Abstract
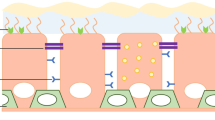
Gene transfer to the respiratory epithelium is currently suboptimal and may be helped by the identification of limiting biological barriers. We have, therefore, developed an ex vivo model which retains many of the characteristics of in vivo native airways including mucociliary clearance, mucus coverage and an intact cellular structure. Using this model we have demonstrated several barriers to gene transfer. Liposome-mediated gene transfer was inhibited by normal mucus, with removal of this layer increasing expression approximately 25-fold. In addition both liposome and adenovirus were inhibited by CF sputum. The apical membrane represented a significant barrier to both agents. Adenovirus-mediated expression could be significantly augmented by increasing contact time or by pretreatment of tissues with a nominally calcium-free medium. The presence of these extracellular and plasma membrane barriers appeared to be the key parameters responsible for the approximately three log difference in gene expression found in vitro compared with our ex vivo model. Cytoskeletal elements and the cell cycle also influenced in vitro gene transfer, and represent further barriers which need to be overcome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout















Similar content being viewed by others
References
Caplen NJ et al. Liposome-mediated CFTR gene transfer to the nasal epithelium of patients with cystic fibrosis Nature Med 1995 1: 39–46
Porteous DJ et al. Evidence for safety and efficacy of DOTAP cationic liposome mediated CFTR gene transfer to the nasal epithelium of patients with cystic fibrosis Gene Therapy 1997 4: 210–218
Gill DR et al. A placebo-controlled study of liposome-mediated gene transfer to the nasal epithelium of patients with cystic fibrosis Gene Therapy 1997 4: 199–209
Zabner J et al. Adenovirus-mediated gene transfer transiently corrects the chloride transport defect in nasal epithelia of patients with cystic fibrosis Cell 1993 75: 207–216
Crystal RG et al. Administration of an adenovirus containing the human CFTR cDNA to the respiratory tract of individuals with cystic fibrosis Nat Genet 1994 8: 42–51
Knowles MR et al. A controlled study of adenovirus-vector-mediated gene transfer in the nasal epithelium of patients with cystic fibrosis New Engl J Med 1995 333: 823–831
Bellon G et al. Aerosol administration of a recombinant adenovirus expressing CFTR to cystic fibrosis patients: A phase I clinical trial Hum Gene Ther 1997 4: 15–25
Zabner J et al. Repeat administration of an adenovirus vector encoding cystic fibrosis transmembrane conductance regulator to the nasal epithelium of patients with cystic fibrosis J Clin Invest 1996 97: 1504–1511
Dorin JR et al. A demonstration using mouse models that successful gene therapy for cystic fibrosis requires only partial gene correction Gene Therapy 1996 3: 797–801
Johnson LG et al. Efficiency of gene transfer for restoration of normal airway epithelial function in cystic fibrosis Nat Genet 1992 2: 21–25
Matsui H, Johnson LG, Randell SH, Boucher RC . Loss of binding and entry of liposome-DNA complexes decreases transfection efficiency in differentiated airway epithelial cells J Biol Chem 1997 272: 1117–1126
Zabner J et al. Cellular and molecular barriers to gene transfer by a cationic lipid J Biol Chem 1995 270: 18997–19007
Lee ER et al. Detailed analysis of structures and formulations of cationic lipids for efficient gene transfer to the lung Hum Gene Ther 1996 7: 1701–1717
Grubb BR et al. Inefficient gene transfer by adenovirus vector to cystic fibrosis airway epithelia of mice and humans Nature 1994 371: 802–806
Zabner J et al. Lack of high affinity fiber receptor activity explains the resistance of ciliated airway epithelia to adenovirus infection J Clin Invest 1997 100: 1144–1149
Pilewski JM et al. Adenonovirus-mediated gene transfer to human bronchial submucosal glands using xenografts Am J Physiol 1995 268: L657–L665
Worgall S, Wolff G, Falck-Pederson E, Crystal RG . Innate immune mechanisms dominate elimination of adenoviral vectors following in vivo administration Hum Gene Ther 1997 8: 37–44
Mack CA et al. Circumvention of anti-adenovirus neutralising immunity by administration of an adenoviral vector of an alternate serotype Hum Gene Ther 1997 8: 99–109
Scaria A et al. Antibody to CD40 ligand inhibits both humoral and cellular immune responses to adenoviral vectors and facilitates repeated administration to mouse airway Gene Therapy 1997 4: 611–617
Yang Y et al. Inactivation of E2a in recombinant adenoviruses improves the prospect for gene therapy in cystic fibrosis Nat Genet 1994 7: 362–369
Zabner J et al. Safety and efficacy of repetitive adenovirus-mediated transfer of CFTR cDNA to airway epithelia of primates and cotton rats Nat Genet 1994 6: 75–83
Brody SL et al. Acute responses of non-human primates to airway delivery of an adenovirus vector containing the human cystic fibrosis transmembrane conductance regulator cDNA Hum Gene Ther 1994 5: 821–836
Dupuit F et al. Regenerating cells in human airway surface epithelium represent preferential targets for recombinant adenovirus Hum Gene Ther 1995 6: 1185–1193
Fasbender A, Zabner J, Zeiher BG, Welsh MJ . A low rate of cell proliferation and reduced DNA uptake limit cationic lipid-mediated gene transfer to primary cultures of ciliated human airway epithelia Gene Therapy 1997 4: 1173–1180
Wills PJ et al. The ciliary transportability of sputum is slow on the mucus-depleted bovine trachea Am J Respir Crit Care Med 1995 151: 1255–1258
Stern M et al. The effect of mucolytic agents on gene transfer across a CF sputum barrierin vitro Gene Therapy 1998 5: 91–98
Bhat M et al. Regulation of tight junction permeability by calcium mediators and cell cytoskeleton in rabbit tracheal epithelium Pharm Res 1993 10: 991–997
Sheils CA et al. Actin filaments mediate DNA fiber formation in chronic inflammatory airway disease Am J Pathol 1996 148: 919–927
Fasbender A et al. Complexes of adenovirus with polycationic polymers and cationic lipids increase the efficiency of gene transfer in vitro and in vivo J Biol Chem 1997 272: 6479–6489
Arcasoy SM et al. Polycations increase the efficiency of adenovirus-mediated gene transfer to epithelial and endothelial cells in vitro Gene Therapy 1997 4: 32–38
Girod S et al. Role of physiochemical properties of mucus in the protection of the respiratory epitheium Eur Respir J 1992 5: 477–487
Lopez-Vidriero MT . Mucus as a natural barrier Respiration 1989 55: 28–32
Albanese CT et al. Role of intestinal mucus in transepithelial passage of bacteria across the intact ileum in vitro Surgery 1994 116: 76–82
Sandberg JW et al. Improving access to intestinal stem cells as a step toward intestinal gene transfer Hum Gene Ther 1994 5: 323–329
Zabner J, Zeiher BG, Friedman E, Welsh MJ . Adenovirus-mediated gene transfer to ciliated airway epithelia requires prolonged incubation time J Virol 1996 70: 6994–7003
Gao X, Huang L . A novel cationic liposome reagent for efficient transfection of mammalian cells Biochem Biophys Res Commun 1991 179: 280–285
Caplen NJ et al. In vitro liposome-mediated DNA transfection of epithelial cell lines using the cationic liposome DC-Chol/DOPE Gene Therapy 1995 2: 603–613
Caplen NJ et al. Gene therapy for cystic fibrosis in humans by liposome-mediated DNA transfer: the production of resources and the regulatory process Gene Therapy 1994 1: 139–147
Muller DWM et al. Catheter-mediated pulmonary vascular gene transfer and expression Circ Res 1994 75: 1039–1049
Jackson AD et al. A human respiratory-tissue organ culture incorporating an air interface Am J Respir Crit Care Med 1996 153: 1130–1135
Alton EWFW et al. Non-invasive liposome-mediated gene delivery can correct the ion transport defect in cystic fibrosis mutant mice Nat Genet 1993 5: 135–142
Chi K et al. Cyclin A expression in normal and transformed alveolar epithelial cells Am J Respir Cell Mol Biol 1993 9: 115–125
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kitson, C., Angel, B., Judd, D. et al. The extra- and intracellular barriers to lipid and adenovirus-mediated pulmonary gene transfer in native sheep airway epithelium. Gene Ther 6, 534–546 (1999). https://doi.org/10.1038/sj.gt.3300840
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3300840
Keywords
This article is cited by
-
Engineered amphiphilic peptides enable delivery of proteins and CRISPR-associated nucleases to airway epithelia
Nature Communications (2019)
-
Overcoming the Cystic Fibrosis Sputum Barrier to Leading Adeno-associated Virus Gene Therapy Vectors
Molecular Therapy (2014)
-
Plasmonic Nanostructures as Accelerators for Nanoparticles: Optical Nanocannon
Plasmonics (2013)
-
Jaagsiekte sheep retrovirus pseudotyped lentiviral vector-mediated gene transfer to fetal ovine lung
Gene Therapy (2012)
-
An ovine tracheal explant culture model for allergic airway inflammation
Journal of Inflammation (2010)