Abstract
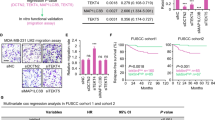
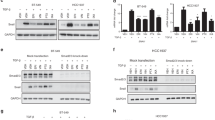
Histone deacetylase (HDAC) 6 is a subtype of the HDAC family; it deacetylates α-tubulin and increases cell motility. Here, we investigate the impact of an alteration of HDAC6 expression in estrogen receptor α (ER)-positive breast cancer MCF-7 cells, as we identified that HDAC6 is a novel estrogen-regulated gene. MCF-7 treated with estradiol showed increased expression of HDAC6 mRNA and protein and a four-fold increase in cell motility in a migration assay. Cell motility was increased to the same degree by stably transfecting the HDAC6 expression vector into MCF-7 cells. In both cases, the cells changed in appearance from their original round shape to an axon-extended shape, like a neuronal cell. This HDAC6 accumulation caused the deacetylation of α-tubulin. Either the selective estrogen receptor modulator tamoxifen (TAM) or the pure antiestrogen ICI 182,780 prevented estradiol-induced HDAC6 accumulation and deacetylation of α-tubulin, leading to reduced cell motility. Tubacin, an inhibitory molecule that binds to the tubulin deacetylation domain of HDAC6, also prevented estradiol-stimulated cell migration. Finally, we evaluated HDAC6 protein expression in 139 consecutively archived human breast cancer tissues by immunohistochemical staining. The prognostic analyses for these patients revealed no significant differences based on HDAC6 expression. However, subset analysis of ER-positive patients who received adjuvant treatment with TAM (n=67) showed a statistically significant difference in relapse-free survival and overall survival in favor of the HDAC6-positive group (P<0.02 and P<0.05, respectively). HDAC6 expression was an independent prognostic indicator by multivariate analysis (odds ratio=2.82, P=0.047). These results indicate the biological significance of HDAC6 regulation via estrogen signaling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Bardou VJ, Arpino G, Elledge RM, Osborne CK and Clark GM . (2003). J. Clin. Oncol., 21, 1973–1979.
Chomczynski P and Sacchi N . (1987). Anal. Biochem., 162, 156–159.
Ciocca DR and Elledge R . (2000). Endocrine, 13, 1–10.
Clark GM, McGuire WL, Hubay CA, Pearson OH and Marshall JS . (1983). N. Engl. J. Med., 309, 1343–1347.
EBCTCG (1998). Lancet, 351, 1451–1467.
Ellis MJ, Coop A, Singh B, Mauriac L, Llombert-Cussac A, Janicke F, Miller WR, Evans DB, Dugan M, Brady C, Quebe-Fehling E and Borgs M . (2001). J. Clin. Oncol., 19, 3808–3816.
Goldhirsch A, Glick JH, Gelber RD, Coates AS and Senn HJ . (2001). J. Clin. Oncol., 19, 3817–3827.
Grozinger CM, Hassig CA and Schreiber SL . (1999). Proc. Natl. Acad. Sci. USA, 96, 4868–4873.
Haggarty SJ, Koeller KM, Wong JC, Grozinger CM and Schreiber SL . (2003). Proc. Natl. Acad. Sci. USA, 100, 4389–4394.
Hubbert C, Guardiola A, Shao R, Kawaguchi Y, Ito A, Nixon A, Yoshida M, Wang XF and Yao TP . (2002). Nature, 417, 455–458.
Inoue A, Yoshida N, Omoto Y, Oguchi S, Yamori T, Kiyama R and Hayashi S . (2002). J. Mol. Endocrinol., 29, 175–192.
Kawaguchi Y, Kovacs JJ, McLaurin A, Vance JM, Ito A and Yao TP . (2003). Cell, 115, 727–738.
Malinda KM, Ponce L, Kleinman HK, Shackelton LM and Millis AJ . (1999). Exp. Cell Res., 250, 168–173.
Omoto Y, Kobayashi Y, Nishida K, Tsuchiya E, Eguchi H, Nakagawa K, Ishikawa Y, Yamori T, Iwase H, Fujii Y, Warner M, Gustafsson JA and Hayashi SI . (2001). Biochem. Biophys. Res. Commun., 285, 340–347.
Osborne CK . (1998). N. Engl. J. Med., 339, 1609–1618.
Palazzo A, Ackerman B and Gundersen GG . (2003). Nature, 421, 230.
Ravdin PM, Green S, Dorr TM, McGuire WL, Fabian C, Pugh RP, Carter RD, Rivkin SE, Borst JR, Belt RJ, Metch B and Osborne CK . (1992). J. Clin. Oncol., 10, 1284–1291.
Saji S, Omoto Y, Shimizu C, Warner M, Hayashi Y, Horiguchi S, Watanabe T, Hayashi S, Gustafsson JA and Toi M . (2002). Cancer Res., 62, 4849–4853.
Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V, Bajamonde A, Fleming T, Eiermann W, Wolter J, Pegram M, Baselga J and Norton L . (2001). N. Engl. J. Med., 344, 783–792.
Verdel A and Khochbin S . (1999). J. Biol. Chem., 274, 2440–2445.
Wade PA . (2001). Hum. Mol. Genet., 10, 693–698.
Yoshida N, Omoto Y, Inoue A, Eguchi H, Kobayashi Y, Kurosumi M, Saji S, Suemasu K, Okazaki T, Nakachi K, Fujita T and Hayashi S . (2004). Cancer Sci., 95, 496–502.
Zhang Z, Yamashita H, Toyama T, Sugiura H, Omoto Y, Ando Y, Mita K, Hamaguchi M, Hayashi S and Iwase H . (2004). Clin. Cancer Res., 10, 6962–6968.
Acknowledgements
We thank Drs Yoko Omoto and Mariko Muta for their help with our experiments. We are grateful to Drs Tso-Pang Yao and Yoshiharu Kawaguchi (Duke University) for their scientific suggestions for our work. This work was supported by the Sato Memorial Foundation for Cancer Research, the Sagawa Cancer Research Fund, and a grant from the Tokyo Metropolitan Government Bureau of Public Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saji, S., Kawakami, M., Hayashi, Si. et al. Significance of HDAC6 regulation via estrogen signaling for cell motility and prognosis in estrogen receptor-positive breast cancer. Oncogene 24, 4531–4539 (2005). https://doi.org/10.1038/sj.onc.1208646
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208646
Keywords
This article is cited by
-
HDAC6 inhibitor ACY-1215 enhances STAT1 acetylation to block PD-L1 for colorectal cancer immunotherapy
Cancer Immunology, Immunotherapy (2024)
-
Selective inhibition of HDAC6 promotes bladder cancer radiosensitization and mitigates the radiation-induced CXCL1 signalling
British Journal of Cancer (2023)
-
Unconventional protein post-translational modifications: the helmsmen in breast cancer
Cell & Bioscience (2022)
-
Structure-based prediction of HDAC6 substrates validated by enzymatic assay reveals determinants of promiscuity and detects new potential substrates
Scientific Reports (2022)
-
HDAC6: A unique HDAC family member as a cancer target
Cellular Oncology (2022)