Key Points
-
Patients with potential for bleeding are common in dental practice.
-
Management of patients with acquired bleeding disorders begins with a careful medical and drug history.
-
Significant postoperative bleeding due to medications is rare in dentistry.
-
Altering a patient's anticoagulant medication often puts them at increased risk of morbidity and mortality.
-
Almost all patients taking anticoagulants can receive dental care in the community setting.
Abstract
Dental patients often give a medical history that suggests the possibility of a coagulopathy from drugs, with a corresponding risk for prolonged bleeding during and following an invasive procedure. Identification of patients who may be prone to oral bleeding requires specific medical history information and the proper use of laboratory tests. Some NSAIDs are reported to cause prolonged oral bleeding, but scientific evidence is lacking. Likewise, the risk of oral bleeding from anticoagulants such as warfarin is often over stated, and unnecessary adjustment of NSAID or warfarin dosage puts patients at risk for significant morbidity and mortality. Some commonly employed laboratory tests such as the prothrombin time provide helpful information when used in the appropriate setting, but others, such as the bleeding time test, provide little or no predictive value in the determination of patients at risk for oral bleeding. Dental management of patients with potential coagulopathies from medications requires an understanding of basic principles of coagulation. The vast majority of these patients can be managed in the community setting without risk and without alteration of anticoagulant drug regimes.
Similar content being viewed by others
Main
An increasing number of dental patients are taking drugs which interfere with haemostasis, and there is concern that they might have prolonged bleeding during or following an invasive dental procedure. Although complete coverage of this subject would include both inherited and acquired coagulopathies, only acquired coagulopathies from medications are addressed in this paper. Acquired coagulopathies from systemic disease are covered in a companion article. (Lockhart et al., Br Dent J 2003; 195: 439–445)
Clinically significant bleeding is an unusual if not rare event in dental practice, even for patients known to be at risk. A review of the medical and dental literature revealed only ten reports of excessive bleeding due to drugs following dental procedures, primarily from the use of warfarin at a time before standardisation of the prothrombin time test used to measure the effectiveness of this drug.1,2 Other reports of prolonged bleeding suggest local factors (eg spitting, smoking) as underlying causes.
The management of a patient at risk for bleeding from medications such as warfarin or aspirin involves careful medical history-taking, consideration of specific laboratory tests and perhaps a discussion with the patient's physician. The dental practitioner must weigh the risk of clinically significant bleeding from an invasive procedure against the risks associated with an alteration in medical management, such as reducing the dose of an anticoagulant. Specific adjustments in dental management are often proposed for a given clinical scenario, usually based on established community standards or clinical guidelines. However, these management guidelines are often based on opinion, case reports, and clinical dogma, rather than clinical research data.3 Recent UK guidelines have taken a stand on the issue of patients using warfarin.4 This paper summarises the dental practice implications for acquired coagulopathies from drugs, based on the literature and official guidelines.
Patient evaluation and management
History taking
Standard medical history questionnaires should be used to gather preliminary information as a starting point for a more detailed questioning about specific medications known or reported to interfere with haemostasis (Table 1). The specific drug, dose, route of administration, and duration of use should be recorded for each medication. The dental surgeon should take the time to read about each drug and its effects from the BNF or similar publications.
Laboratory testing
Several laboratory tests are commonly used to evaluate haemostasis prior to invasive procedures, but some of the common tests (eg bleeding time, prothrombin time) are of little or no value for medication-related bleeding. Appropriate tests for acquired coagulopathies from medications include a platelet count for patients on cancer chemotherapy, International Normalised Ratio (INR) for patients taking warfarin, and partial thromboplastin time (PTT) for patients on heparin (Table 2). What follows is a discussion of the more common coagulopathies from medications, along with the appropriate laboratory test(s) and considerations for dental management.
Coagulopathy as a consequence of medications:
Aspirin, other non-steroidal anti-inflammatory drugs, and dipyridamole
When vascular endothelial cells are damaged, platelets bind to exposed collagen via glycoprotein receptors complexed to Von Willebrand factor. These platelets degranulate, releasing ADP and other substances (eg thromboxane A2, serotonin, epinephrine) that are important in the platelet aggregation process. Since platelets have an important role in coagulation and, in particular, arterial thrombosis, antiplatelet drugs are in widespread use for the prevention of morbidity and mortality from vascular disease. Aspirin has been found to be effective in the prophylaxis of angina, acute myocardial infarction, transient ischaemic attack and stroke, atrial fibrillation, and prevention of clots around prosthetic heart valves. Aspirin, an ingredient of many prescription and over-the-counter drugs (Table 1), has weak antiplatelet activity through acetylating and irreversibly inactivating the enzyme cyclo-oxygenase, which prevents the conversion of arachidonic acid to thromboxane A, the prostaglandin required to stimulate platelet activation and aggregation.5,6,7,8,9,10 Platelet inhibition by aspirin begins at about 1 hour following ingestion, and it is irreversible, lasting for the lifetime of the affected platelets (around 7–10 days).5,9 It has been shown that as little as 80 mg of aspirin will have this effect on all existing platelets, and it is unclear if larger doses or prolonged usage will have a greater effect.5,11 Aspirin, therefore, has its impact on platelet function. The platelet count, or measure of the number of platelets in the systemic circulation, gives no information concerning their viability or functionality. Although some medications (eg cancer chemotherapy) and some diseases (eg alcoholism) can cause bone marrow suppression and a lowered platelet count, this is an unusual situation in community dental practice. Therefore, the platelet count (via the full blood count) has no purpose in the preoperative evaluation of otherwise healthy patients taking non-steroidal anti-inflammatory drugs.
The bleeding time (BT) test has long been thought to predict the degree of interference from aspirin on platelet function, and therefore on bleeding during and following an invasive dental procedure. Although aspirin has been shown to increase the BT, and one study showed an increase in perisurgical blood loss in patients taking aspirin,12 the literature suggests overwhelmingly that prolonged BTs do not translate into increased blood loss from surgery.5,11,13,14,15,16,17,18,19,20 For example, O'Laughlin et al., showed that while standard forearm BTs were elevated in patients taking aspirin, a BT done at the surgical site during liver biopsy was unchanged.21 Because of the nonspecific nature of this test, it should be used only when the medical history identifies a specific reason for doing so.22
Non-aspirin NSAIDs, such as ibuprofen, are known to reduce the production of thromboxane A2 and interfere with platelet aggregation23 and have been reported to cause bleeding in patients taking oral anticoagulants (eg warfarin).24,25 Unlike aspirin, the antiplatelet effect is reversible, since the function of platelet cyclo-oxygenase (COX-1) is restored as the drugs are cleared from the circulation. However, different NSAIDs cause a varied extent and duration of platelet inhibition ranging from 1 to 3 days after discontinuing the drug.26 The effect of NSAIDs on BT is variable, but BT test values usually return to normal within several hours of a single dose. Aspirin is reported to cause surgical bleeding complications in the general, orthopaedic, and cardiac surgery arenas, but this is of questionable significance in the dental setting. However, care must also be exercised with the use of aspirin in patients taking warfarin,27 and with the use of non-aspirin NSAIDs in elderly patients and those taking aspirin for cardiovascular disease.25,28 In addition, a recent study suggests that the pre-treatment of patients taking low dose aspirin for its cardioprotective effects with ibuprofen blocks the inhibition of platelet cyclooxygenase-1 activity and therefore the anti-platelet activity of aspirin.28
Dipyridamole, a more potent anti-platelet drug than aspirin, is currently used as an adjunct therapy to reduce the risk of thrombotic stroke in patients who have failed or who cannot take aspirin, and for those with prosthetic heart valves. This drug affects platelet function by inhibiting the binding of ADP to platelet fibrinogen receptor (glycoprotein IIb-IIIa) and subsequent platelet aggregation.29
Other analgesics — COX-2 inhibitors and paracetamol
The recent introduction of cyclo-oxygenase-2, or COX-2 inhibitors offers an alternative analgesic to NSAIDs for postoperative pain in dental practice.30 This class of drugs also inhibits the activity of cyclo-oxygenase, which mediates the conversion of arachidonic acid to prostaglandins involved in the inflammatory process. Because they lack COX-1 activity, the likelihood of gastrointestinal upset should be lessened, and they are unlikely to have an adverse effect on platelet function. Although clinical research data are lacking, studies using laboratory values as outcomes suggest that COX-2 inhibitors have no effect on platelet aggregation, in contrast to aspirin and non-aspirin NSAIDs. This suggests that COX-2 inhibitors are unlikely to increase bleeding time following invasive dental procedures, although there are no prospective studies in humans.31,32
Although it is well established that paracetamol has little or no effect on platelet function, there have been several reports of an increase of the INR value in patients taking this drug along with warfarin.33,34,35,36 Care must also be exercised with the use of paracetamol in patients with liver disease.
Dental management considerations for patients taking analgesics
A common community standard for patients who have taken aspirin within the past week is to stop the use of any aspirin-containing medications and to delay invasive dental procedures for upwards of 5 days, with the understanding that 50% of the functional platelet population will be restored in that time period.11 The potential problems associated with this practice include a postponement of urgent treatment and, more importantly, a decrease in the cardiovascular benefit of aspirin when prescribed for its antithrombotic properties.15,37,38 A search of the literature failed to uncover a well documented case in which clinically significant bleeding followed tooth extraction in a patient receiving aspirin. Therefore, there is insufficient evidence to support the practice of altering aspirin therapy prior to dental surgery. This would apply to non-aspirin NSAIDs as well, but it is unclear if this applies to other platelet inhibiting drugs such as dipyridamole.39
Warfarin
Since well over a million people in the United States are treated annually with warfarin, and over 300,000 in the UK,40,41 this drug is commonly encountered in dental practice. It is used to prevent various thromboembolic events such as those that occur with prosthetic heart valves and atrial fibrillation (Table 3). Warfarin interferes with the synthesis of Vitamin K, which is necessary for thrombin formation and the synthesis of the Vitamin K-dependent coagulant protein factors II (prothrombin), VII, IX, X, and other vitamin K-dependent proteins C and S.42 Warfarin is bound to albumin, metabolised by the liver, and excreted in the urine. The therapeutic range for warfarin therapy has varied over the last 50 years because there was no standardisation of the prothrombin time (PT) test. When attempts were made to standardise this test in the 1970s, the accepted range of anticoagulation established in the 1940s was found to be too high. This explains the reports in the medical literature of problems with warfarin-related bleeding problems which, along with animal studies, likely initiated the current standards of care for invasive dental procedures.1,43,44 Therefore, the use of the PT test to evaluate the impact of warfarin was deemed inappropriate. In 1983 the World Health Organization adopted the International Normalised Ratio (INR), which calibrates the thromboplastin used and thereby standardizes PT test values.45 The INR specifically measures the responsiveness of the thromboplastin to a specific warfarin-induced defect, and it therefore measures the effect of warfarin and not liver function. The baseline INR is 1.0 An INR of 2.0 to 3.0 roughly correlates with a PT ratio of 1.3 to 1.5, and an INR of 3.0 to 4.5 with a PT ratio of 1.5 to 1.9, depending on the sensitivity index of the prothrombin reagent.45 The therapeutic INR range varies according to the medical indication for its use, but generally falls into three main groups:
-
1
Cardiac arrhythmias, such as atrial fibrillation
-
2
Thromboembolic events, prosthetic heart valves, and valvular heart disease, and
-
3
Mechanical heart valves (Table 3).
Optimal therapeutic ranges for anticoagulation were established in the late 1980s and recommended that the INR value be between 2.0 and 3.0 for most anticoagulation regimens, and in the range of 3.0 to 4.0 for patients with mechanical heart valves and/or a history of recurrent embolism.46,47,48 These guidelines significantly lowered the level of anticoagulation for many patients and have thus decreased the incidence of morbidity from clinical bleeding.43,45 Although the INR system is the appropriate test for patients taking warfarin, its replacement for the PT test in dental practice has been slow.45,49,50,51,52
Dental management considerations for patients taking warfarin
Controversy exists concerning the management of patients taking warfarin who need invasive dental procedures, much of which stems from the legitimate concern and better documented problem of bleeding following non-oral surgical procedures.53 The guidelines adopted by dentistry are generally without science-based evidence, and stem from case reports in the 1950s suggesting that warfarin posed a significant risk for patients having dental surgery.1,2 Today, patients on warfarin are felt to be at higher risk for prolonged postoperative bleeding with high INR values (eg ≥3.5) and when they have an additional coagulopathy.54 Studies done on patients taking warfarin with INRs in the therapeutic range versus controls found little or no difference in the incidence of clinically significant bleeding, even though some had warfarin levels above the present recommended therapeutic levels, and some underwent extensive oral surgery.55,56,57,58,59 Nevertheless, it is a common recommendation and standard of care to stop warfarin 2–3 days prior to an invasive dental procedure to decrease the INR value to less than 2.0-2.5.40,49,60 A survey of practicing physicians in 1996 reports that 73% recommended the withdrawal of warfarin in at least some patients for some dental procedures.61 A Danish study found that 86% of dentists refer patients to their medical practitioner for adjustments of their warfarin dosage.62 The dental literature suggests that the preoperative PT/INR test result should be under two times normal for patients taking warfarin,3 which includes the majority of patients taking this drug. However, there is no scientific evidence that the PT/INR value needs to be this low for invasive dental procedures.
Warfarin has a half life of about 36 hrs, and it may take up to 4 days for the INR to reach 1.5 once warfarin has been stopped depending on the original INR value. However, the INR decreases exponentially and has a wide interpatient variation in the rate of decrease.63 When restarted, it takes about 3 days to reach an INR of 2.0, such that patients may have at least 2-3 days of subtherapeutic anticoagulation around the time of surgery.53 This is an especially worrisome practice in patients that have had a thromboembolic event within the past month. Since recurrent embolic events are more frequent, and some will be fatal, elective surgery should be deferred in this time period. In addition to the risk of morbidity and mortality from thromboembolic events, the literature suggests that lowering the INR level is expensive and unnecessary from the standpoint of the likelihood of bleeding from dental procedures.1,38,50,55,64,65,66,67,68,69
Patients taking more than one anticoagulant, or who have more than one coagulopathy, are at greater risk of intra- and post-operative bleeding from dental procedures. For example, patients taking aspirin are at increased risk for minor prolonged bleeding if also taking warfarin, but there is no difference in major bleeding episodes (Table 1).70 In addition, paracetamol,33 broad spectrum antibiotics, chloral hydrate, metronidazole, miconazole and fluconazole can also potentiate the warfarin effect. Similarly, adrenal corticosteroids, barbiturates, and sucralfate can antagonise the effect of warfarin. Finally, conditions such as old age, liver disease, biliary disease, malabsorption, congestive heart failure, fever, hypo- and hyperthyroidism, malnutrition, cancer, and vitamin K excess or deficiency can all interfere with warfarin activity.
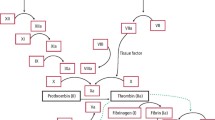
The clinical data and anecdotal evidence from dentists who often manage these patients suggests that INR levels within the accepted therapeutic ranges (ie INR ≤3.5) used to treat most medical conditions do not result in clinically significant increased bleeding during or following invasive dental procedures if local measures are used.64,65,71 The lack of evidence for a risk of bleeding in the dental setting must be contrasted with the case reports of morbidity and mortality in patients who had their continuous anticoagulation withdrawn for any period of time.59,72,73,74,75 A proposed algorithm for treatment of patients on warfarin is shown in Figure 1.
The INR value is accurate only for the time that the blood was taken; and other factors such as dietary considerations (eg vitamin K intake), medications, fever, and liver disease, can all influence the INR test result. Given the fluctuating nature of INR values, the INR test should be repeated within 24 hours of surgery, and ideally the same day. The dental procedure can usually be accomplished with careful attention to local measures, but in rare cases the INR value might need to be lowered. Two days without warfarin will usually bring the INR to an acceptable range, and warfarin can be started again late in the day of surgery. If the need arises to reverse the effect of warfarin, the patient's physician can do so with the administration of vitamin K. For more urgent situations, fresh frozen plasma will provide an almost immediate correction.
Another common protocol calls for the cessation of warfarin altogether until the INR returns to near normal, and then initiating warfarin again after the surgical procedure, which likely puts patients at unnecessary risk. A third protocol, often employed when there is a high risk for complications (eg prosthetic heart valves), is to hospitalise the patient, stop the warfarin, and institute intravenous heparin, which is stopped several hours before surgery.49 Warfarin is then reinstituted 12–18 hours after surgery. Heparin immediately inactivates thrombin and factor Xa, and it is administered to hospitalised patients to prevent clot formation in catheters, shunts, and various pumps and infusion machines (eg dialysis), and therefore it is not relevant to the dental outpatient setting. However, low molecular weight heparin (LMWH) is commonly used for non-hospitalised patients at risk for venous thrombosis. The disadvantages of intravenous heparin use are not shared with LMWH, which inhibits factor Xa and has a lesser effect on inhibition of thrombin, and is therefore more specific in its activity.76,77,78 LMWH is given subcutaneously once or twice daily in a dose based on body weight, and since the half life is twice that of standard heparin, no laboratory monitoring is necessary. The activated partial thromboplastin time (aPTT) test is used to monitor anticoagulation from heparin (but not LMWH), and it measures all intrinsic pathway factors except III, VII, and VIII. Although the aPTT test is usually given as part of the standard coagulation profile from hematology laboratories, its main focus for heparin anticoagulation makes it of little or no value in the evaluation of dental patients in the community setting.
Antibiotics
There have been rare reports suggesting that certain antibiotics (eg metronidazole and erythromycin), and in particular the second and third generation cephalosporins,47 and antifungal drugs may potentiate bleeding. The mechanism is unclear, but it might be reflected in an alteration of PT/INR test values from an interference with vitamin K metabolism.79 This can result from fasting, but it is doubtful that this potential interaction would be of clinical significance in an individual with an adequate diet. There are case reports of increased oral bleeding from antibiotics following oral surgery, but in each case patients were on warfarin as well as an antibiotic.80 Nevertheless, dentists should be aware of this issue and the potential need for appropriate monitoring of the INR for patients taking these drugs.79
Herbs and dietary supplements
At least 15 million Americans are taking herbs, high-dose vitamins, or both, combined with prescription drugs.81 The consumption of alternative supplements by individuals about to undergo surgical procedures have been estimated at over 70%, and 24-35% of patients consumed upwards of 62 different types of herbs, some of which may inhibit coagulation.82 The most commonly used compounds were echinacea, ginko biloba, St. John's wort, garlic, and ginseng.83 It has been suggested that upwards of 70% of patients taking herbs do not disclose this in a pre-anesthesia interview.84 The concern is that many herbs and dietary supplements such as garlic, ginko, and ginseng inhibit platelet adhesion and aggregation, or contain coumarins.85 Such reports of herb-drug interactions are often without confirmation by laboratory drug analysis, and anticoagulation may be more a laboratory phenomenon than a clinical concern.86 Nevertheless, patients taking herbs reported to interfere with coagulation should be advised to stop their use within 2 weeks of a surgical procedure until more is known about the potential for complications.
Alcohol
No discussion of drug-induced coagulopathies would be complete without mentioning chronic exposure to alcohol. Liver disease from alcohol abuse is a common cause of a coagulopathy,87 and the resulting clotting problems could be enhanced by the use of over-the-counter medications, such as NSAIDs. Although alcohol by itself does not cause bleeding, it does potentiate bleeding time prolongation produced by aspirin and non-aspirin NSAIDs.88 This is of particular significance with the alcoholic patient taking NSAIDs who may also have liver failure, bone marrow involvement, and thrombocytopenia from hypersplenism. Heavy alcohol consumption can also lead to thrombocytopenia through folate deficiency or direct suppression of the bone marrow. Paracetamol is of particular concern in high doses as it is not well metabolised in the presence of hepatitis, and acute liver failure can result.
Cancer chemotherapy
Cancer chemotherapy that significantly suppresses the bone marrow will have the result of interfering with haemostasis primarily through a decrease in the number of platelets. Therefore, the platelet count is the most useful laboratory test in this setting, and it will usually be ordered with a full blood count and differential, as the morbidity associated with invasive dental procedures and low white blood cell counts is of at least equal concern. Ambulatory, non-hospitalised patients receiving chemotherapy should be discussed with their haematologist prior to invasive dental procedures. As a temporary measure, patients with acute dental infection can often be medicated with antibiotics and analgesics if necessary until their white blood cell and platelet counts rise to acceptable levels. If their blood counts are in an acceptable range, these patients can receive dental care without risk of bleeding, however, other considerations may preclude treatment.
General Management Considerations
Patients taking drugs thought to predispose to a coagulopathy should have a thorough history taken and, if indicated, specific screening tests and physician consultation should be obtained. Modifications to dental management should be based on the current understanding of the risks of altering the patient's medical management versus the risk for oral bleeding. Invasive procedures should be performed as atraumatically as possible, and the patient should be followed more closely than usual following surgery. Various local haemostatic measures have been proposed following surgery, including: primary wound closure, fibrin adhesive, tranexamic acid, thrombin-soaked gauze, and oxycellulose (Part I: Lockhart et al, Br Dent J 2003; 195: 439–445) Unfortunately, there are few if any prospective, controlled, randomised studies of these measures to show a definitive benefit.
References
Ziffer AM, Scopp IW, Beck J, Baum J, Berger AR . Profound bleeding after dental extractions during dicumarol therapy. N Engl J Med 1957; 256: 351–353.
Scopp IW, Fredrics H . Dental extractions in patients undergoing anticoagulant therapy. Oral Surg Oral Med Oral Pathol 1958; 11: 470–474.
Mulligan R, Weitzel KG . Pretreatment management of the patient receiving anticoagulant drugs. J Am Dent Assoc 1988; 117: 479–483.
Surgical management of the primary care dental patient on warfarin. 2001. Liverpool, England, North West Medicines Information Centre.
George JN, Shattil SJ . The clinical importance of acquired abnormalities of platelet function. N Engl J Med 1991; 324: 27–39.
Coller BS . Platelets and thrombolytic therapy. N Engl J Med 1990; 322: 33–42.
Oates JA, FitzGerald GA, Branch RA, Jackson EK, Knapp HR, Roberts LJ . Clinical implications of prostaglandin and thromboxane A2 formation. N Engl J Med 1988; 319: 689–698.
Ware JA, Heistad DD . Platelet-endothelium interactions. N Engl J Med 1993; 328: 628–635.
Vane JR, Änggård EE, Botting RM . Regulatory functions of the vascular endothelium. N Engl J Med 1990; 323: 27–36.
Schafer AI . Effects of nonsteroidal antiinflammatory drugs on platelet function and systemic hemostasis. J Clin Pharmacol 1995; 35: 209–219.
Mielke CH, Jr. Aspirin prolongation of the template bleeding time: Influence of venostasis and direction of incision. Blood 1982; 60: 1139–1142.
Goldman S, Copeland J, Moritz T, Henderson W, Zadina K, Ovitt T et al. Improvement in early saphenous vein graft patency after coronary artery bypass surgery with antiplatelet therapy: results of a Veterans Administration Cooperative Study. Circulation 1988; 77: 1324–1332.
Amrein PC, Ellman L, Harris WH . Aspirin-induced prolongation of bleeding time and perioperative blood loss. JAMA 1981; 245: 1825–1828.
Pawlak DF, Itkin AB, Lapeyrolerie FM, Zweig B . Clinical effects of aspirin and acetaminophen on hemostasis after exodontics. J Oral Surg 1978; 36: 944–947.
Barber A, Green D, Galluzzo T, Ts'ao C . The bleeding time as a preoperative screening test. Am J Med 1985; 78: 761–764.
Lind SE . The bleeding time does not predict surgical bleeding. J Am Soc Hematol 1991; 77: 2547–2552.
De Caterina R, Lanza M, Manca G, Strata GB, Maffei S, Salvatore L . Time test with parameters of surgical bleeding. Blood 1994; 84: 3363–3370.
Gewirtz AS, Miller ML, Keys TF . The clinical usefulness of the preoperative bleeding time. Arch Pathol Lab Med 1996; 120: 353–356.
Lind SE . Prolonged bleeding time. Am J Med 1984; 77: 305–312.
Ferraris VA, Swanson E . Aspirin usage and perioperative blood loss in patients undergoing unexpected operations. Surg Gynecol Obstet 1983; 156: 439–442.
O'Laughlin JC, Hoftiezer JW, Mahoney JP, Ivey KJ . Does aspirin prolong bleeding from gastric biopsies in man? Gastrointest Endosc 1981; 27: 1–5.
Rapaport SI . Preoperative hemostatic evaluation: Which tests, if any? Blood 1983; 61: 229–231.
Cox SR, VanderLugt JT, Gumbleton TJ, Smith RB . Relationships between thromboxane production, platelet aggregability, and serum concentrations of ibuprofen or flurbiprofen. Clin Pharmacol Ther 1987; 41: 510–521.
Silverstein FE, Faich G, Goldstein JL, Simon LS, Pincus T, Whelton A et al. Gastrointestinal toxicity with celecoxib vs nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis. The CLASS study: a randomized controlled trial. JAMA 2000; 284: 1247–1255.
Shorr RI, Ray WA, Daugherty JR, Griffin MR . Concurrent use of nonsteroidal anti-inflammatory drugs and oral anticoagulants places elderly persons at high risk for hemorrhagic peptic ulcer disease. Arch Intern Med 1993; 153: 1665–1670.
Cronberg S, Wallmark E, Söderberg I . Effect on platelet aggregation of oral administration of 10 non-steroidal analgesics to humans. Scand J Haematol 1984; 33: 155–159.
Petitti DB, Strom BL, Melmon KL . Prothrombin time ratio and other factors associated with bleeding in patients treated with warfarin. J Clin Epidemiol 1989; 42: 759–764.
Catella-Lawson F, Reilly MP, Kapoor SC, Cucchiara AJ, DeMarco S, Tournier B et al. Cyclooxygenase inhibitors and the antiplatelet effects of aspirin. N Engl J Med 2001; 345: 1809–1817.
Sharis PJ, Cannon CP, Loscalzo J . The antiplatelet effects of ticlopidine and clopidogrel. Ann Intern Med 1998; 129: 394–405.
May N, Epstein J, Osborne B . Selective COX-2 inhibitors: A review of their therapeutic potential and safety in dentistry. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2001; 92: 399–405.
Simon LS, Lanza FL, Lipsky PE, Hubbard RC, Talwalker S, Schwartz BD et al. Preliminary study of the safety and efficacy of SC-58635, a novel cyclooxygenase 2 inhibitor: efficacy and safety in two placebo-controlled trials in osteoarthritis and rheumatoid arthritis, and studies of gastrointestinal and platelet effects. Arthritis and Rheumatism 1998.; 41: 1591–1602.
Mengle-Gaw L, Hubbard RC, Karim A, Yu SS, Talwalker S, Isakson PC et al. A study of the platelet effects of SC-58635, a novel COX-2-selective inhibitor. Arthritis and Rheumatism 1997; 40 [9 Suppl.], s88.
Hylek EM, Heiman H, Skates SJ, Sheehan MA, Singer DE . Acetaminophen and other risk factors for excessive warfarin anticoagulation. JAMA 1998; 279: 657–662.
Seymour RA, Williams FM, Oxley A, Ward A, Fearns M, Brighan K et al. A comparative study of the effects of aspirin and paracetamol (acetaminophen) on platelet aggregation and bleeding time. Eur J Clin Pharmacol 1984; 26: 567–571.
Bartle WR, Blakely JA . Potentiation of warfarin anticoagulation by acetaminophen. JAMA 1991; 265: 1260.
Bagheri H . Potentiation of the acenocoumarol anticoagulant effect by acetaminophen. Ann Pharmacother 1999; 33: 506.
De Rossi S S, Glick M . Bleeding time: An unreliable predictor of clinical hemostasis. J Oral Maxillofac Surg 1996; 54: 1119–1120.
Weibert RT . Oral anticoagulant therapy in patients undergoing dental surgery. Clin Pharm 1992; 11: 857–864.
Alexander RE . Eleven myths of dentoalveolar surgery. J Am Dent Assoc 1998; 129: 1271–1279.
Webster K, Wilde J . Management of anticoagulation in patients with prosthetic heart valves undergoing oral and maxillofacial operations. Br J Oral Maxillofac Surg 2000; 38(2): 124–126.
Fihn SD, McDonell M, Martin D . Risk factors for complications of chronic anticoagulation: a multicenter study. Ann Intern Med 1993; 118: 511–520.
Hirsh J . Oral anticoagulant drugs. N Engl J Med 1991; 324: 1865–1875.
Hull R, Hirsh J, Jay R, Carter C, England C, Gent M et al. Different intensities of oral anticoagulant therapy in the treatment of proximal-vein thrombosis. N Engl J Med 1982; 307: 1676–1681.
Turpie AGG, Gunstensen J, Hirsh J, Nelson H, Gent M . Randomised comparison of two intensities of oral anticoagulant therapy after tissue heart valve replacement. Lancet 1988; 1(8597): 1242–1245.
Hirsh J, Poller L, Deykin D, Levine M, Dalen JE . Optimal therapeutic range for oral anticoagulants. Chest 1989; 95: 5S–11S.
Petruff CA, Chopra S . Cirrhosis and portal hypertension: An overview. In Friedman L S, Keeffe E B, Maddrey W C, eds. Handbook of Liver Disease. 133. New York: Churchill Livingstone, 1998.
Hirsh J, Fuster V . Guide to anticoagulant therapy. Part 2: Oral anticoagulants. American Heart Association. Circulation 1994; 89: 1469–1480.
Cannegieter SC, Rosendaal FR, Wintzen AR, van der Meer FJM, Vandenbroucke JP, Briët E . Optimal oral anticoagulant therapy in patients with mechanical heart valves. N Engl J Med 1995; 333: 11–17.
Troulis MJ, Head TW, Leclerc JR . Dental extractions in patients on an oral anticoagulant: A survey of practices in North America. J Oral Maxillofac Surg 1998; 56: 914–917.
Steinburg MJ, Moores JF, III . Use of INR to assess degree of anticoagulation in patients who have dental procedures. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1995; 80: 175–177.
Eckman MH, Levine HJ, Pauker SG . Effect of laboratory variation in the prothrombin-time ratio on the results of oral anticoagulant therapy. N Engl J Med 1993; 329: 696–702.
Stern R, Karlis V, Kinney L, Glickman R . Using the international normalized ratio to standardize prothrombin time. J Am Dent Assoc 1997; 128: 1121–1122.
Kearon C, Hirsh J . Management of anticoagulation before and after elective surgery. N Engl J Med 1997; 336: 1506–1511.
Palareti G, Leali N, Coccheri S, Poggi M, Manotti C, D'Angelo A et al. Bleeding complications of oral anticoagulant treatment: an inception-cohort, prospective collaborative study (ISCOAT). Lancet 1996; 348: 423–428.
Bodner L, Weinstein JM, Baumgarten AK . Efficacy of fibrin sealant in patients on various levels of oral anticoagulant undergoing oral surgery. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1998; 86: 421–424.
Wahl MJ . Dental surgery in anticoagulated patients. Arch Intern Med 1998; 158: 1610–1616.
Behrman SJ, Wright IS . Dental surgery during continuous anticoagulant therapy. J Am Dent Assoc 1961; 62: 172–180.
Campbell JH, Alvarado F, Murray RA . Anticoagulation and minor oral surgery: Should the anticoagulation regimen be altered? J Oral Maxillofac Surg 2000; 58: 131–135.
Devani P, Lavery KM, Howell CJT . Dental extractions in patients on warfarin: is alteration of anticoagulant regime necessary? Br J Oral Maxillofac Surg 1998; 36: 107–111.
Russo G, Dal Corso L, Biasiolo A, Berengo M, Pengo V . Simple and safe method to prepare patients with prosthetic heart valves for surgical dental procedures. Clin Appl Thrombosis/Hemostasis 2000; 6: 90–93.
Wahl MJ, Howell J . Altering anticoagulation therapy: A survey of physicians. J Am Dent Assoc 1996; 127: 625–638.
Lippert S, Gutschik E . Views of cardiac-valve prosthesis patients and their dentists on anticoagulation therapy. Scand J Dent Res 1994; 102: 168–171.
White RH, McKittrick T, Hutchinson R, Twitchell J . Temporary discontinuation of warfarin therapy: Changes in the international normalized ratio. Ann Intern Med 1995; 122: 40–42.
Benoliel R, Leviner E, Katz J, Tzukert A . Dental treatment for the patient on anticoagulant therapy: Prothrombin time value – what difference does it make? Oral Surg Oral Med Oral Pathol 1986; 62: 149–151.
Beirne OR, Koehler JR . Surgical management of patients on warfarin sodium. J Oral Maxillofac Surg 1996; 54: 1115–1118.
Rooney TP . General dentistry during continuous anticoagulation therapy. Oral Surg Oral Med Oral Pathol 1983; 56: 252–255.
Mulligan R . Response to anticoagulant drug withdrawal. J Am Dent Assoc 1987; 115: 435–438.
Eckman MH, Beshansky JR, Durand-Zaleski I, Levine HJ, Pauker SG . Anticoagulation for noncardiac procedures in patients with prosthetic heart valves. Does low risk mean high cost? JAMA 1990; 263: 1513–1521.
Saour JN, Ali HA, Mammo LARF, Sieck JO . Dental procedures in patients receiving oral anticoagulation therapy. J Heart Valve Dis 1994; 3: 315–317.
Hurlen M, Erikssen J, Smith P, Arnesen H, Rollag A . Comparison of bleeding complications of warfarin and warfarin plus acetylsalicylic acid: a study in 3166 outpatients. J Intern Med 1994; 236: 299–304.
Herman WW, Konzelman JL, Sutley SH . Current perspectives on dental patients receiving coumarin anticoagulant therapy. J Am Dent Assoc 1997; 128: 327–335.
Akbarian M, Austen WG, Yurchak PM, Scannell JG . Thromboembolic complications of prosthetic cardiac valves. Circulation 1968; 37: 826–831.
Blinder D, Manor Y, Martinowitz U, Taicher S . Dental extractions in patients maintained on continued oral anticoagulant. Comparison of local hemostatic modalities. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1999; 88: 137–140.
Marshall J . Rebound phenomena after anticoagulant therapy in cerebrovascular disease. Circulation 1963; 28: 329–332.
Ogiuchi H, Ando T, Tanaka M, Kuwasawa T, Sangu Y, Abe H et al. Clinical reports on dental extraction from patients undergoing oral anticoagulant therapy. Bull Tokyo Dent Coll 1985; 26: 205–212.
Koopman MMW, Prandoni P, Piovella F, Ockelford P, Brandjes DPM, van der Meer J et al. Treatment of venous thrombosis with intravenous unfractionated heparin administered in the hospital as compared with subcutaneous low-molecular-weight heparin administered at home. N Engl J Med 1996; 334: 682–687.
Levine M, Gent M, Hirsh J, Leclerc J, Anderson D, Weitz J et al. A comparison of low-molecular-weight heparin administered primarily at home with unfractionated heparin administered in the hospital for proximal deep-vein thrombisis. N Engl J Med 1996; 334: 677–681.
Harenberg J, Huhle G, Piazolo L, Giese C, Heene D . Long-term anticoagulation of outpatients with adverse events to oral anticoagulants using low-molecular-weight heparin. Semin Thromb Hemost 1997; 23: 167–172.
Mailloux AT, Gidal BE, Sorkness CA . Potential interaction between warfarin and dicloxacillin. Ann Pharmacother 1996; 30: 1402–1407.
Bandrowsky T, Vorono AA, Borris TJ, Marcantoni HW . Amoxicillin-related postextraction bleeding in an anticoagulated patient with tranexamic acid rinses. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1996; 82: 610–612.
Eisenberg DM, Davis RB, Ettner SL, Appel S, Wilkey S, Van Rompay M et al. Trends in alternative medicine use in the United States, 1990-1997: Results of a follow-up national survey. JAMA 1998; 280: 1569–1575.
Norred CL, Zamudio S, Palmer SK . Use of complementary and alternative medicines by surgical patients. AANA J 2000; 68: 13–18.
Tsen LC, Segal S, Pothier M, Bader AM . Alternative medicine use in presurgical patients. Anesthesiol 2000; 93: 148–151.
Kaye AD, Clarke RC, Sabar R, Vig S, Dhawan KP, Hofbauer R et al. Herbal medicines: Current trends in anesthesiology practice – a hospital survey. J Clin Anesth 2000; 12: 468–471.
Ang-Lee MK, Moss J, Yuan CS . Herbal medicines and perioperative care. JAMA 2001; 286: 208–216.
Lumb AB . Effect of dried ginger on human platelet function. Thromb Haemost 1994; 71: 110–111.
Friedlander AH, Mills MJ, Gorelick DA . Alcoholism and dental management. Oral Surg Oral Med Oral Pathol 1987; 63: 42–46.
Deykin D, Janson P, McMahon L . Ethanol potentiation of aspirin-induced prolongation of the bleeding time. N Engl J Med 1982; 306: 852–854.
Acknowledgements
The authors would like to thank Philip C. Fox, DDS; Mack Mitchell, MD; George Hart, MD; and Charles H. Packman, MD, for their help in reviewing this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Refereed Paper
Rights and permissions
About this article
Cite this article
Lockhart, P., Gibson, J., Pond, S. et al. Dental management considerations for the patient with an acquired coagulopathy. Part 2: Coagulopathies from drugs. Br Dent J 195, 495–500 (2003). https://doi.org/10.1038/sj.bdj.4810660
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bdj.4810660
This article is cited by
-
Acquired bleeding disorders through antithrombotic therapy: the implications for dental practitioners
British Dental Journal (2020)
-
Prevalence of and risk factors for postoperative hemorrhage after lower third molar extraction on warfarin therapy: a multicenter retrospective study in Japan
Odontology (2020)
-
Characteristics, treatment and outcome of bleeding after tooth extraction in patients on DOAC and phenprocoumon compared to non-anticoagulated patients—a retrospective study of emergency department consultations
Clinical Oral Investigations (2019)
-
An Assessment of the Incidence of Prolonged Postoperative Bleeding After Dental Extraction Among Patients on Uninterrupted Low Dose Aspirin Therapy and to Evaluate the Need to Stop Such Medication Prior to Dental Extractions
Journal of Maxillofacial and Oral Surgery (2017)
-
Prevention of post-operative bleeding in hemostatic compromised patients using native porcine collagen fleeces—retrospective study of a consecutive case series
Oral and Maxillofacial Surgery (2016)