Abstract
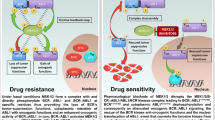
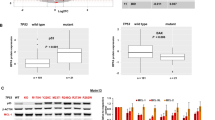
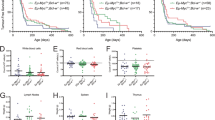
The Wilms' tumour gene 1 (WT1) protein is highly expressed in most leukaemias. Co-expression of WT1 and the fusion protein AML1-ETO in mice rapidly induces acute myeloid leukaemia (AML). Mechanisms behind expression of WT1, as well as consequences thereof, are still unclear. Here, we report that the fusion protein BCR/ABL1 increases expression of WT1 mRNA and protein via the phosphatidylinositol-3 kinase (PI3K)–Akt pathway. Inhibition of BCR/ABL1 or PI3K activity strongly suppressed transcription from WT1 promoter/enhancer reporters. Forced expression of BCR/ABL1 in normal human progenitor CD34+ cells increased WT1 mRNA and protein, further supporting the notion of BCR/ABL1-driven expression of WT1 in human haematopoietic cells. Forced expression of WT1 in K562 cells provided protection against cytotoxic effects of the ABL1 tyrosine kinase inhibitor imatinib, as judged by effects on viability measured by trypan blue exclusion, metabolic activity, annexin V and DAPI (4′, 6-diamidino-2-phenylindole) staining. None of the isoforms provided any detectable protection against apoptosis induced by arsenic trioxide and only very weak protection against etoposide, indicating that WT1 interferes with specific apoptotic signalling pathways. Our data demonstrate that WT1 expression is induced by oncogenic signalling from BCR/ABL1 and that WT1 contributes to resistance against apoptosis induced by imatinib.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Lee SB, Haber DA . Wilms tumour and the WT1 gene. Exp Cell Res 2001; 264: 74–99.
Scharnhorst V, van der Eb AJ, Jochemsen AG . WT1 proteins: functions in growth and differentiation. Gene 2001; 273: 141–161.
Call KM, Glaser T, Ito CY, Buckler AJ, Pelletier J, Haber DA et al. Isolation and characterization of a zinc finger polypeptide gene at the human chromosome 11 Wilms' tumor locus. Cell 1990; 60: 509–520.
Gessler M, Poustka A, Cavenee W, Neve RL, Orkin SH, Bruns GAP . Homozygous deletion in Wilms tumours of a zinc-finger gene identified by chromosome jumping. Nature 1990; 343: 774–778.
Fraizer GC, Patmasiriwat P, Zhang X, Saunders GF . Expression of the tumor suppressor gene WT1 in both human and mouse bone marrow. Blood 1995; 86: 4704–4706.
Baird PN, Simmons PJ . Expression of the Wilms' tumor gene (WT1) in normal hemopoiesis. Exp Hematol 1997; 25: 312–320.
Maurer U, Brieger J, Weidmann E, Mitrou PS, Hoelzer D, Bergmann L . The Wilms' tumor gene is expressed in a subset of CD34+ progenitors and downregulated early in the course of differentiation in vitro. Exp Hematol 1997; 25: 945–950.
Menssen HD, Renkl HJ, Entezami M, Thiel E . Wilms' tumor gene expression in human CD34+ hematopoietic progenitors during fetal development and early clonogenic growth. Blood 1997; 89: 3486–3487.
Hosen N, Sonoda Y, Oji Y, Kimura T, Minamiguchi H, Tamaki H et al. Very low frequencies of human normal CD34+ haematopoietic progenitor cells express the Wilms' tumour gene WT1 at levels similar to those in leukaemia cells. Br J Haematol 2002; 116: 409–420.
Hosen N, Shirakata T, Nishida S, Yanagihara M, Tsuboi A, Kawakami M et al. The Wilms' tumor gene WT1-GFP knock-in mouse reveals the dynamic regulation of WT1 expression in normal and leukemic hematopoiesis. Leukemia 2007; 21: 1783–1791.
King-Underwood L, Little S, Baker M, Clutterbuck R, Delassus S, Enver T et al. Wt1 is not essential for hematopoiesis in the mouse. Leuk Res 2005; 29: 803–812.
Alberta JA, Springett GM, Rayburn H, Natoli TA, Loring J, Kreidberg JA . Role of the WT1 tumor suppressor in murine hematopoiesis. Blood 2003; 101: 2570–2574.
Miwa H, Beran M, Saunders GF . Expression of the Wilms' tumor gene (WT1) in human leukemias. Leukemia 1992; 6: 405–409.
Miyagi T, Ahuja H, Kubota T, Kubonishi I, Koeffler HP, Miyoshi I . Expression of the candidate Wilm's tumor gene, WT1, in human leukemia cells. Leukemia 1993; 7: 970–977.
Inoue K, Sugiyama H, Ogawa H, Nakagawa M, Yamagami T, Miwa H et al. WT1 as a new prognostic factor and a new marker for the detection of minimal residual disease in acute leukemia. Blood 1994; 84: 3071–3079.
Menssen HD, Renkl HJ, Rodeck U, Maurer J, Notter M, Schwartz S et al. Presence of Wilms' tumor gene (wt1) transcripts and the WT1 nuclear protein in the majority of human acute leukemias. Leukemia 1995; 9: 1060–1067.
Tamaki H, Ogawa H, Inoue K, Soma T, Yamagami T, Miyake S et al. Increased expression of the Wilms tumor gene (WT1) at relapse in acute leukemia. Blood 1996; 88: 4396–4398.
Phelan SA, Lindberg C, Call KM . Wilms' tumor gene, WT1, mRNA is down-regulated during induction of erythroid and megakaryocytic differentiation of K562 cells. Cell Growth Differ 1994; 5: 677–686.
Sekiya M, Adachi M, Hinoda Y, Imai K, Yachi A . Downregulation of Wilms' tumor gene (wt1) during myelomonocytic differentiation in HL60 cells. Blood 1994; 83: 1876–1882.
Inoue K, Tamaki H, Ogawa H, Oka Y, Soma T, Tatekawa T et al. Wilms' tumor gene (WT1) competes with differentiation-inducing signal in hematopoietic progenitor cells. Blood 1998; 91: 2969–2976.
Svedberg H, Chylicki K, Baldetorp B, Rauscher III FJ, Gullberg U . Constitutive expression of the Wilms' tumor gene (WT1) in the leukemic cell line U937 blocks parts of the differentiation program. Oncogene 1998; 16: 925–932.
Tsuboi A, Oka Y, Ogawa H, Elisseeva OA, Tamaki H, Oji Y et al. Constitutive expression of the Wilms' tumor gene WT1 inhibits the differentiation of myeloid progenitor cells but promotes their proliferation in response to granulocyte-colony stimulating factor (G-CSF). Leuk Res 1999; 23: 499–505.
Nishida S, Hosen N, Shirakata T, Kanato K, Yanagihara M, Nakatsuka S et al. AML1-ETO rapidly induces acute myeloblastic leukemia in cooperation with the Wilms tumor gene, WT1. Blood 2006; 107: 3303–3312.
Cilloni D, Messa F, Gottardi E, Fava M, Arruga F, Defilippi I et al. Sensitivity to imatinib therapy may be predicted by testing Wilms tumor gene expression and colony growth after a short in vitro incubation. Cancer 2004; 101: 979–988.
Ren R . Mechanisms of BCR-ABL in the pathogenesis of chronic myelogenous leukaemia. Nat Rev Cancer 2005; 5: 172–183.
Svensson E, Eriksson H, Gekas C, Olofsson T, Richter J, Gullberg U . DNA-binding dependent and independent functions of WT1 protein during human hematopoiesis. Exp Cell Res 2005; 308: 211–221.
Kelly PF, Vandergriff J, Nathwani A, Nienhuis AW, Vanin EF . Highly efficient gene transfer into cord blood nonobese diabetic/severe combined immunodeficiency repopulating cells by oncoretroviral vector particles pseudotyped with the feline endogenous retrovirus (RD114) envelope protein. Blood 2000; 96: 1206–1214.
Edvardsson L, Dykes J, Olsson ML, Olofsson T . Clonogenicity, gene expression and phenotype during neutrophil versus erythroid differentiation of cytokine-stimulated CD34+ human marrow cells in vitro. Br J Haematol 2004; 127: 451–463.
Ginzinger DG . Gene quantification using real-time quantitative PCR: an emerging technology hits the mainstream. Exp Hematol 2002; 30: 503–512.
Hakansson P, Lassen C, Olofsson T, Baldetorp B, Karlsson A, Gullberg U et al. Establishment and phenotypic characterization of human U937 cells with inducible P210 BCR/ABL expression reveals upregulation of CEACAM1 (CD66a). Leukemia 2004; 18: 538–547.
Zhang X, Xing G, Fraizer GC, Saunders GF . Transactivation of an intronic hematopoietic-specific enhancer of the human Wilms' tumor 1 gene by GATA-1 and c-Myb. J Biol Chem 1997; 272: 29272–29280.
Wu Y, Fraizer GC, Saunders GF . GATA-1 transactivates the WT1 hematopoietic specific enhancer. J Biol Chem 1995; 270: 5944–5949.
Chalandon Y, Jiang X, Hazlewood G, Loutet S, Conneally E, Eaves A et al. Modulation of p210(BCR-ABL) activity in transduced primary human hematopoietic cells controls lineage programming. Blood 2002; 99: 3197–3204.
Zhao RC, Jiang Y, Verfaillie CM . A model of human p210(bcr/ABL)-mediated chronic myelogenous leukemia by transduction of primary normal human CD34(+) cells with a BCR/ABL-containing retroviral vector. Blood 2001; 97: 2406–2412.
Algar EM, Khromykh T, Smith SI, Blackburn DM, Bryson GJ, Smith PJ . A WT1 antisense oligonucleotide inhibits proliferation and induces apoptosis in myeloid leukaemia cell lines. Oncogene 1996; 12: 1005–1014.
Yamagami T, Sugiyama H, Inoue K, Ogawa H, Tatekawa T, Hirata M et al. Growth inhibition of human leukemic cells by WT1 (Wilms tumor gene) antisense oligodeoxynucleotides: implications for the involvement of WT1 in leukemogenesis. Blood 1996; 87: 2878–2884.
Ito K, Oji Y, Tatsumi N, Shimizu S, Kanai Y, Nakazawa T et al. Antiapoptotic function of 17AA(+)WT1 (Wilms' tumor gene) isoforms on the intrinsic apoptosis pathway. Oncogene 2006; 25: 4217–4229.
Miller Jr WH, Schipper HM, Lee JS, Singer J, Waxman S . Mechanisms of action of arsenic trioxide. Cancer Res 2002; 62: 3893–3903.
Glienke W, Chow KU, Bauer N, Bergmann L . Down-regulation of wt1 expression in leukemia cell lines as part of apoptotic effect in arsenic treatment using two compounds. Leuk Lymphoma 2006; 47: 1629–1638.
Fukumi S, Horiguchi-Yamada J, Nakada S, Nagai M, Ohno T, Yamada H . Differential responses of Bcl-2 family genes to etoposide in chronic myeloid leukemia K562 cells. Mol Cell Biochem 2000; 206: 43–50.
Floros KV, Thomadaki H, Florou D, Talieri M, Scorilas A . Alterations in mRNA expression of apoptosis-related genes BCL2, BAX, FAS, caspase-3, and the novel member BCL2L12 after treatment of human leukemic cell line HL60 with the antineoplastic agent etoposide. Ann NY Acad Sci 2006; 1090: 89–97.
Chalandon Y, Jiang X, Christ O, Loutet S, Thanopoulou E, Eaves A et al. BCR-ABL-transduced human cord blood cells produce abnormal populations in immunodeficient mice. Leukemia 2005; 19: 442–448.
Skorski T, Kanakaraj P, Nieborowska-Skorska M, Ratajczak MZ, Wen SC, Zon G et al. Phosphatidylinositol-3 kinase activity is regulated by BCR/ABL and is required for the growth of Philadelphia chromosome-positive cells. Blood 1995; 86: 726–736.
Stokoe D . The phosphoinositide 3-kinase pathway and cancer. Expert Rev Mol Med 2005; 7: 1–22.
Blume-Jensen P, Hunter T . Oncogenic kinase signalling. Nature 2001; 411: 355–365.
Tuna M, Chavez-Reyes A, Tari AM . HER2/neu increases the expression of Wilms' Tumor 1 (WT1) protein to stimulate S-phase proliferation and inhibit apoptosis in breast cancer cells. Oncogene 2005; 24: 1648–1652.
Bowman T, Garcia R, Turkson J, Jove R . STATs in oncogenesis. Oncogene 2000; 19: 2474–2488.
Fraizer GC, Wu YJ, Hewitt SM, Maity T, Ton CC, Huff V . Transcriptional regulation of the human Wilms' tumor gene (WT1). Cell type-specific enhancer and promiscuous promoter. J Biol Chem 1994; 269: 8892–8900.
Ray S, Lu Y, Kaufmann SH, Gustafson WC, Karp JE, Boldogh I et al. Genomic mechanisms of p210BCR-ABL signaling: induction of heat shock protein 70 through the GATA response element confers resistance to paclitaxel-induced apoptosis. J Biol Chem 2004; 279: 35604–35615.
Zhao W, Kitidis C, Fleming MD, Lodish HF, Ghaffari S . Erythropoietin stimulates phosphorylation and activation of GATA-1 via the PI3-kinase/AKT signaling pathway. Blood 2006; 107: 907–915.
Dame C, Kirschner KM, Bartz KV, Wallach T, Hussels CS, Scholz H . The Wilms' tumor suppressor, Wt1, is a transcriptional activator of the erythropoietin gene. Blood 2006; 107: 4282–4290.
Na IK, Kreuzer KA, Lupberger J, Dorken B, le Coutre P . RT-PCR of Wilms tumor gene transcripts (WT1) for the molecular monitoring of patients with accelerated phase bcr/abl + CML. Leuk Res 2005; 29: 343–345.
Mayo MW, Wang CY, Drouin SS, Madrid LV, Marshall AF, Reed JC et al. WT1 modulates apoptosis by transcriptionally upregulating the bcl-2 proto-oncogene. EMBO J 1999; 18: 3990–4003.
Carrington D, Algar E . Overexpression of murine WT1 +/+ and −/− isoforms has no effect on chemoresistance but delays differentiation in the K562 leukemia cell line. Leuk Res 2000; 24: 927–936.
Kuroda J, Puthalakath H, Cragg MS, Kelly PN, Bouillet P, Huang DC et al. Bim and Bad mediate imatinib-induced killing of Bcr/Abl+ leukemic cells, and resistance due to their loss is overcome by a BH3 mimetic. Proc Natl Acad Sci USA 2006; 103: 14907–14912.
Acknowledgements
We thank Dr E Buchdunger (Novartis, Basel, Switzerland) for providing imatinib mesylate, Dr F Rauscher III (Philadelphia, PA, USA) for providing the WT1 cDNA, Dr C Eaves (Terry Fox Laboratory, Vancouver, Canada) for the gift of MSCV-BCR/ABL1-IRES-GFP vector and Dr G Fraizer (Houston, Texas, USA) for the gift of pCB.7PH, pCB.7e250, pCB.7e258 and pCB.7e250e258 vectors. This work was supported by grants from the Swedish Cancer Society, the Swedish Research Council (project no. 11546), the Swedish Children's Cancer Foundation, the Gunnar Nilsson Cancer Foundation, the Österlund Foundation and from Funds of Lund University Hospital.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Svensson, E., Vidovic, K., Lassen, C. et al. Deregulation of the Wilms' tumour gene 1 protein (WT1) by BCR/ABL1 mediates resistance to imatinib in human leukaemia cells. Leukemia 21, 2485–2494 (2007). https://doi.org/10.1038/sj.leu.2404924
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2404924
Keywords
This article is cited by
-
Curcumin attenuates Adriamycin-resistance of acute myeloid leukemia by inhibiting the lncRNA HOTAIR/miR-20a-5p/WT1 axis
Laboratory Investigation (2021)
-
Identification of TCR Vβ11-2-Dβ1-Jβ1-1 T cell clone specific for WT1 peptides using high-throughput TCRβ gene sequencing
Biomarker Research (2019)
-
Loss of Wilms tumor 1 protein is a marker for apoptosis in response to replicative stress in leukemic cells
Archives of Toxicology (2018)
-
Is Wilms’ tumor gene 1 a useful biomarker for detecting minimal residual disease in chronic myeloid leukemia (BCR-ABL1-p210-positive) patients?
Comparative Clinical Pathology (2016)
-
Wilms’ tumour suppressor gene 1 (WT1) is involved in the carcinogenesis of Lung cancer through interaction with PI3K/Akt pathway
Cancer Cell International (2013)