-
PDF
- Split View
-
Views
-
Cite
Cite
Thomas S. Harrison, George E. Griffin, Stuart M. Levitz, Conditional Lethality of the Diprotic Weak Bases Chloroquine and Quinacrine against Cryptococcus neoformans, The Journal of Infectious Diseases, Volume 182, Issue 1, July 2000, Pages 283–289, https://doi.org/10.1086/315649
Close - Share Icon Share
Abstract
Chloroquine at 10 μM enhances the activity of macrophages against Cryptococcus neoformans but does not directly inhibit cryptococcal growth. The antifungal activity of higher chloroquine concentrations likely to be found within the acidic cryptococcal phagosome was tested. Concentrations of ⩽30 μM inhibited cryptococcal growth, and there was fungal killing at concentrations of ⩽100 μM. Activity was dependent on physiologic temperature and pH. Quinacrine was 50-fold more active than chloroquine, and concentrations as low as 100 nM enhanced macrophage anticryptococcal activity. Quinacrine was concentrated within a vacuolar system within the fungal cell and highly concentrated within intracellular C. neoformans. Ammonium chloride and bafilomycin A both inhibited cryptococcal growth, suggesting that the activity of chloroquine and quinacrine may in part be due to disruption of pH-dependent processes. These findings add to the known spectrum of activity of chloroquine and quinacrine. These, and related compounds, may have utility for the treatment of cryptococcosis.
Over recent years, the incidence of systemic fungal infections has increased, primarily because of the increased number of immunocompromised patients [1, 2]. For example, the encapsulated yeast Cryptococcus neoformans has emerged as a major cause of life-threatening meningoencephalitis in immunocompromised patients, especially those with human immunodeficiency virus (HIV) infection [3]. Unfortunately, at present, only a limited number of antifungal agents are available to treat these infections, and their efficacy is variable. Furthermore, resistance to some of these agents is now emerging [4]. Clearly, new antifungal drugs are urgently required.
A major hurdle to antifungal drug development is that mammalian and fungal cells are eukaryotic and thus have similar mechanisms for protein, RNA, and DNA synthesis, which reduces the potential for specific targets. However, protozoa are also eukaryotes, and we hypothesized that drugs active against protozoa could also have clinically significant antifungal activity. Two such antiprotozoal drugs are the 9-aminoacridine quinacrine and the 4-aminoquinoline chloroquine [5]. Quinacrine has activity against a number of protozoa, including Plasmodium species, Giardia lamblia, and Leishmania species. Chloroquine was developed in the late 1930s from quinacrine, which it replaced in clinical use for the treatment of malaria. Both drugs are concentrated within the acidic food vacuole of the malarial parasite, where they are thought to inhibit hematin polymerization. Very high concentrations of antimalarials have been found to inhibit the growth of the yeast Saccharomyces cerevisiae [6–8], and quinacrine has been used as a marker for the S. cerevisiae vacuole [9]. However, to our knowledge, no direct antifungal activity has previously been reported for these or other related antimalarial compounds against any of the pathogenic fungi at pharmacologically relevant concentrations.
We have previously shown that chloroquine, at a concentration of 10 μ M, markedly enhances the antimicrobial activity of human mononuclear phagocytes against C. neoformans [10]. At this concentration, chloroquine did not directly inhibit cryptococcal growth. However, because chloroquine is known to be concentrated by ion trapping within acidified compartments of eukaryotic cells [11], we tested whether chloroquine had any direct activity, in the absence of effector cells, at the higher concentrations likely to be found within the acidic cryptococcal phagosome [12]. Here, we investigated the antifungal activity of chloroquine and quinacrine against C. neoformans and explored the mechanisms of this activity.
Materials and Methods
Materials
All reagents were obtained from Sigma (St. Louis) except as noted. Cell culture medium was RPMI 1640 supplemented with L-glutamine, penicillin, streptomycin, and, where indicated, 5% pooled human serum (PHS). PHS was obtained by combining serum from ⩽ 10 healthy volunteers under ice-cold conditions and stored at −70°C to preserve complement activity. Chloroquine and quinacrine, dissolved in RPMI, and bafilomycin A, dissolved in dimethyl sulfoxide, were stored in aliquots at −70°C until required. Unless otherwise noted, assays were done at 37°C in humidified air supplemented with 5% CO2.
C. neoformans. Serotype A strain 145 of C. neoformans was a gift of T. Mitchell (Duke University, Durham, NC). Strains 201, 371 (serotype A), 444, 4506 (serotype B), and 18 (serotype C) were gifts of J. Kwon-Chung (National Institute of Allergy and Infectious Diseases, Bethesda, MD). Strain TM was a recent clinical isolate from an HIV-infected patient with cryptococcal meningitis. Fungi were harvested after 4 days of growth on Sabouraud dextrose agar at 30°C, counted in a hemocytometer, and resuspended at the desired concentration.
Monocyte-derived macrophages (MDMs)
Buffy coat cells from a single blood donor were obtained from the UK blood transfusion service. After dextran sedimentation to further deplete red cells, monocytes were purified by counterflow centrifugation [13] by means of an elutriation system (JE-5.0; Beckman Instruments, Fullerton, CA). For each donor, cytospin slides were prepared and examined by light microscopy after Giemsa staining to determine the percentages of monocytes, neutrophils, and lymphocytes (77% ± 5%, 16% ± 3%, and 8% ± 3%, respectively [mean ± SE]). Monocytes (7 × 104/well) in culture medium were then added to the wells of half-area 96-well plates (Costar; Corning, Corning, NY) and incubated for 90 min, after which the wells were washed to remove nonadherent cells, and adherent monocytes were incubated for a further 7 days in 100 μL of medium supplemented with 5% PHS to obtain MDMs. MDMs were washed again before assay.
Assay of antifungal activity
Antifungal activity of compounds in the absence or presence of macrophages was determined as previously described [10]. In assays in the absence of macrophages, C. neoformans (1 × 104) was incubated in wells of half-area 96-well plates in 100 μL of culture medium containing the indicated concentrations of drug. In experiments in the presence of macrophages, C. neoformans (1 × 104) was added to the wells of half-area 96-well plates containing 7 × 104 MDMs in 100 μL of culture medium containing the indicated concentrations of drug supplemented with 5% PHS. We found 5% serum to be sufficient to promote efficient binding and phagocytosis of C. neoformans by MDMs. We have previously shown that chloroquine does not affect the binding and internalization of C. neoformans [10]. Plates were incubated for 18 h, after which the number of colony-forming units of C. neoformans was determined by dilution and spread plates on Sabouraud dextrose agar. For each assay, sets of wells containing medium and C. neoformans were incubated at 4°C to determine the inoculum of live organisms. Replication of C. neoformans does not occur after incubation at 4°C for 18 h. In experiments with macrophages, 0.1% Triton X-100 was added to the 4°C control wells and to the experimental wells before plating to lyse the cells. Results are expressed as percentage of growth and were calculated as follows: [(experimental cfu/inoculated cfu) −1] × 100. Thus, a value of zero indicates no net fungal growth. Values of 100% and 300% indicate an average of 1 and 2 replications, respectively, and a negative value indicates that the number of colony-forming units decreased during the incubation period and therefore some fungal killing had occurred.
Accumulation of quinacrine within C. neoformans
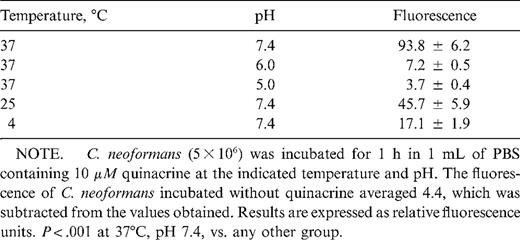
This was visualized by confocal microscopy and quantitated spectrofiuorimetrically. C. neoformans was incubated with 10 μM quinacrine in PBS for 15 min, washed once in PBS, and examined immediately on a Zeiss LSM 510 laser confocal scanning microscope (Carl Zeiss Inc., Thornwood, NY) equipped with a ×63 oil objective lens. For accumulation of quinacrine within phagocytosed C. neoformans, macrophages adherent to a glass slide were incubated with C. neoformans in PBS containing 10% PHS and 1 μM quinacrine. After 1 h, slides were washed in PBS and immediately examined on the confocal microscope. In quantitative studies, 5 × 106C. neoformans was incubated for 1 h in 1 mL of PBS containing 10 μM quinacrine at the indicated temperature and pH. The fungi were washed 3 times and resuspended in PBS, and fluorescence was determined by use of a spectrofluorimeter (F-4500; Hitachi Scientific Instruments, Mountain View, CA), with the excitation and emission wavelengths set at 450 and 520 nm, respectively.
Statistics
Means and SEs of sample groups were compared by use of the 2-tailed Student's t test.
Results
Direct antifungal activity of chloroquine against C. neoformans
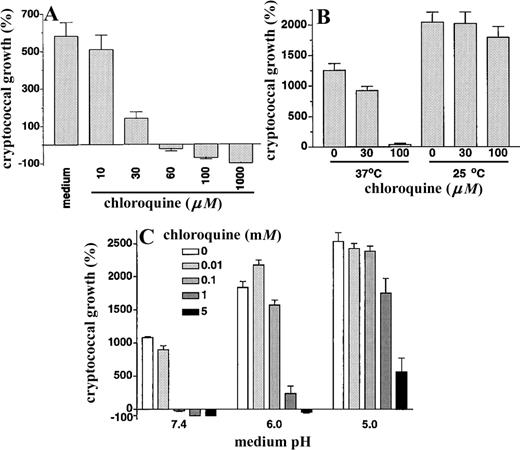
In initial experiments, we tested whether chloroquine had direct activity against C. neoformans by using the colonyforming unit, growth inhibition, and killing assay developed in previous studies [10]. We found that chloroquine did have potent antifungal activity (figure 1A). Chloroquine at 30 μM resulted in significant inhibition of cryptococcal growth, and at ⩽100 μM there was fungal killing, as demonstrated by an overall reduction in colony-forming units compared with the inoculum. Reductions in colony-forming units were also seen when the duration of the assay was reduced to 2 h, confirming the rapid fungicidal activity of chloroquine at >100 μM (data not shown). The above assays were done at physiologic temperature and pH. We next tested whether varying these conditions would affect the anticryptococcal activity of chloroquine. As expected, C. neoformans divided more rapidly at 25°C than at 37°C [3]. At 25°C, no inhibition of cryptococcal growth was seen with concentrations of chloroquine up to 100 μM (figure 1B). In addition to temperature, the efficacy of chloroquine was also dependent on the pH of the medium, with a progressive diminution in antifungal activity as the pH was reduced from 7.4 to 5.0 (figure 1C). For example, at pH 7.4, 100 μM chloroquine was fungicidal, whereas at pH 5 this concentration had no effect on fungal growth (figure 1C). Consistent with our previous results, under these assay conditions C. neoformans replicated more rapidly at lower pH [10].
A, Direct anticryptococcal activity of chloroquine. Cryptococcus neoformans strain 145 was incubated for 18 h at 37°C in humidified air with 5% CO2 in the presence of increasing concentrations of chloroquine, after which colony-forming units were determined by dilution and spread plates on Sabouraud dextrose agar. Data are mean ± SE for 3 separate experiments, each done in triplicate. For concentrations of ⩽30 μM, P < .001 in presence vs. absence of chloroquine. B, Activity of chloroquine is temperature dependent. C. neoformans strain 145 was incubated for 18 h at either 37°C or 25°C in humidified air with 5% CO2 in the presence of increasing concentrations of chloroquine, after which colony-forming units were determined as above. Data are mean ± SE for 4 separate experiments, each done in triplicate. At 37°C, P = .01 and P < .001 in presence of 30 μM and 100 μM chloroquine, respectively, vs. absence of chloroquine. C, Activity of chloroquine is dependent on external pH. pH of medium containing increasing concentrations of chloroquine was adjusted by addition of hydrochloric acid and then filter-sterilized. C. neoformans was added and incubated for 18 h at 37°C in humidified air with 5% CO2, after which colony-forming units were determined as above. Data are mean ± SE for 2 separate experiments, each done in triplicate. At pH 7.4, P < .001 in the presence of 0.1, 1, or 5 mM chloroquine vs. absence of chloroquine. At pH 6, P < .001 in the presence of 1 or 5 mM chloroquine vs. absence of chloroquine. At pH 5, P = .01 and P < .001 in the presence of 1 and 5 mM chloroquine, respectively, vs. absence of chloroquine.
Anticryptococcal activity of quinacrine
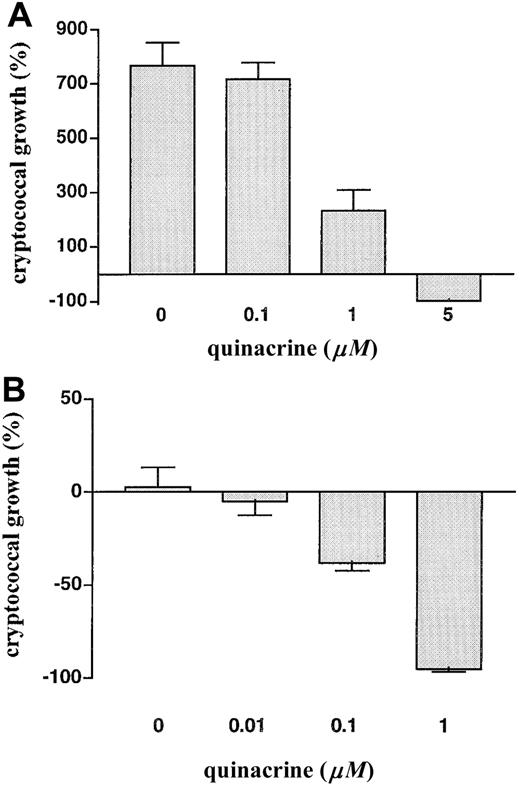
Next we tested the anticryptococcal activity of the related 9-aminoacridine quinacrine. Compared with chloroquine, quinacrine has a broader spectrum of activity against eukaryotic pathogens. In addition, the fluorescence of quinacrine allows study of its distribution within C. neoformans cells. Interestingly, we found quinacrine to be between 10- and 100-fold more active than chloroquine on a molar basis. Quinacrine at 1 μM caused marked inhibition of growth, and 5 μM was fungicidal (figure 2A). Like chloroquine activity, quinacrine activity was maximal at a temperature of 37°C and an external pH of 7.4 (data not shown). Furthermore, when tested in the presence of effector cells, concentrations of quinacrine as low as 100 nM significantly enhanced the anticryptococcal activity of human MDM (figure 2B).
A, Anticryptococcal activity of quinacrine. Cryptococcus neoformans was incubated for 18 h at 37°C in humidified air with 5% CO2 in the presence of increasing concentrations of quinacrine, after which colony-forming units were determined by dilution and spread plates. Data are mean ± SE for 2 separate experiments, each done in triplicate. P < .001 in the presence of 1 or 5 μM quinacrine vs. absence of quinacrine. B, Quinacrine enhances anticryptococcal activity of macrophages at nanomolar concentrations. Human monocyte-derived macrophages were incubated for 18 h with C. neoformans in the presence of increasing concentrations of quinacrine, after which macrophages were lysed and cryptococcal colony-forming units were determined. Data are mean ± SE for 4 separate experiments, each done in triplicate. P = .002 in the presence of 100 nM quinacrine vs. absence of quinacrine.
Accumulation of quinacrine within C. neoformans
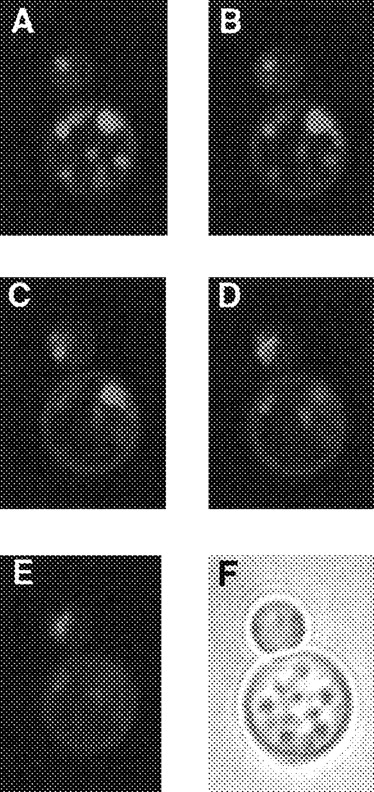
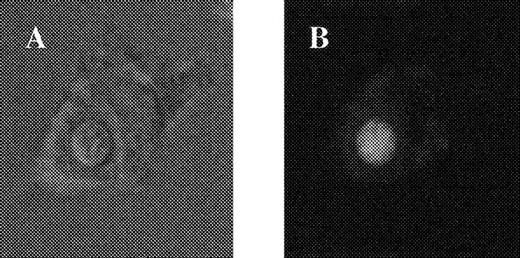
We hypothesized that the dependence of the activity of the diprotic lipophilic weak bases chloroquine and quinacrine on external pH could be explained if this activity depended on the accumulation of the drugs by ion trapping within acidified compartments of the fungal cell, as occurs in other eukaryotic cells [11]. At a low external pH, no pH gradient would exist to drive such accumulation. C. neoformans was incubated for 15 min at 37°C in PBS, pH 7.4, containing 10 μM quinacrine, washed in PBS, and examined immediately by confocal microscopy. Figure 3 shows a series of cuts through a representative cryptococcal cell and demonstrates accumulation of fluorescent quinacrine within what appears to be a complex vacuolar system. Because chloroquine and quinacrine would be expected to be concentrated within the acidic cryptococcal phagolysosome, we also examined quinacrine accumulation within phagocytosed C. neoformans. Macrophages were infected with C. neoformans and exposed to 1 μM quinacrine for 1 h. Figure 4 shows brightfield and fluorescence views of a macrophage containing a single cryptococcal cell and confirms selective concentration of quinacrine within the intracellular C. neoformans with relatively little fluorescence within other macrophage compartments.
Accumulation of quinacrine within discrete compartments within cryptococcal cell. Cryptococcus neoformans was incubated with 10 μM quinacrine in PBS for 15 min, washed in PBS, and examined immediately on a laser confocal scanning microscope equipped with ×63 oil objective lens. A-E, Fluorescence images through consecutive levels of representative budding cryptococcal cell; F, brightfield view of same cell.
Accumulation of quinacrine within phagocytosed Cryptococcus neoformans. Macrophages adherent to glass slide were incubated with C. neoformans in PBS containing 10% pooled human serum and 1 μM quinacrine. After 1 h, slides were washed in PBS and immediately examined on laser confocal scanning microscope. A, Brightfield image of macrophage containing single internalized C. neoformans; B, fluorescence view of same cell.
Next, the effect of external pH and temperature on quinacrine uptake by C. neoformans was determined spectrofluorimetrically (table 1). Uptake of quinacrine by C. neoformans was maximal under the physiologic conditions of pH 7.4 and 37°C temperature. Decreasing the extracellular pH to 6.0 or 5.0 resulted in a >90% decrease in fungal accumulation of quinacrine. Lowering the incubation temperature to 25°C and 4°C resulted in 51% and 82% reductions, respectively, in quinacrine concentrations.
Effect of temperature and pH on quinacrine uptake by Cryptococcus neoformans.
Growth inhibition of C. neoformans by other lysosomotropic agents and bafilomycin A
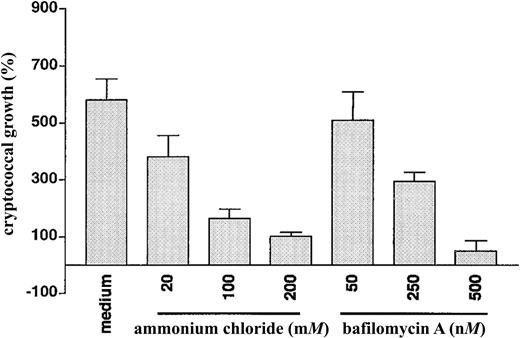
Accumulation of chloroquine and quinacrine within acidified compartments within the cryptococcal cell would be expected to raise the pH of such compartments and disrupt pH-dependent processes. To test whether raising the pH of such compartments affects cryptococcal growth, we tested the activity of ammonium chloride, a structurally unrelated weak base, and of bafilomycin A, an inhibitor of eukaryotic vacuolar adenosinetriphosphatase (vATPase) [14]. Both compounds were found to cause a dose-dependent inhibition of cryptococcal growth (figure 5).
Inhibition of cryptococcal growth by ammonium chloride and bafilomycin A. Cryptococcus neoformans was incubated for 18 h in the presence of medium alone or increasing concentrations of ammonium chloride or bafilomycin A, after which colony-forming units were determined by dilution and spread plates. Bafilomycin A was dissolved in dimethyl sulfoxide (DMSO). Final concentration of DMSO (⩾1%) had no effect on cryptococcal growth (for 3 triplicate experiments, mean ± SE cryptococcal growth in 1% DMSO was 547% ± 54%). Data are mean ± SE for 3 separate experiments, each done in triplicate. P < .001 in the presence of 100 or 200 mM ammonium chloride vs. medium alone. P = .002 and P < .001 in the presence of 250 and 500 nM bafilomycin A, respectively, vs. medium alone.
Activity of chloroquine and quinacrine against diverse cryptococcal strains
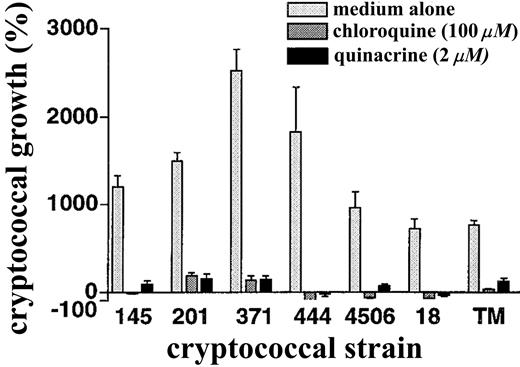
Finally, to test the general nature of our results, we tested the activity of chloroquine and quinacrine against a number of cryptococcal strains. Laboratory strains representing different cryptococcal serotypes and a recent clinical isolate from an HIV-infected patient with cryptococcal meningitis all showed similar susceptibility to chloroquine (at 100 μM) and quinacrine (at 2 μM;figure 6).
Susceptibility of diverse cryptococcal strains to chloroquine and quinacrine. Indicated Cryptococcus neoformans strains were incubated in the presence of medium alone, 100 μM chloroquine, or 2 μM quinacrine, after which colony-forming units were determined by dilution and spread plates. Incubations were for 18 h except for the more slowly replicating strain 18, which was incubated for 30 h. Data are mean ± SE for 2 separate experiments, each done in duplicate. For all strains, P ⩾ .01 in the presence of chloroquine or quinacrine vs. medium alone.
Discussion
These findings add significantly to the known spectrum of activity of chloroquine and quinacrine and suggest that these drugs should be further investigated as possible adjunctive agents in the treatment of cryptococcosis. Furthermore, over more than 60 years, in the course of research into more effective antimalarials, hundreds of related compounds have been synthesized that, to our knowledge, have not been appropriately tested for antifungal activity. The results presented here raise the possibility that this large group of compounds may contain some other agents with potentially useful antifungal activity.
In contrast to our findings, 2 other groups have reported no direct activity of chloroquine against C. neoformans. However, Mazzolla et al. [15] tested chloroquine concentrations of ⩾20 μM only [15], and Wang and Casadevall [16], although testing up to 5 mM, used a minimal medium that has a low pH and a temperature of 30°C [16]. Interestingly, we found the antifungal effects of chloroquine and quinacrine to be maximal at pH 7.4 and a temperature of 37°C. Accumulation of quinacrine within C. neoformans also was maximal at that pH and temperature, suggesting that the antifungal activity is directly dependent on drug uptake. The diminished uptake of quinacrine at low pH is consistent with its weak base properties. At low extracellular pH, there will not be a pH gradient driving it into acidified intracellular compartments [5]. Although there was some reduction in quinacrine uptake at 25°C compared with 37°C, the dependence of antifungal activity on temperature suggests the possibility of interactions of chloroquine and quinacrine with recently elucidated signaling pathways involved in survival of C. neoformans at physiologic temperature [17]. Experiments are ongoing to examine whether chloroquine and quinacrine have additive or synergistic effects with known inhibitors of these pathways, such as cyclosporine and FK 506 [17].
The direct antifungal activity of chloroquine and quinacrine may be due to inhibition of processes, such as protein processing, secretion, and degradation, that require acidified compartments within the cryptococcal cell. Support for this concept comes from work with S. cerevisiae. Mutations in the vATPase, responsible for the generation of proton gradients across the membranes of the vacuolar system, result in yeast cells that fail to grow in medium with a pH >6.5 [18]. Moreover, S. cerevisiae strains with defective vacuolar acidification fail to sort and process vacuolar proteins normally [19]. The confocal studies confirm the existence of a complex vacuolar system in C. neoformans [20]. By use of the pH-sensitive fluorescent probe carboxy-2′,7′-dichlorofluorescein diacetate, which accumulates in yeast vacuoles, we have found the average pH of this compartment to be 5.7 (authors' unpublished data). Our results suggest that inhibition of the acidification of these intracellular compartments per se can limit cryptococcal growth. The structurally unrelated weak base ammonium chloride and the vATPase inhibitor bafilomycin A both inhibited cryptococcal growth in a concentration-dependent manner. However, neither was fungicidal, so that neutralization of the acid pH of compartments within the cryptococcal cell may be only partly responsible for the activity of chloroquine and quinacrine.
In addition to such pH-dependent effects, there may be specific fungal targets to which chloroquine and quinacrine bind and whose function is thereby affected. Of note, both are diprotic weak bases that might be expected to have a similar effect on the pH of acidic compartments within the cryptococcal cell. Despite this, the potencies of the 2 drugs against C. neoformans are significantly different, suggesting that there may be one or more specific fungal targets that are preferentially affected by quinacrine. In this regard, chloroquine and quinacrine have been shown to have a wide range of effects on eukaryotic cells, including DNA binding and inhibition of phospholipases, electron transport, and calmodulin-mediated signaling [21–26], some of which may be relevant to their anticryptococcal activity. In S. cerevisiae, high concentrations of chloroquine have been shown to inhibit protein phosphorylation and some glycolytic enzymes, in particular phosphoglycerate kinase and pyruvate decarboxylase [27], whereas resistance in S. cerevisiae to antimalarials has been associated with overexpression of several genes whose function has yet to be determined [8]. Further experiments are under way to look for specific cryptococcal targets that are affected by chloroquine and quinacrine.
As we demonstrated previously for chloroquine [10], the anticryptococcal activity of quinacrine was markedly enhanced in the presence of human MDM. Chloroquine enhances the activity of MDM against Legionella pneumophila and Histoplasma capsulatum by a mechanism dependent on iron deprivation [28, 29]. However, distinct antimicrobial mechanisms appear to be operative, because the effects of chloroquine on C. neoformans are independent of iron deprivation [10]. Rather, our data suggest that chloroquine and quinacrine are concentrated within the MDM phagolysosome, resulting in attainment of fungicidal concentrations of these drugs. In addition to this direct antifungal effect, chloroquine and quinacrine could have indirect effects as a result of raising the pH of cryptococcal phagolysosomes [12]. Over a range of pH from 5 to 8, the growth of C. neoformans in cell-free medium was found to be progressively reduced as the pH of the medium was raised [10], suggesting that some of the effect of chloroquine may be related to the neutralization of phagosome pH [12].
Very high concentrations of hydroxychloroquine (0.6 nmol/106 cells or 1.3 mM, assuming an average cell volume of 450 fL [30]), have been measured in peripheral blood mononuclear cells of rheumatoid patients treated with hydroxychloroquine at a dose of 400 mg/day [31]. These levels were similar to those measured after in vitro exposure of peripheral blood mononuclear cells to concentrations of 10–100 μM hydroxychloroquine [31]. Chloroquine and quinacrine are further concentrated within the cryptococcal phagolysosome (figure 4) [12], which normally has a pH ∼5.0 [12]. Thus, concentrations well above those needed to kill C. neoformans should be realized in patients. However, in human cases of cryptococcosis, C. neoformans is found both as a facultative intracellular parasite of macrophages and as an extracellular pathogen [32]. Chloroquine and quinacrine would be expected to have greatest activity against the intracellular fungi. Nevertheless, we and others have demonstrated in vivo benefits of chloroquine in murine models of cryptococcosis [10, 15]. In addition, of interest, there is a report of a patient with simultaneous malaria and cryptococcosis whose cryptococcal disease improved, with disappearance of the organisms from the cerebrospinal fluid, when the malaria was treated with quinacrine [33].
For a number of additional reasons, chloroquine and quinacrine deserve further investigation as adjunctive agents in the treatment of cryptococcosis. Both drugs are generally safe, established oral agents with a long half-life, allowing for infrequent administration. Chloroquine and quinacrine are both protein-bound (50%–60% and ∼85%, respectively). For chloroquine this is unlikely to have important pharmacokinetic effects, although for quinacrine it is possible that this could reduce penetration of drug into the cerebrospinal fluid [34]. Of note, both are concentrated within brain tissue [35–38], although to a lesser extent than in some other organs. Not least, chloroquine and quinacrine are cheap and available in the developing world, where the burden of cryptococcosis is greatest and where treatment with established agents often is not available. In a wider context, the results suggest that the aminoquinolines, aminoacridines, and related compounds should be screened by appropriate in vitro systems for activity against C. neoformans and the other important pathogenic fungi.
Acknowledgment
We thank Ryan Hastey for help with the confocal microscopy.
References
Presented in part: 4th International Conference on Cryptococcus and Cryptococcosis, London, September 1999; Royal Society of Tropical Medicine and Hygiene, Research in Progress, London, November 1999.
Informed consent was obtained from all blood donors. US Department of Health and Human Services and Boston University Medical Center guidelines for human experimentation were followed.
Financial support: NIH (AI-37532, AI-25780, TW-00940); Wellcome Trust (Advanced Training Fellowship to T.S.H.); Burroughs Wellcome Fund Scholar Award in Pathogenic Mycology (to S.M.L.).