Abstract
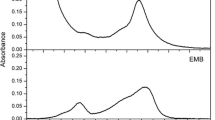
Chlorin-e6 (chl-e6) and a hydrogenated derivative (chl-e6H) were semi-synthesized, and their photophysical properties and photodynamic activity against Escherichia coli, Staphylococcus aureus and Candida albicans evaluated. Methyl pheophorbide-a (Mepheo-a) was obtained from S. maxima using methanolic extraction with acid catalysis (CH3OH—H2SO4). Chlorin-e6 was prepared from Mepheo-a by basic hydrolysis with H2O―acetone and NaOH. Hydrogenated Chlorin-e6 was synthesized by a similar procedure starting from the hydrogenated methyl pheophorbide-a (Mepheo-aH). Photophysical studies were performed in order to determine the singlet oxygen quantum yield of chl-e6H which is higher than that of chl-e6. The microorganism inactivation of chl-e6 and chl-e6H was investigated at two concentrations and three fluence levels. Both chl-e6 and chl-e6H showed microorganism inactivation against Gram-positive bacteria and a fungus.
Similar content being viewed by others
Notes and references
R. R. Allison, V. S. Bagnato, R. Cuenca, G. H. Downie, C. H. Sibata, The future of photodynamic therapy on oncology, Future Oncol., 2006, 2, 53–71.
K. Plaetzer, B. Krammer, J. Berlanda, F. Berr, T. Kiesslich, Photophysics photochemistry of photodynamic therapy: fundamental aspects, Laser Med. Sci., 2009, 24, 259–268.
D. J. Granville, B. M. McManus, D. W. C. Hunt, Photodynamic therapy: shedding light on the biochemical pathways regulating porphyrin-mediated cell death, Histol. Histopathol., 2001, 16, 309–317.
M. R. Hamblin, T. Hasan, Photodynamic therapy: a new antimicrobial approach to infectious disease, Photochem. Photobiol. Sci., 2004, 3, 436–450.
G. Jori, C. Fabris, M. Soncin, S. Ferro, O. Coppellotti, D. Dei, L. Fantetti, G. Chiti, G. Roncucci, Photodynamic therapy in the treatment of microbial infections: basic principles and perspective applications, Laser Surg. Med., 2006, 38, 468–481.
X. Zhang, W. Guo, Imidazole functionalized magnesium phthalocyanine photosensitizer: modified photophysics, singlet oxygen generation and photooxidation mechanism, J. Phys. Chem., 2012, 116, 7651–7657.
P. Calzavara-Pinton, M. T. Rossi, R. Sala, M. Venturini, Photodynamic antifungal chemotherapy, Photochem. Photobiol., 2012, 88, 512–522.
R. Ackroyd, C. Kelty, N. Brown, M. Reed, The history of photodetection and photodynamic therapy, Photochem. Photobiol., 2001, 74, 656–669.
R. J. Porra, Recent progress in phorphyrin and chlorophyll biosynthesis, Photochem. Photobiol., 1997, 65, 492–516.
M. Wainwright, Photodynamic antimicrobial chemotherapy (PACT), J. Antimicrob. Chemother., 1998, 42, 13–28.
G. Bertoloni, F. M. Lauro, G. Cortrella, M. Merchat, Photosensitizing activity of hematoporphyrin on Staphylococcus aureus cells, Biochim. Biophys. Acta, 2000, 1475, 169–174.
K. T. de Oliveira, A. M. S. Silva, A. C. Tomé, M. G. P. M. S. Neves, C. R. Neri, V. S. Garcia, O. A. Serra, Y. Iamamoto, J. A. S. Cavaleiro, Synthesis of new amphiphilic chlorin derivatives from protoporphyrin IX dimethyl ester, Tetrahedron, 2008, 64, 8709–8715.
A. F. Uchoa, K. T. de Oliveira, M. S. Baptista, A. J. Bortoluzzi, Y. Iamamoto, O. A. Serra, Chlorin photosensitizer sterically designed to prevent self-aggregation, J. Org. Chem., 2011, 76, 8824–8832.
F. F. de Assis, J. M. de Souza, B. H. K. Assis, T. J. Brocksom, K. T. de Oliveira, Synthesis and photophysical studies of a chlorin sterically designed to prevent self-aggregation, Dyes Pigm., 2013, 98, 153–159.
E. S. Nyman, P. H. Hynninen, Research advances in the use of tetrapyrrolic photosensitizes for photodynamic therapy, J. Photochem. Photobiol., B, 2004, 73, 1–28.
A. D. Djalil, N. A. Nurulita, L. W. Limantara, S. Ibrahim, D. H. Tjahjono, Biological evaluations of protoporphyrin IX, pheophorbide a and its 1-hidroxyethyl derivatives for applications in photodynamic therapy, Int. J. Pharm. Pharm. Sci., 2012, 4, 741–746.
N. S. Soukos, L. A. Ximenez-Fyvie, M. R. Hamblin, S. S. Socransky, T. Hasan, Targeted Antimicrobial Photochemotherapy, Antimicrob. Agents Chemother., 1998, 42, 10 2595.
S. Douillard, D. Olivier, T. Patrice, In vitro and in vivo evaluation of Radachlorin sensitizer for photodynamic therapy, Photochem. Photobiol. Sci., 2009, 8, 405–413.
A. P. J. Maestrin, C. R. Neri, K. T. de Oliveira, O. A. Serra, Y. Iamamoto, Extração e purificação de clorofila a, da alga Spirulina maxima: um experimento para os cursos de química, Quim. Nova, 2009, 32, 1670–1672.
R. W. Redmond, J. N. Gamlin, A compilation of singlete oxygen yields from biologically relevant molecules, Photochem. Photobiol., 1999, 70, 391–475.
M. G. Lagorio, L. E. Dicello, E. S. Roman, Quantum yield of singlet molecular oxygen sensitization by copper(II) tetracarboxyphthalocyanine, J. Photochem. Photobiol., B, 1989, 3, 615–624.
M. Goksel, M. Durmus, D. Atilla, A comparative study on photophysical and photochemical properties of zinc phthalocyanines with different molecular symmetries, J. Porphyrins Phthalocyanines, 2012, 16, 895–906.
P. F. C. Menezes, C. A. S. Melo, V. S. Bagnato, H. Imasato, J. R. Perussi, Dark cytotoxicity of the photoproducts of the photosensitezer photogen after photobleacheaching induced by a laser, Laser Phys., 2005, 15, 435–442.
V. Engelhardt, B. Krammer, K. Plaetzer, Antibacterial photodynamic therapy using water-soluble formulation of hypericin or m-THPC is effective in inactivation of Staphylococcus aureus, Photochem. Photobiol. Sci., 2010, 9, 365–369.
P. S. Thakuri, R. Joshi, S. Basnet, S. S. Pandey, S. D. Taujale, N. Mishra, Antibacterial photodynamic therapy on Staphylococcus aureus and pseudomonas aeruginosa in vitro, Nepal Med. Coll. J., 2011, 13, 281–284.
M. Grinholc, B. Szramka, K. Olender, A. Graczyk, Bactericidal effect of photodynamic therapy against methicillin-resistant Staphylococcus aureus strain with the use of various porphyrin photosensitizers, Acta Biochim. Pol., 2007, 54, 665–670.
Z. Malik, H. Ladan, Y. Nitzan, Photodynamic inactivation of Gram-negative bacteria: problems and possible solutions, J. Photochem. Photobiol., B, 1992, 14, 262–266.
G. P. Tegos, M. Anbe, C. Yang, T. N. Demidova, M. Satti, P. Mroz, S. Janjua, F. Gad, M. R. Hamblin, Protease-stable polycationic photosensitizer conjugates between polyethileneimine and chlorin (e6) for broad-spectrum antimicrobial photoinactivation, Antimicrob. Agents Chemother., 2006, 50, 1402–1410.
Z. Malik, J. Hanania, Y. Nitzan, New trends in photobiology bactericidal effects of photoactivated porphyrins–an alternative approach to antimicrobial drugs, J. Photochem. Photobiol., B, 1990, 5, 281–293.
Y. Nitzan, M. Gutterman, Z. Malik, B. Ehrenberg, Inactivation of gram-negative bacteria by photosensitized porphyrins, Photochem. Photobiol., 1992, 55, 89–96.
J. Park, Y. Moon, I. Bang, Y. Kim, S. Kim, S. Ahn, J. Yoon, Antimicrobial effect of photodynamic therapy using a highly pure chlorin e6, Laser Med. Sci., 2010, 25, 705–710.
A. Preuß, L. Zeugner, S. Hackbarth, M. A. F. Faustino, M. G. P. M. S. Neves, J. A. S. Cavaleiro, B. Roeder, Photoinactivation of Escherichia coli (SURE2) without intracellular uptake of the photosensitizer, J. Appl. Microbiol., 2004, 114, 36–43.
G. Jori, S. B. Brown, Photosensitized inactivation of microorganisms, Photochem. Photobiol. Sci., 2004, 3, 403–405.
A. Minnock, D. I. Vernon, J. Schofield, J. Griffiths, J. H. Parish, S. B. Brown, Photoinactivation of bacteria. Use of a cationic water-soluble zinc phthalocyanine to photoinactivate both Gram-negative and Gram-positive bacteria, J. Photochem. Photobiol., B, 1996, 32, 159–164.
M. R. Hamblin, T. Hasan, Photodynamic therapy: a new antimicrobial approach to infectious disease?, Photochem. Photobiol. Sci., 2004, 3, 436–450.
T. Maisch, R. Szeimies, G. Jori, G. Abels, Antibacterial photodynamic therapy in dermatology, Photochem. Photobiol. Sci., 2004, 3, 907–917.
L. N. Dovigo, A. C. Pavarina, D. G. Ribeiro, C. S. Adriano, V. S. Bagnato, Photodynamic inactivation of four Candida species induced by photogem®, Braz. J. Microbiol., 2010, 41, 42–49.
Z. E. Hughes, A. E. Mark, R. L. Mancera, Molecular Dynamics Simulations of the Interactions of DMSO with DPPC and DOPC Phospholipid Membranes, J. Phys. Chem. B, 2012, 116, 11911–11923.
A. C. Williams, B. W. Barry, Penetration enhancers, Adv. Drug Delivery Rev., 2004, 56, 603–618.
F. He, W. Liu, S. Zheng, L. Zhou, B. Ye, Z. Qi, Ion transport through dimethyl sulfoxide (DMSO) induced transient water pores in cell membranes, Mol. Membr. Biol., 2012, 29, 107–113.
M. C. Gomes, S. Silva, M. A. F. Faustino, M. G. P. M. S. Neves, A. Almeida, J. A. S. Cavaleiro, J. P. C. Tomé, A. Cunha, Cationic galactoporphyrin photosensitisers against UV-B resistant bacteria: oxidation of lipids and proteins by 1O2, Photochem. Photobiol. Sci., 2013, 12, 262–271.
E. Alves, L. Costa, C. M. B. Carvalho, J. P. C. Tomé, M. A. Faustino, M. G. P. M. S. Neves, A. C. Tomé, J. A. S. Cavaleiro, Â. Cunha, A. Almeida, Charge effect on the photoinactivation of Gram-negative and Gram-positive bacteria by cationic meso-substituted porphyrins, BMC Microbiol., 2009, 9, 70 1–13.
C. M. B. Carvalho, E. Alves, L. Costa, J. P. C. Tome, M. A. F. Faustino, M. G. P. M. S. Neves, A. C. Tomé, J. A. S. Cavaleiro, A. Almeida, A. Cunha, Z. Lin, J. Rocha, Functional Cationic Nanomagnet_Porphyrin Hybrids for the Photoinactivation of Microorganisms, ACS Nano, 2010, 4, 7133–7140.
J. A. Hargus, F. R. Fronczek, M. G. H. Vicente, K. M. Smith, Mono(L)-aspatylchlorin-e6, Photochem. Photobiol., 2007, 83, 1006–1015.
N. R. S. Gobo, T. J. Brocksom, J. Zukerman-Schpector, K. T. de Oliveira, Synthesis of an octa-tert-buthylphthalocyanine: a low-aggregation and photochemically stable photosensitizer, Eur. J. Org. Chem., 2013, 5028–5031.
F. A. B. dos Santos, A. F. Uchoa, M. S. Baptista, Y. Iamamoto, O. A. Serra, T. J. Brocksom, K. T. de Oliveira, Synthesis of functionalized chlorins sterically-prevented from self-aggregation, Dyes Pigm., 2013, 99, 402–411.
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary information (ESI) available. See DOI: 10.1039/c3pp50376c
Rights and permissions
About this article
Cite this article
Uliana, M.P., Pires, L., Pratavieira, S. et al. Photobiological characteristics of chlorophyll a derivatives as microbial PDT agents. Photochem Photobiol Sci 13, 1137–1145 (2014). https://doi.org/10.1039/c3pp50376c
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c3pp50376c