Abstract
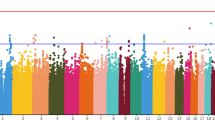
QT prolongation is associated with increased risk of cardiac arrhythmias. Identifying the genetic variants that mediate antipsychotic-induced prolongation may help to minimize this risk, which might prevent the removal of efficacious drugs from the market. We performed candidate gene analysis and five drug-specific genome-wide association studies (GWASs) with 492K single-nucleotide polymorphisms to search for genetic variation mediating antipsychotic-induced QT prolongation in 738 schizophrenia patients from the Clinical Antipsychotic Trial of Intervention Effectiveness study. Our candidate gene study suggests the involvement of NOS1AP and NUBPL (P-values=1.45 × 10−05 and 2.66 × 10−13, respectively). Furthermore, our top GWAS hit achieving genome-wide significance, defined as a Q-value <0.10 (P-value=1.54 × 10−7, Q-value=0.07), located in SLC22A23, mediated the effects of quetiapine on prolongation. SLC22A23 belongs to a family of organic ion transporters that shuttle a variety of compounds, including drugs, environmental toxins and endogenous metabolites, across the cell membrane. This gene is expressed in the heart and is integral in mouse heart development. The genes mediating antipsychotic-induced QT prolongation partially overlap with the genes affecting normal QT interval variation. However, some genes may also be unique for drug-induced prolongation. This study demonstrates the potential of GWAS to discover genes and pathways that mediate antipsychotic-induced QT prolongation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Viskin S, Justo D, Halkin A, Zeltser D . Long QT syndrome caused by noncardiac drugs. Prog Cardiovasc Dis 2003; 45: 415–427.
Zeltser D, Justo D, Halkin A, Prokhorov V, Heller K, Viskin S . Torsade de pointes due to noncardiac drugs: most patients have easily identifiable risk factors. Medicine (Baltimore) 2003; 82: 282–290.
Ray WA, Meredith S, Thapa PB, Meador KG, Hall K, Murray KT . Antipsychotics and the risk of sudden cardiac death. Arch Gen Psychiatry 2001; 58: 1161–1167.
Kane JM, Marder SR . Psychopharmacologic treatment of schizophrenia. Schizophr Bull 1993; 19: 287–302.
Kane JM, McGlashan TH . Treatment of schizophrenia. Lancet 1995; 346: 820–825.
Glassman AH, Bigger Jr JT . Antipsychotic drugs: prolonged QTc interval, torsade de pointes, and sudden death. Am J Psychiatry 2001; 158: 1774–1782.
Taylor DM . Antipsychotics and QT prolongation. Acta Psychiatr Scand 2003; 107: 85–95.
Yerrabolu M, Prabhudesai S, Tawam M, Winter L, Kamalesh M . Effect of risperidone on QT interval and QT dispersion in the elderly. Heart Dis 2000; 2: 10–12.
Paulussen AD, Gilissen RA, Armstrong M, Doevendans PA, Verhasselt P, Smeets HJ et al. Genetic variations of KCNQ1, KCNH2, SCN5A, KCNE1, and KCNE2 in drug-induced long QT syndrome patients. J Mol Med 2004; 82: 182–188.
Arking DE, Pfeufer A, Post W, Kao WH, Newton-Cheh C, Ikeda M et al. A common genetic variant in the NOS1 regulator NOS1AP modulates cardiac repolarization. Nat Genet 2006; 38: 644–651.
Aarnoudse AJ, Newton-Cheh C, de Bakker PI, Straus SM, Kors JA, Hofman A et al. Common NOS1AP variants are associated with a prolonged QTc interval in the Rotterdam Study. Circulation 2007; 116: 10–16.
Post W, Shen H, Damcott C, Arking DE, Kao WH, Sack PA et al. Associations between genetic variants in the NOS1AP (CAPON) gene and cardiac repolarization in the old order Amish. Hum Hered 2007; 64: 214–219.
Tobin MD, Kahonen M, Braund P, Nieminen T, Hajat C, Tomaszewski M et al. Gender and effects of a common genetic variant in the NOS1 regulator NOS1AP on cardiac repolarization in 3761 individuals from two independent populations. Int J Epidemiol 2008; 37: 1132–1141.
Pfeufer A, Sanna S, Arking DE, Muller M, Gateva V, Fuchsberger C et al. Common variants at ten loci modulate the QT interval duration in the QTSCD Study. Nat Genet 2009; 41: 407–414.
Newton-Cheh C, Eijgelsheim M, Rice KM, de Bakker PI, Yin X, Estrada K et al. Common variants at ten loci influence QT interval duration in the QTGEN Study. Nat Genet 2009; 41: 399–406.
Volpi S, Heaton C, Mack K, Hamilton JB, Lannan R, Wolfgang CD et al. Whole genome association study identifies polymorphisms associated with QT prolongation during iloperidone treatment of schizophrenia. Mol Psychiatry 2009; 14: 1024–1031.
Lieberman JA, Stroup TS, McEvoy JP, Swartz MS, Rosenheck RA, Perkins DO et al. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med 2005; 353: 1209–1223.
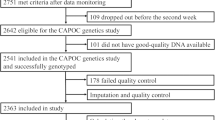
Stroup TS, McEvoy JP, Swartz MS, Byerly MJ, Glick ID, Canive JM et al. The National Institute of Mental Health Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) project: schizophrenia trial design and protocol development. Schizophr Bull 2003; 29: 15–31.
First MB, Spitzer R, Gibbon M, Williams J . Structured Clinical Interview for DSM IV Axis I Disorders—Administration Booklet. American Psychiatric Press, Inc.: Washington D.C.:, 1994.
Bazett H . An analysis of the time-relations of electrocardiograms. Heart 1920; 7: 353–370.
Sullivan PF, Lin D, Tzeng JY, van den Oord E, Perkins D, Stroup TS et al. Genomewide association for schizophrenia in the CATIE study: results of stage 1. Mol Psychiatry 2008; 13: 570–584.
van den Oord EJ, Adkins DE, McClay J, Lieberman J, Sullivan PF . A systematic method for estimating individual responses to treatment with antipsychotics in CATIE. Schizophr Res 2009; 107: 13–21.
Aberg K, Adkins DE, Bukszar J, Webb BT, Caroff SN, Miller del D et al. Genomewide association study of movement-related adverse antipsychotic effects. Biol Psychiatry 2010; 67: 279–282.
McClay JL, Adkins DE, Aberg K, Stroup S, Perkins DO, Vladimirov VI et al. Genome-wide pharmacogenomic analysis of response to treatment with antipsychotics. Mol Psychiatry 2009; e-pub ahead of print 1 September 2009; doi:10.1038/mp.2009.89.
Goldstein H . Multilevel statistical models. Arnold: London, 1995.
Searle SR, Casella G, McCulloch CE . Variance components. Wiley: New York, 1992.
Pinheiro JC, Bates DM . Mixed-Effects Models in S and S-Plus. Springer: New York, NY, 2000.
Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, Bender D et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 2007; 81: 559–575.
Price AL, Patterson NJ, Plenge RM, Weinblatt ME, Shadick NA, Reich D . Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet 2006; 38: 904–909.
Storey JD . The positive false discovery rate: A Bayesian interpretation and the q-value. Annals of Statistics 2003; 31: 2013–2035.
Storey JD, Tibshirani R . Statistical significance for genomewide studies. Proc Natl Acad Sci USA 2003; 100: 9440–9445.
van den Oord EJ, Sullivan PF . False discoveries and models for gene discovery. Trends Genet 2003; 19: 537–542.
Bukszar J, McClay J, van den Oord E . Estimating the posterior probability that genomewide association findings are true or false. Bioinformatics 2009, Jul 14.
Glonek G, Soloman P . Discussion of resampling-based multiple testing for microarray data analysis by ge, dudoit and speed. Test 2003; 12: 1–77.
Evans WE, Johnson JA . Pharmacogenomics: the inherited basis for interindividual differences in drug response. Annu Rev Genomics Hum Genet 2001; 2: 9–39.
Devlin B, Roeder K . Genomic control for association studies. Biometrics 1999; 55: 997–1004.
Koepsell H, Endou H . The SLC22 drug transporter family. Pflugers Arch 2004; 447: 666–676.
Jacobsson JA, Haitina T, Lindblom J, Fredriksson R . Identification of six putative human transporters with structural similarity to the drug transporter SLC22 family. Genomics 2007; 90: 595–609.
Christoforou N, Miller RA, Hill CM, Jie CC, McCallion AS, Gearhart JD . Mouse ES cell-derived cardiac precursor cells are multipotent and facilitate identification of novel cardiac genes. J Clin Invest 2008; 118: 894–903.
Chang KC, Barth AS, Sasano T, Kizana E, Kashiwakura Y, Zhang Y et al. CAPON modulates cardiac repolarization via neuronal nitric oxide synthase signaling in the heart. Proc Natl Acad Sci USA 2008; 105: 4477–4482.
Acknowledgements
This work was supported by the National Institute of Mental Health (grant number N01 MH90001). Eli Lilly funded the GWAS genotyping done at Perlegen Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr PF Sullivan reports receiving research funding from Eli Lilly in connection with this project. Dr TS Stroup reports receiving consulting funds from Janssen and Lilly. Dr D Perkins reports having received research funding from AstraZeneca Pharmaceuticals LP, Bristol-Myers Squibb, Otsuka Pharmaceutical, Eli Lilly, Janssen Pharmaceutica Products and Pfizer; and consulting and educational fees from AstraZeneca Pharmaceuticals LP, Bristol-Myers Squibb, Eli Lilly, Janssen Pharmaceuticals, GlaxoSmithKline, Forest Labs, Pfizer Inc and Shire. Dr JA Lieberman reports having received research funding from AstraZeneca Pharmaceuticals, Bristol-Myers Squibb, GlaxoSmithKline, Janssen Pharmaceutica, Wyeth, Merck, Forest Labs, Allon and Pfizer, has participated in research, consulting, advisory board or DSMB activities from Chephalon, Eli Lilly, Pfizer, Bioline, Lilly, AstraZeneca, Forest Labs, GlaxoSmithKline, Janssen Pharmaceutica, Otsuka, Wyeth and Solvay, and a patent with Repligen. Drs K Aberg, JL McClay, Y Liu, DE Adkins, J Bukszár, P Jia, Z Zhao and EJCG van den Oord report no financial relationships with commercial interests and have nothing to disclose.
Additional information
Supplementary Information accompanies the paper on the The Pharmacogenomics Journal website
Supplementary information
PowerPoint slides
Rights and permissions
About this article
Cite this article
Åberg, K., Adkins, D., Liu, Y. et al. Genome-wide association study of antipsychotic-induced QTc interval prolongation. Pharmacogenomics J 12, 165–172 (2012). https://doi.org/10.1038/tpj.2010.76
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/tpj.2010.76
Keywords
This article is cited by
-
Delineating significant genome-wide associations of variants with antipsychotic and antidepressant treatment response: implications for clinical pharmacogenomics
Human Genomics (2020)
-
Investigation of the SLC22A23 gene in laryngeal squamous cell carcinoma
BMC Cancer (2018)
-
QTc prolongation in short-term treatment of schizophrenia patients: effects of different antipsychotics and genetic factors
European Archives of Psychiatry and Clinical Neuroscience (2018)
-
Pharmakogenetik in der Psychiatrie: eine Standortbestimmung
Der Nervenarzt (2018)
-
Drug–gene interactions and the search for missing heritability: a cross-sectional pharmacogenomics study of the QT interval
The Pharmacogenomics Journal (2014)