Abstract
Study design: Review article on spinal involvement of hydatid disease.
Objectives: A better understanding of this rare but clinically challenging disease is intended. An overview of the epidemiology, pathogenesis, presentation and diagnosis of spinal hydatid disease is provided. Management problems and frequent pitfalls are discussed as well as current therapeutic options, results and outcome.
Methods: Thirty-seven reports of spinal hydatid disease published between 1964 and 2000 were reviewed.
Results: Most of the reported cases of spinal hydatid disease presented with spinal cord compression syndrome. Due to the relative rarity of the problem the diagnosis was frequently made during surgery. Surgical intervention by decompressive laminectomy was the most frequent first management. Reports of anterior procedures as well as spinal stabilization exist. Intraoperative prophylaxis to reduce spillage as well as pharmacotherapy were usually instituted. Results of surgery were generally reported to be poor. Progressive neurological and mechanical deterioration over the years was the most frequently reported disease course. Anecdotal reports of alternative management strategies exist.
Conclusion: Spinal hydatid disease should be considered in the differential diagnosis of spinal cord compression syndrome in endemic countries and sought after with imaging and serology. Treatment is based on surgical decompression. Despite therapy the disease frequently relapses with progressive destruction of the vertebral column and neurological deterioration. Retention of spinal stability is the major long term concern. Overall outcome is poor with few reports of disease-free long term survival.
Similar content being viewed by others
Introduction
Hydatid – Greek for ’watery cyst‘ – disease is a rare but significant parasitic disease in endemic areas. Bone involvement is a rare complication of this disease. According to Işlekel et al1 bone disease was first described by Bidloo in 1708, the first description of spinal hydatid disease was made by Churrier in 1807 and the first surgical intervention was reported by Reydellet in 1819.
Despite the introduction of modern surgical and pharmacological therapy the disease still continues to be difficult to cure and highly prone to recurrence. The infestation persistently erodes the spinal column, eventually leading to its destruction and neurological deterioration. The prognosis is poor, comparable to that of malignancies.2
Pathogenesis
Hydatid disease is caused by the larval stage of the tapeworm Echinococcus Granulosus. Far less commonly infestation by Echinococcus Multilocularis (alveolar echinococcus) or Oligoarthus may be responsible for the disease.3,4,5,6
As is the case with other cestodes, Echinococcus has definitive and intermediate hosts. The 3–6 mm long adult worm resides in jejunum of the definitive host: mainly a domestic dog and occasionally other carnivores. The parasite has three proglottids: One immature, one mature and one gravid. The gravid proglottid is shed in the excreta of the dog and ruptures to release 500 to 800 ova which are accidentally ingested by cows, sheep, mice or humans. In the intermediate hosts, after digestion of the chitin, outer layer embryos (oncospheres) develop in the intestine to penetrate the duodenal mucosa and disseminate via the portal circulation. They then either develop into cysts in tissues or are destroyed by the host response. The cysts consist of an outer fibrous layer and a cestode-derived inner germinal membrane containing scolices.3,4,5,6,7,8
The liver is the most commonly involved site, followed by the lungs. Bone involvement is rare and in most of these cases involves the spine. Infestation is thought to begin primarily in the center of the vertebral body and subsequently to extend extraduraly or paravertebraly. Isolated extradural spinal infestation has been reported.9 Secondary extension from paravertebral tissues (costal, mediastinal lesions or cysts involving the Erector Spina muscles) or even from pulmonary cysts are occasionally seen.3,10,11
In 90% of the cases the disease is confined to the bone and the epidural space.6,11,12,13,14,15,16 Intradural extramedullary involvement is found in 9% of the cases.12,13,17,18,19 This can be primary or complicate prior to surgery with dural opening or tear. Occasionally, extension from paravertebrally located cysts is encountered. Intramedullary hydatid disease is described in the literature.20
Pathology
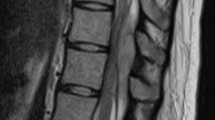
Primary infestation of the spine is thought to occur through the portovertebral shunts and the center of the vertebral body is the first site to be involved.1,21 In contrast to extraosseous involvement, larval penetration of the cancellous bone in the vertebrae causes a mutlivesicular and diffuse infiltration.21 Cortical weakening due to compression or avascular necrosis22,23 may eventually lead to pathological fractures7,13,23 and spread to the pedicles, laminae or the ribs may subsequently occur.7,13 Death of the cyst results in dystrophic calcification, which may be radiographically demonstrated. The intervertebral discs are spared in the early stages of the disease,2 as is the case in tuberculosis. Finally, the infestation may extend beyond the vertebral body and periosteum with the development of a spherical cyst with space occupying symptomatology (Figure 1). Bone destruction and pus-like fluid may be encountered during operation, leading to the name ’ossifluent abscess‘.24
(A–D). A 35-year-old male patient presented to our clinic with recurrence two years after operation for epidurally located spinal hydatid disease. (A, B) T2W sagital and axial images showing multiloculated cysts extending into the thoracic cavity and subcutaneous tissue (arrows). (C) AP pulmonary plain x-ray showing posteriorly located mass lateral to the vertebral body (arrows). (D) T2W coronal MR image shows pulmonary extension of the multiloculated cyst. (arrow)
Incidence
Although rare in developed countries, the disease continues to be a significant health problem in endemic areas.13,16,25 The disease is especially prevalent in areas where livestock is raised in association with dogs: Mediterranean, eastern European, east African, central Asian and South American countries, Australia and New Zealand. The disease is rare in Western Europe and north America, but reviewing the cases in North America, Rao et al25 noted that sporadic cases from California, Utah, Lower Mississippi valley, Alaska and northwest Canada were reported. Sapkas et al7 stated that the infestation may be seen anywhere in the world due to frequent overseas travel.
In contrast to visceral disease, bone or central nervous system involvement occurs much less frequently. Only 0.5 to 3.1% of patients suffer from bone involvement, half of which occurs in the spine.1,12,13,16,17,26
Spinal hydatitosis is a significant cause of spinal cord compression syndrome in endemic areas with incidences ranging from 1.1 in Australia to as high as 14% in Tunisia.1,2,7,12,16,17,21,23,24,25 Spinal hydatid disease in Turkey is responsible for 3.8% of all spinal cord compression syndromes.12
Spinal disease most frequently involves the thoracic segments (46% to 50%). Lumbar (20% to 29%) and sacral (20% to 23%) diseases also occur.1,7,10,12,13,14,15,16,17,21,25,27,28 Cervical spine is the least commonly involved area.
Clinical manifestations
Usually clinical manifestations appear long after the initial infestation as cysts grow slowly over a period of several years hampered by the resistance of the surrounding bone. There are no characteristic signs or symptoms. A long history of back pain and/or signs of spinal cord compression are the usual manifestations. Back pain was present in 85%, radicular pain in 25% to 95% and paraparesis in 25% to 77% of patients.12,21,25 Cauda equina syndrome also may be noted.2,15 It is noted that a history of trauma was present in up to 20% of the cases.1 Özek et al21 reported that rapid onset of neurological dysfunction is due to vascular compromise and associated with a slow and incomplete recovery. Baysefer et al25 noted that severe paraparesis is usually secondary to epidural disease and that this is associated with a more favorable prognosis.
Paraplegia due to recurrent disease is reported to be as high as 45%.12,23,26
Rupture of a cyst with spillage of the content may provoke a variety of hypersensitivity reactions such as pruritus, urticaria, edema, dyspnoea, asthma, vomiting, diarrhea, colicky abdominal pain and even anaphylactic shock.3,5
Secondary bacterial infection may occur and further complicates the diagnosis.13,21 This should be considered whenever fever and irritability are present21 (Figure 2).
(A, B). A 21-year-old female patient presented to our clinic with motor weakness and presthesias in the lower extremities and a dermal fistula in the back with purulent discharge. The patient was shown to have secondary pyogenic infection of spinal hydatid cysts. (A): T2W NMR image showing epidural spinal cysts in dorsal spine. (B) Contrast enhanced T1W axial MR image shows epidurally located hydatid cyst (arrows). Note that the spinal cord is displaced anteriorly and laterally
Diagnosis
Spinal hydatid disease should be considered in the differential diagnosis of spinal cord compression syndrome in endemic countries. Due to the relative rarity of the entity most diagnoses are made intraoperatively which increases the risk of future recurrences. As the choice of the most suitable surgical approach relies on correct diagnosis and because special measures are required to prevent intraoperative spillage, preoperative diagnosis is of high importance. The differential diagnosis should include tuberculosis, pyogenic infections, fibrous dysplasia, enchondroma, metastatic malignancy, multiple myeloma and giant cell tumor.7
Serodiagnostic tests may be used for preoperative diagnosis in suspected cases.4 Detection of antibody directed to echinococcal polypeptide antigens has the highest degree of specificity.4,5 ELISA, indirect hemagglutination and complement fixation tests are reported to be 80% to 100% sensitive and 88% to 96% specific in abdominal disease.5 However, the sensitivity decreases abruptly to 25% to 56% in extrahepatic disease which limits their use in the diagnosis or follow up for primary bone disease.5 Second line tests like immunoblotting (Antigen 5 precipitation: Arc-5 Test) may be used for confirmation after ELISA and hemagglutination.4 Imaging is more sensitive than serodiagnosis in spinal hydatid disease.6 A negative serology should not exclude the diagnosis of hydatid disease in the presence of suggestive imaging results.5,6 Diagnostic fine needle aspiration is usually avoided because of the risk of spillage or anaphylaxis due to cyst rupture. Not uncommonly, the final diagnosis is delayed until the surgery and direct visualization of the lesions.1,3,7,25
Other laboratory tests are usually not informative except that eosinophilia may be noted in the peripheral blood.29
Although plain X-rays are not diagnostic of the disease, ’moth eaten‘ lesions with surrounding sclerosis and calcifications extending into paravertebral soft tissues, when present, are highly suggestive. Bone changes are reported to be present in only 27% of cases.12 Compression fractures with loss of the corpus height or erosion of the pedicles may be noted.7,12
Myelography, which is of little diagnostic value, carries the risk of cyst puncture with intradural dissemination or anaphylaxis and therefore should be avoided.6
CT has excellent bone resolution and usually shows irregular erosions of the cancellous bone devoid of any subperiosteal reaction or enlargement of the vertebra.1,12,21,25,27 Tekkök and Benli11 concluded that CT myelography, although inferior to MRI in showing paravertebral soft tissue extension is of great diagnostic value in showing cysts in bone and their intraspinal extension.
The MRI with its superior soft tissue resolution is the most sensitive diagnostic method as well as the method of choice for determination of the extent of the disease.6,16,27,30 Berk et al30 reviewed MRI characteristics of the lesions and concluded that they had a unique appearance: A sausage-like shape with two dome shaped ends, thin and regular walls and no septation or debris in the lumen. The lesions are occasionally spherical. Signal characteristics of the cyst content are usually similar to that of CSF.27,30,31 On T1W images the cyst wall appears iso- or slightly more hypo-intense than the cyst content and enhances slightly after contrast injection. T2W images demonstrate a low intensity rim which correlates to the pathological findings of reactive fibrosis and degeneration surrounding the parasitic membrane. It has also been reported that viability of the cysts could be determined by their MRI characteristics.30 Namely, on T2W images cysts tend to lose their hyperintensity and the walls tend to lose their hypointense signal upon inactivation.13,30,31 The authors noted that the differential diagnosis included dorsal arachnoid diverticula, dorsal intrathoracic and lateral thoracic meningocoeles.
The CT and MRI provide complementary data both in diagnosis and follow up of patients with spinal echinococcosis.21,27,30 Early postoperative MRI is valuable in demonstrating lesions missed during the operation.18 However, the diagnosis of recurrent disease should not rely solely on imaging but should also be based on symptomatology.6
Head, thorax and abdomen should be scanned for extra-spinal cysts upon the diagnosis of hydatid disease.6,32
Treatment
Surgery
Surgery is currently considered the treatment of choice for spinal hydatid disease.1,12,21,25 Provided that most of the cases are admitted with signs and symptoms of spinal cord compression syndrome, urgent surgery is validated and most surgeons act accordingly.12,26,27
Preoperative diagnosis of spinal hydatid disease, as well as the determination of its extent, is of crucial importance for planning the operative approach. Posterior or anterior approaches may be considered.
Pure intradurally or pure epidurally located lesions in all spinal levels are approached posteriorly. In these cases surgical excision without intraoperative rupture results in cure.12,17,18
Bone involvement is a major challenge for the treating surgeon. Due to the infiltrative nature of the lesion, spillage of scolices is unavoidable during the surgery for lesions involving the bone.1,12,21,25 Even laminectomy may result in rupture of the cysts and spillage.1 It must be kept in mind that the disease is highly prone to recurrence, and plans for the surgical approach and stabilization must be made accordingly.38
Simple decompression with laminectomy was by far the most commonly reported procedure.1,7,10,11,12,14,15,18,22,25,26,27,29,33,34,35,36,37 Except for one case,12 all cases in the literature were first managed by laminectomy. This approach allows simple and easy decompression with little morbidity and mortality. However, access to the vertebral body is limited which may be a problem especially in recurrent disease. Stability of the anterior column must be ascertained to prevent postoperative kyphosis. Some authors advocate a more radical approach with as much bone removal as possible.1 Radical removal is not shown to preclude disease progression. However, extensive resection with stabilization and grafting is shown to prolong survival in selected cases.38
Several procedures were reported in the cervical region. Posterior approaches including laminectomy, laminectomy with lateral mass plating were reported to be effective with low or no morbidity.22,38 Reported anterior approaches were corpectomy with simple bony fusion and halo bracing and corpectomy with anterior plating.22 In the only reported case of anterior corpectomy with simple bony fusion and external halo bracing22 the authors reported development of postoperative kyphosis with progressive myelopathy. The patient was successfully managed by lateral mass plating.
Operations in thoracic cases were mainly posterior.1,7,10,12,14,25,26,29 More extensive lesions with involvement of the vertebral body can be approached via a transpedicular route.26 Three cases of anterior approach to thoracic lesions were reported.1,7,12 The first patient had presented with a pathologic compression fracture of the T9 vertebral body. The patient underwent transthoracic corpectomy with anterior fusion.12 An autologous costal graft was well tolerated by the patient who was asymptomatic at the third year follow-up. The second patient7 was first approached posteriorly with laminectomy and transpedicular curettage. However, cysts were noted to extend into the thoracic cavity via neural foraminae and a second anterior procedure was carried out after posterior fusion. Reportedly the patient did well after the operation but suffered local recurrence with complete paraplegia 1 year after the operation and succumbed to the disease 2 years later having denied further therapy. In the third case,1 cysts residing anterior to the vertebral body were simply evacuated with costotransversectomy.
The lumbar region was the second most commonly reported disease location and simple laminectomy was the most common surgery performed.1,11,12,18,29,38 Retroperitoneal abdominal corpectomy with posterior fusion with subsequent surgery for total destruction of the vertebral body is reported in lumbar disease.2 No long-term outcome was included in the report regarding disease spread to retroperitoneal tissues.
Five cases of bone fusion were reported in cases with spinal hydatid disease.7,12,17,22,25 Autologous pelvic and costal bone grafts were reported. Bhojraj et al26 speculated that extensive fusion with decortication might result in spread to uninvolved adjacent bone and reported a successful stabilization with a Luque system and sublaminar wiring after laminectomy and transpedicular curretage. However, the authors provided follow-up data of only 3 weeks which makes the evaluation of their hypothesis impossible. Sapkas et al7 reported his concerns about the possible infestation of graft material and advocated the use of acrylic cement for osteosynthesis. However, no cases are reported. Keller et al22 reported successful graft fusion and preservation of spinal stability even in the presence of verified active disease. Recurrence was noted in three of these cases. In one case, long-term outcome was not reported.4 In another, recurrence was seen 1 year7 and in the third, 3 years22 after bone fusion. Spinal stabilization was regained after revision in one of the cases.22 The other case7 reportedly refused therapy and died of disease progression 2 years after recurrence.
Complications
Reported surgical complications are perioperative death, misdiagnosis with local spread, recurrence, increasing neurological deficit, secondary infection, CSF fistula and anaphylaxis.12,13 A mortality rate of 0 to 3% is reported which progressively increases to 15% in subsequent reoperations.1,12,13,21 Extreme caution should be taken not to rupture intradural or epidural solitary cysts as complete removal might potentially result in cure. Işlekel1 reported a case misdiagnosed as a dermoid cyst. In this case, dural opening was reported to result in intradural invasion and recurrences. Although the effectivity is not proven13,38 most surgeons advocate intraoperative scolicidal agents as well as adjuvant pharmacotherapy. Scolicidal agents like 3% hypertonic saline, chlorhexidine, 80% alcohol, 0.5% silver nitrate or 10% formalin are used in abdominal disease. Use of Povidone iodide15 as a scolicidal agent is reported in spinal cases but long-term results are lacking. Hypertonic saline is the most frequently used agent in spinal disease. Işlekel et al1 reported an operative mortality due to toxic myelitis caused by formalin after a dural tear. Formalin irrigation is also reported to add to the morbidity by increasing tissue necrosis13 and should be avoided in spinal cases.
Recurrence
Unfortunately even with extensive surgery results are far from being curative and recurrences are the rule.1,2,12,21,29,38,39,40 Up to 40% of patients are reported to have recurrent symptomatology within 2 years of initial operation.2,17,21,29 In recurrent lesions the major goal is restoration and preservation of neurological status and spinal stability. Several authors advocate the reinstitution of antihelmintic pharmacotherapy which should be continued indefinitely.21,22 Presence of disease spread to paravertebral tissues further warrants the use of adjuvant therapy.21 However, most patients experience a downhill course with worsening neurological status and progressive destruction of the vertebral column with multiple recurrences.21,38 Incidence of paraplegia in recurrent disease is reported to be as high as 45%.12,34 Recurrence is considered a bad prognostic factor in spinal hydatid disease.21 Operative mortalities of up to 14.4% were reported in recurrent cases.12 Keller et al22 reported a cervical case operated on six times with preservation of spinal stability and neurological status. The authors concluded that despite multiple recurrences, spinal stability could be achieved and should be the goal.
Minimally invasive techniques
Diagnostic or therapeutic CT guided fine needle aspiration of hydatid cysts is frequently used in abdominal hydatid disease.39,41 This technique however is usually avoided in spinal disease due to the risk of cyst rupture and severe anaphylaxis.13 In the single reported case, Spektor et al41 described the successful decompression of an extradural cervical spinal hydatid cyst by CT guided fine needle aspiration with complete resolution of quadriplegia. The authors advocate that with adequate prophylaxis against anaphylaxis and dissemination, CT guided aspiration of spinal hydatid cysts with hypertonic saline irrigation and subsequent chemotherapy may be an alternative to surgical therapy in selected cases.39,41 Other minimally invasive techniques such as endoscopic evacuation of cyst material has been reported in the literature.31 We think that minimally invasive methods may be of value in selected patients with localized spinal disease, in whom surgical therapy is contraindicated. However, these methods are still experimental and the efficacy and safety have not been determined.
Simple drainage of the infested cystic area is reported to result in pyogenic osteomyelitis and is strongly discouraged.13
Pharmacotherapy
Drug therapy with benzimidazole derivatives is proven to be effective in abdominal disease.8 A course of Albendazole (800 mg daily in two divided doses) is continued for 1 to 6 months (usually 3 months). Albendazole is preferred over Mebendazole due to better pharmacokinetic properties and superior efficacy against helminths.2,9 While these agents do not cure the bone disease they are used in conjunction with surgery to decrease avoid recurrences due to intraoperative spillage.7,9,12 Their effectiveness in preventing recurrence is also debatable. There are reports, however, that they may be effective in inoperable patients, prolonging the length of survival.2 It should be kept in mind that both of these drugs are teratogenic and embriotoxic, and both may cause alterations in liver function and hematological adverse reactions.5 It is reported that the drug Praziquantel is also effective against Echinococci alone or in combination with Albendazole. Lam et al2 reported an inoperable case of recurrent spinal hydatitosis treated with a combination of Albendazole plus Praziquantel having survived for 34 years.
Prognosis
Even with the use of scolicidal agents like hypertonic saline or formalin, intraoperative seeding is commonly leading to recurrence rates up to 40%.12,13 Recurrences occur 2 to 28 months (mean: 25.2 months) after the initial operation.1,21 The recurrence rate for intradural extramedullary disease is reported to be low.12
The high recurrence rate is primarily responsible for the poor survival of only 5 years after the onset of symptoms.2,7 Lam et al2 reported that the average age at death is 41 years. Operative mortality rates up to 15% are noted in the literature and are known to increase with subsequent operations. Paraplegia due to recurrent disease is reported to be as high as 45%.12,26
Conclusion
Spinal hydatid disease, although rare in developed countries, must be considered in the differential diagnosis of spinal cord compression syndrome especially in endemic areas. Surgical decompression is the treatment of choice. Due to the invasive nature of the disease, surgery alone is seldom curative. Adjuvant drug therapy as well as intraoperative prophylaxis are indicated. Despite all measures, the disease has a high propensity for recurrence and leads to progressive destruction of the vertebral column and neurological deterioration. Instrumentation with bony fusion may be required to preserve spinal stability. Despite aggressive treatment, overall outcome is still poor leading to a prognosis measured by years.
References
İşlekel S et al. Spinal hydatid disease Spinal Cord 1998 36: 166–170
Lam KS, Faraj A, Mulholland RC, Finch RG . Medical decompression of vertebral hydatidosis Spine 1997 22: 2050–2055
Sparks AK, Connor DH, Neafie RC . Echinococcosis In: Binford CH, Connor DH (eds) Pathology of Tropical and Extraordinary Diseases Vol.2: AFIP: Washington D.C. 1976 pp 530–534
Lightfowlers M, Goldstein B . Immunodiagnosis of Echinococcus In: Thompson RCA, Lymberty AJ (eds) Echinococcus and Hydatidosis Wallingford: CAB intern 1995 pp 355–410
King C . Cestodes In: Mandel GL, Bennet JE, Dolin R (eds) Principles and practice of Infectious diseases 4th edn Churchill & Livingstone 2000 pp 2957–2965
Killough KK, Lusbough WB, Harkey HL . Parasitic infestations of the spine In: Osenbach RK, Zeidman SM (eds) Infections in Neurosurgery Lippincott & Raven: Philadelphia 1999 pp 305–339
Sapkas GS, Stathakopulos DP, Babis GC, Tsarouchas JK . Hydatid disease of bones and joints Acta Orthop Scand 1998 69: 89–94
Gil-Grande L et al. Randomised controlled trial of efficacy of albendazole in intraabdominal hydatid diasease Lancet 1993 342: 1269–1272
Fiennes AG, Thomas DG . Combined medical and surgical treatment of spinal hydatid disease: a case report J Neurol Neurosurg Psychiatry 1982 45: 927–931
İplikçioglu AC et al. Spinal invasion of pulmonary hydatidosis: Computed tomographic demonstration Neurosurgery 1991 29: 467–468
Tekkök H, Benli K . Primary spinal extradural hydatid disease: report of a case with magnetic resonance chararcteristics and pathological correlation Neurosurgery 1993 33: 320–323
Pamir MN, Akalan N, Özgen T, Erbengi A . Spinal hydatid cysts Surg Neurol 1984 21: 54–57
Rao S, Parikh S, Kerr R . Echinococcal infestation of the spine in North America Clin Orthop Relat Res 1991 271: 164–169
Baybek M, İnci S, Tahta K, Bertan V . Primary multiple spinal extradural hydatid cysts of the literature: Case report and review Paraplegia 1992 30: 517–519
Pandey M, Cahudhari MP . Primary hydatid cyst of sacral spinal canal: case report Neurosurgery 1997 40: 407–409
Altinörs N, Bavbek M, Caner H, Erdoğan B . Central nervous system hydatitosis in Turkey: a cooperative study and literature survey analysis of 458 cases J Neurosurg 2000 93: 1–8
Bettaieb A, Khalid T, Ben Rhouma T, Touibi S . L'echinococcose vertebro-medullarie Neurochirurgie 1978 24: 205–210
İşlekel S, Zileli M, Erşahin Y . Intradural spinal hydatid cysts Eur Spine J 1998 7: 162–164
Sharma A, Kashyap V, Abraham J, Kurian S . Intradural hydatid cysts Surg Neurol 1981 16: 235–237
Ley A, Marti A . Intramedullary hydatid cyst J Neurosurg 1970 33: 257–259
Özek MM . Complications of central nervous system hydatid disease Pediatr Neurosurg 1994 20: 84–91
Keller TM, Schweitzer JS, Helfend LK, Chappel T . Treatment of progressive cervical spinal instability secondary to hydatid disease Spine 1997 22: 915–919
Mills TJ . Paraplegia due to hydatid disease J Bone Joint Surg 1956 38B: 884–890
Rayport M, Wisoff HS, Zaiman H . Vertebral echinococcus J Neurosurg 1964 21: 647–659
Baysefer A et al. Hydatid disease of the spine Spinal Cord 1996 34: 297–300
Bhojraj SJ, Shetty NR . Primary hydatid disease of the spine: An unusual cause of progessive paraplegia J Neurosurg (Spine 2) 1999 91: 216–218
Özer F, Özek MM, Pamir MN, Erzen C . Magnetic resonance imaging in the diagnosis of spinal hydatid cyst disease Paraplegia 1993 31: 338–340
Turgut M . Hydatid disease of the spine: a survey from Turkey Infection 1997 25: 221–226
Kaoutzanis M, Anagnostopoulos D . Hydatid disease affecting the vertebrae ActaNeurochir (Wien) 1989 98: 60–65
Berk Ç, Çiftçi, Erdoğan A . MRI in primary intraspinal extradural hydatid disease: case report Neuroradiology 1998 40: 390–392
Açikgöz B et al. Endoscopic evacuation of Sacral hydatid cysts: case report Spinal cord 1996 34: 361–364
Ranganadham P . Posterior mediastinal paravertebral hydatid cyst prtesenting as spinal compression Clin Neurol Neurosurg 1990 2-2: 149–151
Murray RO, Haddad F . Hydatid disease of the spine J Bone Joint Surg 1959 41: 499–506
Morshed AA . Hydatid disease of spine Neurochirurgia 1977 20: 211–215
Malloch JD . Hydatid disease of spine Br Med J 1965 1: 633
Wani et al. Primary spinal extradural hydatid cyst Neurosurgery 1989 4: 631–632
Charles RW, Govender S, Naidoo KS . Echinococcal infection of the spine with neural involvement Spine 1988 1: 47–49
Turtas S, Sehrbundt Viale E, Pau A . Long term results of surgery for hydatid disease of the spine Surg Neurol 1980 13: 468–470
Sinner WNV, Nyman R, Linjawi T, Ali AM . Fine needle aspiration biopsy of hydatid cysts Acta Radiologica 1995 36: 168–172
Robinson RG . Hydatid disease of the spine and its neurological complications Br J Surg 1959 47: 301–306
Spektor S, Gomori JM, Beni-Adani L, Constantini S . Spinal echinococcal cyst: treatment using computerized tomography guided needle aspiration and hypertonic saline irrigation J Neurosurg 1997 87: 464–467
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pamir, M., Ozduman, K. & Elmaci, I. Spinal hydatid disease. Spinal Cord 40, 153–160 (2002). https://doi.org/10.1038/sj.sc.3101214
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.sc.3101214
Keywords
This article is cited by
-
Hydatid disease of the brain and spine
Child's Nervous System (2023)
-
Twenty-six years of involvement with cystic echinococcosis: a case report
Journal of Medical Case Reports (2021)
-
Isolated primary spinal epidural hydatid cyst in a child with progressive paraparesis
Child's Nervous System (2021)
-
Hydatid disease (Echinococcus) of the central nervous system
Child's Nervous System (2018)
-
Primary intramedullary hydatid cyst: a case report and literature review
European Spine Journal (2017)