered statistically significant. Individual tests are mentioned in the corresponding table legends.
Results
A total of n = 751 neonates < 32 weeks of gestation were included in this study (Table 1). In total, 397 infants developed ROP of which n = 81 developed severe ROP (≥stage 3) (Table 2). In total, n = 63 (8.3%) mothers indicated at admission to the Department of Obstetrics that they had been smoking during pregnancy, with a median of 10 cigarettes per day. Amongst established risk factors, univariate analysis revealed a significantly higher percentage of smoking mothers (17%) of infants who developed severe ROP compared with smoking mothers of infants who did not develop severe ROP (7%) (Table 1). Frequency distribution of ROP stages differed between infants who were or were not exposed to cigarette smoke during pregnancy (Table 2, p = 0.0115). More specifically, 22% of infants born to smoking mothers developed ROP ≥ stage 3, compared with only 10% in mothers who did not smoke (Table 2). Within the subgroup of smoking mothers, we found no difference in the number of cigarettes smoked between infants with severe ROP (cigarettes per day, median 10; min 1/max 30) and infants without severe ROP (median 10; min 1/max: 38) (p = 0.9326).
Multivariate analysis revealed smoking during pregnancy to be an independent risk factor for the development of severe ROP (Table 3, OR 2.591, 95% CI 1.097–6.122). Furthermore, known risk factors for the development of ROP like low birth weight (per additional kg: OR 0.079, 95% CI 0.013–0.486), low gestational age (per additional day: OR 0.961, 95% CI 0.931–0.991) and prolonged respiratory support (per additional hour: OR 1.001, 95% CI 1.000–1.001) were confirmed by multivariate analysis (Table 3).
Discussion
Maternal smoking during pregnancy is a risk factor for preterm birth and a determinant of adverse neonatal outcome [18, 28]. Very preterm infants are at high risk to develop severe ROP (>stage 3), which may lead to visual impairment and blindness [5]. The current study therefore focusses on this high-risk population. Maternal smoking during pregnancy has been associated to the development of ROP [5, 15, 17], however small study populations have yielded conflicting results. A recent review by Kim et al. on risk factors for ROP highlighted the need to conduct further studies that help clarify these uncertainties [5]. We now report maternal smoking during pregnancy to be an independent risk factor for the development of severe ROP.
The prevalence of maternal smoking during pregnancy depends on the social, cultural and ethnical background [29]. In our study population, the prevalence of smoking was 8.4%, which is lower than estimations for high-income countries (10–20%) and within the range of low- and middle-income countries (0–15%) [22, 30]. The lower-than-expected fraction of smokers in our study is most likely partially due to under-reporting, as this has been described for studies relying on self-reporting of maternal smoking [31].
There is no known single mechanism by which smoking may promote ROP development. Maternal smoking can cause nicotine-induced vasoconstriction, carbon monoxide-induced foetal hypoxia, cadmium disruption of calcium signalling, altered steroid hormone production, disruption of prostaglandin synthesis and changed responses to oxytocin, as described by Ion and Bernal [21]. Through these mechanisms, uterine blood flow may be impaired leading to intrauterine growth restriction of the foetus and preterm birth [21, 29]. In addition, smoking is associated with a higher risk of preterm rupture of the membrane, incompetent cervix and chorioamnionitis [20], which in turn has been suggested to increase the risk of developing ROP [32, 33]. Animal models indicate a dose-dependent effect of gestational nicotine exposure on histopathological parameters of the developing retina [34]. However, our study found no dose effect relationship for the number of cigarettes per day and the development of ROP. It should be noted that no data was available as to when during pregnancy mothers were smoking or whether they were still smoking after delivery, which might also factor into the development of ROP.
Contrary to our findings, Hirabayashi et al. propose inhibitory effects of maternal smoking on the development of severe ROP based on a study population of 86 very preterm infants [15]. At such low sample sizes (out of 27 infants that developed severe ROP, only one single woman smoked) conclusions need to be drawn with caution, as highlighted by Ram and McDonald for this particular study [35]. Further, as most studies on risk factors for ROP rely on retrospective clinical data, methodological differences in diagnosing ROP stages by ophthalmologists have to be taken into consideration as confounding factors [5, 36, 37]. There are many additional reasons why results from clinical studies may differ, including heterogeneity of the study population regarding ethnicity, differences in baseline patient characteristics and clinical neonatal care protocols. For example, a recent study by Yusuf et al. did not find an association of ROP stage ≥ 3 and cigarette smoking, however, the study was carried out on more premature infants (<29 weeks of gestation) and a total of four study groups were defined (including hypertensive mothers) [16].
In addition to maternal smoking, multivariate analysis confirmed known risk factors for the development of ROP. Low gestational age and birth weight are the main determinants of ROP [5, 38], which was also recapitulated in the current study. Of note, infants which are born small for gestational age (SGA) have also been shown to be at higher risk for the development of ROP and it has been suggested to include SGA as a screening parameter to identify vulnerable patients [39]. As SGA is derived from gestational age and birth weight, this association may also be deducted from our results. Very preterm neonates require respiratory support, which has been shown to increase the odds for ROP development [40, 41]. In line, our study confirmed an association of the duration of total respiratory support with the development of ROP. Controversial risk factors like maternal age, mode of delivery, gender, multiple pregnancies or low Apgar Score were not corroborated by our data.
Conclusion
Our study identifies smoking as an independent factor associated with the development of severe ROP in very premature infants. This finding underlines the importance to quit smoking during pregnancy and may provide additional insights for clinicians who counsel pregnant women.
Summary
What was known before
-
Retinopathy of prematurity (ROP) is a severe neonatal complication leading to visual impairment and blindness.
-
Known risk factors include preterm birth, low birth weight and respiratory support.
-
Contradictory data exist on the risk of maternal smoking during pregnancy on the development of ROP.
What this study adds
-
Maternal smoking is an independent risk factor for the development of severe ROP in preterm infants born < 32 weeks of gestation.
-
Low gestational age, low birth weight and prolonged respiratory support were confirmed as risk factors for the development of severe ROP.
References
Vogel JP, Chawanpaiboon S, Moller A-B, Watananirun K, Bonet M, Lumbiganon P. The global epidemiology of preterm birth. Best Pract Res Clin Obstet Gynaecol. 2018;52:3–12.
Chawanpaiboon S, Vogel JP, Moller A-B, Lumbiganon P, Petzold M, Hogan D, et al. Global, regional, and national estimates of levels of preterm birth in 2014: a systematic review and modelling analysis. Lancet Glob Health. 2019;7:e37–e46.
Natarajan G, Shankaran S, Nolen TL, Sridhar A, Kennedy KA, Hintz SR, et al. Neurodevelopmental outcomes of preterm infants with retinopathy of prematurity by treatment. Pediatrics. 2019;144:e20183537.
Solebo AL, Teoh L, Rahi J. Epidemiology of blindness in children. Arch Dis Child. 2017;102:853–7.
Kim SJ, Port AD, Swan R, Campbell JP, Chan RVP, Chiang MF. Retinopathy of prematurity: a review of risk factors and their clinical significance. Surv Ophthalmol. 2018;63:618–37.
Blencowe H, Lawn JE, Vazquez T, Fielder A, Gilbert C. Preterm-associated visual impairment and estimates of retinopathy of prematurity at regional and global levels for 2010. Pediatr Res. 2013;74(Suppl 1(S1)):35–49.
Schaffer DB, Palmer EA, Plotsky DF, Metz HS, Flynn JT, Tung B, et al. Prognostic factors in the natural course of retinopathy of prematurity. The Cryotherapy for Retinopathy of Prematurity Cooperative Group. Ophthalmology. 1993;100:230–7.
Kanungo J, James A, McMillan D, Lodha A, Faucher D, Lee SK, et al. Advanced maternal age and the outcomes of preterm neonates: a social paradox? Obstet Gynecol. 2011;118:872–7.
Uchida A, Miwa M, Shinoda H, Koto T, Nagai N, Mochimaru H, et al. Association of maternal age to development and progression of retinopathy of prematurity in infants of gestational age under 33 weeks. J Ophthalmol. 2014;2014:187929–5.
Wu W-C, Ong FS-C, Kuo JZ-C, Lai C-C, Wang N-C, Chen K-J, et al. Retinopathy of prematurity and maternal age. Retina. 2010;30:327–31.
Abdel HA, Mohamed GB, Othman MF. Retinopathy of prematurity: a study of incidence and risk factors in NICU of Al-Minya University Hospital in Egypt. J Clin Neonatol. 2012;1:76–81.
Darlow BA, Hutchinson JL, Henderson-Smart DJ, Donoghue DA, Simpson JM, Evans NJ, et al. Prenatal risk factors for severe retinopathy of prematurity among very preterm infants of the Australian and New Zealand Neonatal Network. Pediatrics. 2005;115:990–6.
Figueras-Aloy J, Alvarez-Domínguez E, Morales-Ballus M, Salvia-Roiges MD, Moretones-Suñol G. [Early administration of erythropoietin in the extreme premature, a risk factor for retinopathy of prematurity?]. Pediatrics. 2010;73:327–33.
Wikstrand MH, Hård A-L, Niklasson A, Smith L, Löfqvist C, Hellström A. Maternal and neonatal factors associated with poor early weight gain and later retinopathy of prematurity. Acta Paediatr. 2011;100:1528–33.
Hirabayashi H, Honda S, Morioka I, Yokoyama N, Sugiyama D, Nishimura K, et al. Inhibitory effects of maternal smoking on the development of severe retinopathy of prematurity. Eye. 2010;24:1024–7.
Yusuf K, Alshaikh B, da Silva O, Lodha AK, Wilson RD, Alvaro RE, et al. Neonatal outcomes of extremely preterm infants exposed to maternal hypertension and cigarette smoking. J Perinatol. 2018;38:1051–9.
Spiegler J, Jensen R, Segerer H, Ehlers S, Kühn T, Jenke A, et al. Influence of smoking and alcohol during pregnancy on outcome of VLBW infants. Z Geburtshilfe Neonatol. 2013;217:215–9.
Hackshaw A, Rodeck C, Boniface S. Maternal smoking in pregnancy and birth defects: a systematic review based on 173 687 malformed cases and 11.7 million controls. Hum Reprod Update. 2011;17:589–604.
Isayama T, Shah PS, Ye XY, Dunn M, da Silva O, Alvaro R, et al. Adverse impact of maternal cigarette smoking on preterm infants: a population-based cohort study. Am J Perinatol. 2015;32:1105–11.
Hayashi K, Matsuda Y, Kawamichi Y, Shiozaki A, Saito S. Smoking during pregnancy increases risks of various obstetric complications: a case-cohort study of the Japan Perinatal Registry Network database. J Epidemiol. 2011;21:61–66.
Ion R, Bernal AL. Smoking and preterm birth. Reprod Sci. 2015;22:918–26.
Caleyachetty R, Tait CA, Kengne AP, Corvalan C, Uauy R, Echouffo-Tcheugui JB. Tobacco use in pregnant women: analysis of data from Demographic and Health Surveys from 54 low-income and middle-income countries. Lancet Glob Health. 2014;2:e513–e520.
Hartnett ME, Penn JS. Mechanisms and management of retinopathy of prematurity. N Engl J Med. 2012;367:2515–26.
Aiello LP, Pierce EA, Foley ED, Takagi H, Chen H, Riddle L, et al. Suppression of retinal neovascularization in vivo by inhibition of vascular endothelial growth factor (VEGF) using soluble VEGF-receptor chimeric proteins. Proc Natl Acad Sci USA. 1995;92:10457–61.
Pons M, Marin-Castaño ME. Nicotine increases the VEGF/PEDF ratio in retinal pigment epithelium: a possible mechanism for CNV in passive smokers with AMD. Investig Ophthalmol Vis Sci. 2011;52:3842–53.
Zhang Y, Ma A, Wang L, Zhao B. Nornicotine and Nicotine Induced Neovascularization via Increased VEGF/PEDF Ratio. Ophthalmic Res. 2015;55:1–9.
International Committee for the Classification of Retinopathy of Prematurity. The International Classification of Retinopathy of Prematurity revisited. Arch Ophthalmol. 2005;123:991–9.
Leonardi-Bee J, Smyth A, Britton J, Coleman T. Environmental tobacco smoke and fetal health: systematic review and meta-analysis. Arch Dis Child Fetal Neonatal Ed. 2008;93:F351–61.
Wagijo M-A, Sheikh A, Duijts L, Been JV. Reducing tobacco smoking and smoke exposure to prevent preterm birth and its complications. Paediatr Respir Rev. 2017;22:3–10.
Miyazaki Y, Hayashi K, Imazeki S. Smoking cessation in pregnancy: psychosocial interventions and patient-focused perspectives. Int J Women’s Health. 2015;7:415–27.
Bakker R, Kruithof C, Steegers EAP, Tiemeier H, Mackenbach JP, Hofman A, et al. Assessment of maternal smoking status during pregnancy and the associations with neonatal outcomes. Nicotine Tob Res. 2011;13:1250–6.
Dammann O. Inflammation and retinopathy of prematurity. Acta Paediatr. 2010;99:975–7.
Kuon RJ, Hudalla H, Seitz C, Hertler S, Gawlik S, Fluhr H, et al. Impaired neonatal outcome after emergency cerclage adds controversy to prolongation of pregnancy. PLoS ONE. 2015;10:e0129104.
Evereklioglu C, Ozkiriş A, Alaşehirli B, Sari I, Güldür E, Cengiz B, et al. Effect of gestational nicotine treatment on newborn rat retina: a histopathological and morphometric analysis. Ophthalmic Physiol Opt. 2003;23:527–33.
Ram FSF, McDonald EM. Response to ‘Inhibitory effects of maternal smoking on the development of severe retinopathy of prematurity’. Eye. 2011;25:123–4.
Chiang MF, Jiang L, Gelman R, Du YE, Flynn JT. Interexpert agreement of plus disease diagnosis in retinopathy of prematurity. Arch Ophthalmol. 2007;125:875–80.
Campbell JP, Ryan MC, Lore E, Tian P, Ostmo S, Jonas K, et al. Diagnostic discrepancies in retinopathy of prematurity classification. Ophthalmology. 2016;123:1795–801.
Lundgren P, Kistner A, Andersson EM, Hansen Pupp I, Holmström G, Ley D, et al. Low birth weight is a risk factor for severe retinopathy of prematurity depending on gestational age. PLoS ONE. 2014;9:e109460.
Razak A, Faden M. Association of small for gestational age with retinopathy of prematurity: a systematic review and meta-analysis. Arch Dis Child Fetal Neonatal Ed. 2020;105:270–8.
Slidsborg C, Jensen A, Forman JL, Rasmussen S, Bangsgaard R, Fledelius HC, et al. Neonatal risk factors for treatment-demanding retinopathy of prematurity: a danish national study. Ophthalmology. 2016;123:796–803.
York JR, Landers S, Kirby RS, Arbogast PG, Penn JS. Arterial oxygen fluctuation and retinopathy of prematurity in very-low-birth-weight infants. J Perinatol. 2004;24:82–87.
Author information
Authors and Affiliations
Contributions
HH: protocol/project development, data collection or management, data analysis, paper writing/editing. TB: data collection or management, data analysis. JP: paper editing. TS: paper editing. RJK: protocol/project development, data collection or management, data analysis, paper writing/editing. All authors read and approved the final paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was approved by the Heidelberg University institutional review board (IRB, S-094/2013). All procedures performed in the study were in accordance with the ethical standards and data safety of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments.
Informed consent
The institutional ethics committee waived informed consent due to the retrospective nature of the study, and pseudonymization of data
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hudalla, H., Bruckner, T., Pöschl, J. et al. Maternal smoking as an independent risk factor for the development of severe retinopathy of prematurity in very preterm infants. Eye 35, 799–804 (2021). https://doi.org/10.1038/s41433-020-0963-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-020-0963-4
This article is cited by
-
Toxische Retinopathien
Der Ophthalmologe (2020)