-
PDF
- Split View
-
Views
-
Cite
Cite
GEERT DOM, BIEKE DE WILDE, WOUTER HULSTIJN, WIM VAN DEN BRINK, BERNARD SABBE, BEHAVIOURAL ASPECTS OF IMPULSIVITY IN ALCOHOLICS WITH AND WITHOUT A CLUSTER-B PERSONALITY DISORDER, Alcohol and Alcoholism, Volume 41, Issue 4, July/August 2006, Pages 412–420, https://doi.org/10.1093/alcalc/agl030
Close - Share Icon Share
Abstract
Aims: Studies have shown that alcoholics with a cluster-B personality disorder (cluster-B PD) are characterized by high levels of impulsivity. However, impulsivity has mainly been studied as a broad concept without its different aspects being considered. The present study compared abstinent alcoholic inpatients without any personality disorder (PD) and abstinent alcoholics with cluster-B PD on different aspects of impulsivity, i.e. self-reported impulsivity and neuropsychological indicators such as behavioural control and delay of gratification. Methods: Forty alcohol-dependent inpatients without PD and 22 alcohol-dependent inpatients with a cluster-B PD were compared on two self-report impulsivity questionnaires (Barratt impulsiveness scale; sensation-seeking scales) and three behavioural impulsivity tasks [Go/No-Go task; delay discounting task (DDT); Stroop colour word test]. Tests were administered after stable abstinence of at least 3 weeks. Results: Self-report measures of impulsivity were higher in cluster-B alcoholics than in alcoholics without PD. Behavioural tasks revealed a more differentiated pattern of impairments. On the Go/No-Go task, cluster-B alcoholics were impaired in inhibitory control but not in reaction time compared with alcoholics without PD. In contrast, no significant differences on the DDT and the Stroop were observed. Conclusion: Alcohol-dependent patients with and without a cluster-B PD differ in terms of behavioural inhibition but not in terms of activation or the ability to delay gratification. This finding may partly account for their impulsive and (self-) destructive behaviours. Treatment planning should pay specific attention to these impairments in behavioural control.
(Received 2 September 2005; first review notified 14 October 2005; in revised form 23 February 2006; accepted 13 March 2006)
INTRODUCTION
Research on substance use disorders (SUDs) has recently taken great interest in the role of personality in the pathogenesis of addictive disorders. One of the personality traits that have consistently been linked to substance abuse is impulsivity. Dawe et al. (2004) proposed that impulsivity was critically involved in both the initiation and end-stage of addiction. Similarly, Koob and Le Moal (2001) have suggested that addiction is to be regarded as the progression from an impulse-control disorder (i.e. high reward drive) to a compulsive disorder in its end-stage (i.e. relapse after abstinence and loss of control).
Impulsivity is now widely viewed as a multidimensional construct consisting of different, related dimensions and corresponding neuroanatomical pathways (Winstanley et al., 2004). Specifically the anterior cingulate cortex (ACC) and the orbitofrontal cortex have been reported to be critically involved in impulsive behaviours (Rolls, 2004). It is noteworthy that abnormalities in these brain regions have recently been proposed to underlie both addictive processes and cluster-B disorders, i.e. borderline and antisocial personality disorders (PDs) (Lubman et al., 2004; Seguin, 2004; Berlin et al., 2005; Dom et al., 2005; Kalivas and Volkow, 2005).
Factor analyses of the various self-report instruments used to measure impulsivity support a two-factor model to underlie the trait (Dawe et al., 2004). In addition, animal and human behavioural models of impulsivity discriminate at least two dimensions (Winstanley et al., 2004). Dickman (1990) distinguished functional and dysfunctional impulsivity. Dawe and Loxton (2004) argue for the two facets reward drive and rash impulsiveness. De Wit and Richards (2004) identify two behavioural processes. The first refers to the degree to which immediate (rewarding) consequences have more control over an individual's behaviour than consequences that are delayed, as measured in a delay discounting task (DDT). The second is behavioural inhibition and denotes the ability of an individual to appropriately inhibit thoughts or actions. Reflecting on the relationship between personality and substance abuse, Verheul (Verheul et al., 1999; Verheul, 2001) argues for a three-pathway model with behavioural disinhibition, stress reactivity and reward sensitivity, where the behavioural-disinhibition pathway predicts that individuals scoring high on traits such as antisocial behaviour and impulsiveness have lower thresholds for deviant behaviours such as alcohol and drug abuse (Verheul, 2001). Substance misuse like stimulant abuse in individuals with an antisocial or borderline PD is likely to develop along this pathway. It may be hypothesized that specifically rash impulsiveness (or behavioural disinhibition) may be involved in this association. The stress-reduction pathway to addiction predicts that individuals scoring high on traits like stress reactivity typically respond to stress with anxiety and mood instability, which in turn become a motive for substance use as self-medication. Avoidant, dependent and schizotypical PDs have been associated with this second pathway. Finally, the reward-sensitivity pathway predicts that individuals scoring high on traits such as novelty seeking and reward seeking will be motivated to substance use for its positive reinforcing properties, e.g. people with a histrionic, antisocial or narcissistic PD. Specifically reward drive (cf. delay discounting) may be involved in this developmental pathway.
Of the three proposed pathways, the disinhibition pathway has been documented best. A high co-morbidity has indeed been observed between SUDs and Axis-II disorders from the impulse-control spectrum, i.e. cluster-B PDs such as the antisocial and borderline syndromes (e.g. Bowden-Jones et al., 2004). Furthermore, several longitudinal studies have shown that childhood personality traits of behavioural disinhibition, antisocial behaviour and low harm avoidance were associated with an early engagement in substance use and subsequent early development of SUDs in adolescence and young adulthood (Cloninger et al., 1988; Masse and Tremblay, 1997; Tarter et al., 2003; Kirisci et al., 2004). Taken together, evidence is accumulating that the behavioural-disinhibition pathway may play an important role in subgroups of drug-abusing populations and it is hypothesized that impulsivity, especially its disinhibition dimension (or rash impulsiveness), may be of particular relevance.
Various instruments have been developed to assess impulsive behaviours. These can roughly be divided into subjective self-report measures of personality that rely on an individual's self-perception of his or her behaviour, and objective behavioural tasks that evaluate the subject's performance patterns in relation to specific dimensions of impulsivity. Research on impulsivity has generally relied on self-reports or on measurements or observations whose respective power to gauge impulsivity is open to interpretation: the relationship between how individuals behave and how they report they behave is an ongoing topic of debate in personality research. The correlation between the two techniques has been reported to be only weak to moderate (Reynolds et al., 2006).
A limited number of studies have explored impulsive personality traits within different alcoholic subpopulations. Adult early-onset alcoholics have been found to have higher levels of self-reported impulsivity than late-onset alcoholics (Lykouras et al., 2004; Dom et al., 2006a). It is suggested that in subtypes of alcoholics impulsivity-related personality traits are not directly associated with alcohol abuse but rather with elevated levels of (antisocial) psychopathology (Whiteside and Lynam, 2003). This would support Verheul's disinhibition pathway, although none of the studies using self-report measures of impulsivity to examine alcoholic populations actually explored the association with co-morbid personality pathology.
Different operant behavioural tasks can be employed to measure the two main dimensions of impulsivity, i.e. reward drive and behavioural disinhibition (de Wit and Richards, 2004). The delay discounting paradigm has been studied extensively in both animals and humans. It assesses the subject's preference for a more immediate and more certain reward relative to a delayed or uncertain reward (Mazur, 1987; Richards et al., 1999). Numerous studies have demonstrated higher discount rates in non-clinical illicit-drug users (Kollins, 2003) and in subjects dependent on heroin (Madden et al., 1997, 1999; Kirby et al., 1999), cocaine (Coffey et al., 2003; Kirby and Petry, 2004) and nicotine (Bickel et al., 1999; Reynolds et al., 2004), all relative to the rates found for healthy controls. However, studies investigating alcohol-dependent populations are scarce and results are conflicting. Petry (2001) found higher discount rates in actively using alcoholics than in alcoholics who were abstinent at the time of testing and non-addicted controls. Kirby and Petry (2004) could not find differences in discounting between alcoholics and controls and although Bjork et al. (2004) did report higher discount rates in alcoholics relative to controls, they did not find any differences between Type-I and Type-II alcoholics. In contrast, we demonstrated that early-onset alcoholics had higher discount rates than late-onset alcoholics (Dom et al., 2006b). Possibly, differences in co-morbid personality pathology may account for these inconsistent findings.
Go/No-Go paradigms are widely used to test the behavioural-disinhibition dimension of impulsivity. Since successful performance requires prepotent behaviours to be inhibited, the tasks provide a measure of behavioural control. In drug addicts impairments in behavioural control have frequently been reported (e.g. Hester and Garavan, 2004; Moeller et al., 2004, 2005; Lee et al., 2005). A neuroimaging study showed ACC hypo-activity in cocaine users during such a behavioural suppression task (Kaufman et al., 2003). In their recent fMRI study Forman et al. (2004) revealed attenuation of the error signal in the ACC of opiate addicts and proposed that this might play a role in the loss of control observed in addictions and other forms of impulsive behaviours.
With respect to alcohol-use disorders, behavioural-inhibition paradigms have been mainly restricted to the study of acute effects of alcohol priming in non-dependent, social drinkers (e.g. Fillmore and Weafer, 2004). Studies examining (stable abstinent) alcohol-dependent populations are limited. Björk et al. (2004) found Type-II alcoholics to make more errors on a behavioural-suppression task than Type-I alcoholics but they failed to correct for co-morbid (antisocial) PDs, which are known to be highly prevalent in Type-II alcoholics.
In conclusion, research relating personality traits to impulsivity in particular mainly concerned drug-abusing populations and the limited number of studies that did investigate alcohol-dependent subjects did not examine the two aspects simultaneously. To amend for this lack, in the current study we compared abstinent alcohol-dependent patients with and without a co-morbid cluster-B PD. We tested the disinhibition pathway as proposed by Verheul (2001) and its relation with impulsivity. We hypothesized that for Verheul's pathway to be valid the alcoholics with cluster-B PD would need to score higher on self-report measures of impulsivity and exhibit more impairments on tasks measuring behavioural disinhibition than alcoholics without co-morbid PDs.
METHODS
Sample
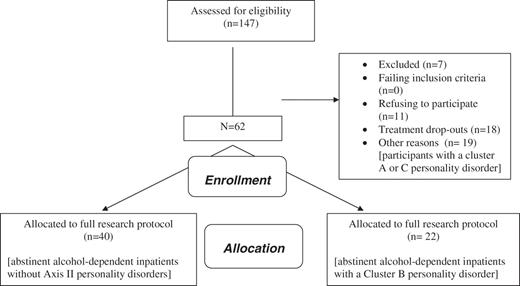
Participants were DSM-IV alcohol-dependent adults recruited from an inpatient treatment facility (see also Dom et al., 2006a,b). During a period of 18 months (2003–2004) every second patient admitted to the treatment unit was asked to participate in the study (n = 147). Exclusion criteria were current or lifetime history of psychotic disorders (n = 0), amnesic disorders (n = 2), neurological disorders such as trauma capitis or epilepsy (n = 2), and severe somatic disorders such as liver cirrhosis, AIDS, thyroid disorders or visual problems that could interfere with concentration during the interviews or with task execution (n = 3). Ten patients refused participation while another 30 patients continued their treatment on an outpatient basis after detoxification. Eligible patients (n = 100) were those patients who engaged in the clinic's long-term inpatient treatment after having been fully detoxified (and whose abstinence could be monitored daily). Of this sample, 18 patients prematurely ceased treatment and in the course of the study 1 patient refused further participation. A trained interviewer (MA in psychology) evaluated the remaining 81 participants for Axis-II PDs during a structured interview [SCID-II; DSM-IV; American Psychiatric Association (APA) 1994]. Forty patients had no co-morbid PD (49.5%), 22 were diagnosed with a cluster-B PD (27%), and 19 (23.5%) with a cluster-A and/or cluster-C PD. The participants with a cluster-A or C PD were subsequently excluded. Ultimately, the study sample comprised 62 patients: 40 alcoholics without co-morbidity and 22 with a cluster-B PD. In the cluster-B group 15 patients met criteria for borderline, 3 for antisocial, 2 for both borderline and antisocial, 0 for histrionic and 2 for narcissistic PD (Fig. 1).
Diagnoses of alcohol dependence and co-morbid illicit-drug abuse or dependence based on DSM-IV criteria (APA, 1994) were made during a clinical psychiatric interview, supplemented with the data from the European version of the addiction severity index (EuropASI), and, if possible, collateral information (spouse, family). The patient's family history of alcoholism was determined using the information obtained by a clinical interview using the family-tree method (Mann et al., 1985) and completed with the data from the EuropASI interview, and, if possible, collateral information.
The alcoholics without PD and those with a cluster-B PD did not significantly differ in terms of sex (71% male), age (mean 42.1 years; SD 9.5), years of education (mean 13.0; SD 2.5), years of alcohol dependence (mean 15.8; SD 8.9), days of abstinence before testing (mean 34.0; SD 15.3), IQ (mean Full IQ 96.3; SD 12.5), and paternal or maternal family history of alcoholism (positive history in 40 and 13% of the participants, respectively). In contrast, the cluster-B participants' age-at-onset of alcohol dependence was significantly lower (23 years; SD 9) than that of the alcoholics without PD (28 years; SD 9) [t(60) = −2.095; P = 0.040]. Also, more cluster-B alcoholics had a history of co-morbid illicit-drug abuse or dependence and nicotine dependence (see Table 1).
Differences between the alcoholics without PDs and those with a cluster-B PD on the EuropASI severity scores, the BDHI direct-aggression subscale (BDHI_DA), the BDI, the percentages of cigarette-dependent alcoholics (Smokers), alcoholics with co-morbid illicit-drug abuse or dependence (Drug) and alcoholics undergoing pharmacotherapy on the day of behavioural testing
| EuropASI severity . | Alcoholics without any PD . | Alcoholics with Cluster-B . | t(P) . |
|---|---|---|---|
| Medical | 1.85 (1.53) | 2.45 (1.65) | 1.448 (ns) |
| Work | 2.45 (1.93) | 3.81 (1.81) | 2.657 (P < 0.05) |
| Alcohol | 5.97 (1.78) | 5.82 (1.89) | −0.321 (ns*) |
| Drug | 1.85 (2.30) | 3.41 (2.77) | 2.365 (P < 0.05) |
| Legal | 1.23 (1.46) | 2.41 (2.15) | 2.543 (P < 0.05) |
| Social | 3.10 (1.21) | 3.86 (1.86) | 1.937 (ns) |
| Psychiatric | 3.92 (2.00) | 5.14 (1.64) | 2.416 (P < 0.05) |
| BDHI-DA | 6.34 (3.82) | 10.24 (3.56) | −3.838 (P < 0.001) |
| BDI | 13.24 (6.60) | 18.44 (10.84) | −2.003 (0.052) |
| % Smokers | 72 | 91 | χ2(1) = 3.064 (ns) |
| % Drug | 30 | 59 | χ2(1) = 4.992 (P<0.05) |
| % Pharmacotherapy | 56 | 62 | χ2(1) = 0.170 (ns) |
| EuropASI severity . | Alcoholics without any PD . | Alcoholics with Cluster-B . | t(P) . |
|---|---|---|---|
| Medical | 1.85 (1.53) | 2.45 (1.65) | 1.448 (ns) |
| Work | 2.45 (1.93) | 3.81 (1.81) | 2.657 (P < 0.05) |
| Alcohol | 5.97 (1.78) | 5.82 (1.89) | −0.321 (ns*) |
| Drug | 1.85 (2.30) | 3.41 (2.77) | 2.365 (P < 0.05) |
| Legal | 1.23 (1.46) | 2.41 (2.15) | 2.543 (P < 0.05) |
| Social | 3.10 (1.21) | 3.86 (1.86) | 1.937 (ns) |
| Psychiatric | 3.92 (2.00) | 5.14 (1.64) | 2.416 (P < 0.05) |
| BDHI-DA | 6.34 (3.82) | 10.24 (3.56) | −3.838 (P < 0.001) |
| BDI | 13.24 (6.60) | 18.44 (10.84) | −2.003 (0.052) |
| % Smokers | 72 | 91 | χ2(1) = 3.064 (ns) |
| % Drug | 30 | 59 | χ2(1) = 4.992 (P<0.05) |
| % Pharmacotherapy | 56 | 62 | χ2(1) = 0.170 (ns) |
*ns = non-significant (α = 0.05).
Differences between the alcoholics without PDs and those with a cluster-B PD on the EuropASI severity scores, the BDHI direct-aggression subscale (BDHI_DA), the BDI, the percentages of cigarette-dependent alcoholics (Smokers), alcoholics with co-morbid illicit-drug abuse or dependence (Drug) and alcoholics undergoing pharmacotherapy on the day of behavioural testing
| EuropASI severity . | Alcoholics without any PD . | Alcoholics with Cluster-B . | t(P) . |
|---|---|---|---|
| Medical | 1.85 (1.53) | 2.45 (1.65) | 1.448 (ns) |
| Work | 2.45 (1.93) | 3.81 (1.81) | 2.657 (P < 0.05) |
| Alcohol | 5.97 (1.78) | 5.82 (1.89) | −0.321 (ns*) |
| Drug | 1.85 (2.30) | 3.41 (2.77) | 2.365 (P < 0.05) |
| Legal | 1.23 (1.46) | 2.41 (2.15) | 2.543 (P < 0.05) |
| Social | 3.10 (1.21) | 3.86 (1.86) | 1.937 (ns) |
| Psychiatric | 3.92 (2.00) | 5.14 (1.64) | 2.416 (P < 0.05) |
| BDHI-DA | 6.34 (3.82) | 10.24 (3.56) | −3.838 (P < 0.001) |
| BDI | 13.24 (6.60) | 18.44 (10.84) | −2.003 (0.052) |
| % Smokers | 72 | 91 | χ2(1) = 3.064 (ns) |
| % Drug | 30 | 59 | χ2(1) = 4.992 (P<0.05) |
| % Pharmacotherapy | 56 | 62 | χ2(1) = 0.170 (ns) |
| EuropASI severity . | Alcoholics without any PD . | Alcoholics with Cluster-B . | t(P) . |
|---|---|---|---|
| Medical | 1.85 (1.53) | 2.45 (1.65) | 1.448 (ns) |
| Work | 2.45 (1.93) | 3.81 (1.81) | 2.657 (P < 0.05) |
| Alcohol | 5.97 (1.78) | 5.82 (1.89) | −0.321 (ns*) |
| Drug | 1.85 (2.30) | 3.41 (2.77) | 2.365 (P < 0.05) |
| Legal | 1.23 (1.46) | 2.41 (2.15) | 2.543 (P < 0.05) |
| Social | 3.10 (1.21) | 3.86 (1.86) | 1.937 (ns) |
| Psychiatric | 3.92 (2.00) | 5.14 (1.64) | 2.416 (P < 0.05) |
| BDHI-DA | 6.34 (3.82) | 10.24 (3.56) | −3.838 (P < 0.001) |
| BDI | 13.24 (6.60) | 18.44 (10.84) | −2.003 (0.052) |
| % Smokers | 72 | 91 | χ2(1) = 3.064 (ns) |
| % Drug | 30 | 59 | χ2(1) = 4.992 (P<0.05) |
| % Pharmacotherapy | 56 | 62 | χ2(1) = 0.170 (ns) |
*ns = non-significant (α = 0.05).
The patients in our clinical sample received usual care, i.e. they participated in the centre's regular treatment programme comprising both psychosocial and pharmacological interventions. Of the total sample, 58% received pharmacological treatment including antidepressants (50%), anti-psychotics (14%) and mood-stabilizing anticonvulsants (5%). None of the participants were taking anti-alcohol drugs (e.g. disulfiram or acamprosate) at the time of testing (if indicated, these drugs were started at the end of their inpatient treatment).
All patients gave their written and informed consent. The study was approved by the medical ethics committee of the Brothers of Charity psychiatric hospitals, Belgium.
Assessment instruments
In order to prevent contamination of the current levels of psychopathology with symptoms of chronic intoxication or withdrawal, patients were only interviewed and tested after full detoxification, i.e. after at least 3 weeks of controlled abstinence.
Instruments for sample description
European addiction severity index
The Belgian adaptation of the Dutch version of the EuropASI (Raes, 1996) is widely used in addiction research and has been tested for reliability and validity (Kokkevi and Hartgers, 1995). It provides problem-severity scores in seven domains (medical, work, alcohol, illicit-drug, legal, psychiatric and social) for the preceding 30-day period, with higher scores reflecting greater problems.
Structured clinical interview for DSM-IV Axis-II PDs
The SCID-II (First et al., 1997) is a semi-structured diagnostic interview developed to measure the 10 DSM-IV (APA, 1994) Axis-II PDs. We used the Dutch version (Weertman et al., 1999).
Wechsler adult intelligence scale
The Wechsler adult intelligence scale (WAIS-III) (Dutch version; 2000) consists of an individually administered battery of tests designed to measure verbal and performance intelligence. An overall measure of intelligence, combining the two scores, is indicated as full-scale IQ.
The Beck depression inventory.
For the Dutch version of the Beck depression inventory (BDI) that was administered in this study (Beck et al., 1961; Bouman et al., 1985) sufficient reliability and validity have been established (Bosscher et al., 1986).
Buss–Durkee hostility inventory
The Buss–Durkee hostility inventory (BDHI) (Buss and Durkee, 1957) is a widely used true–false questionnaire. The Dutch version (BDHI-D) comprises 40 items and its psychometric properties have been well examined and show good reliability and validity (Lange et al., 1995a,b). We used its direct-aggression subscale to assess aggression.
Instruments assessing aspects of impulsivity
Self-report questionnaires
Barratt impulsiveness scale: The Dutch adaptation of the Barratt impulsiveness scale (BIS-11) (Patton et al., 1995) is a 30-item self-report questionnaire with a total score (BIS_T) and three subscales assessing different aspects of impulsiveness: non-planning (BIS_NP), motor (BIS_M) and cognitive impulsiveness (BIS_C).
Sensation-seeking scale: The Dutch adaptation sensation-seeking scale (SSS-D) is based on earlier versions of Zuckerman's Sensation-Seeking Scale (Zuckerman et al., 1964). It was tested in a Dutch population and showed good reliability and validity. Norm scores for different age groups are available (Feij et al., 1997). The 67-item, self-report questionnaire provides a general score (SSS_G) as well as scores on four subscales assessing disinhibition (SSS_DIS), thrill and adventure seeking (SSS_TAS), experience seeking (SSS_ES) and boredom susceptibility (SSS_BS).
Behavioural tasks
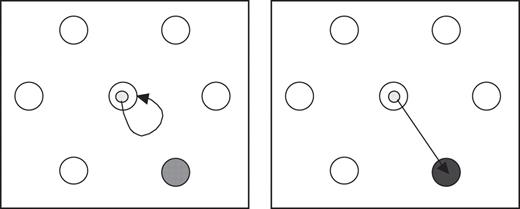
Go/No-Go task: A classical Go/No-Go paradigm was presented in a computerized design. The materials and software used are described in De Jong et al. (1996). Participants were seated in a quiet room at a table on which a computer screen and writing tablet were mounted. They were asked to move a screen cursor (yellow dot), controlled by displacements executed with an electronic pen, as fast and as accurately as possible into a newly presented target circle if this was coloured dark blue (Go trial) and to withhold a reaction if the target was light blue (No-Go trial; see Fig. 2). Participants were presented with four series each comprising 56 trials (ratio Go/No-Go was 1/8).
The Go/No-Go task with the left-hand panel illustrating a No-Go trial and the right-hand panel a Go trial
The dependent variables were the number of errors (Error) in the No-Go trials and the reaction (RT) and movement times (MT) in the Go trials. An error on a No-Go signal was defined as any movement away from the start circle. RT was defined as the time between the appearance of a new target circle and response initiation, and MT as the interval from response initiation until the target circle was reached.
Stroop colour word test: The Stroop colour word test (Stroop, 1935) is a behavioural task traditionally employed to measure attentional bias. We opted for this test as it is especially useful at assessing whether stimuli are differentially attended to by participants. It determines to what extent the lexical information processing of the stimulus word overrules the perception of the colour in which the word is printed. The interference effect (defined as the time to read card series 3 minus the time to read card series 2) is hypothesized to be a behavioural measure of response inhibition (Lezak, 1995). The Stroop colour word test has frequently been used to investigate behavioural inhibition within drug- and alcohol-dependent populations (see e.g. Tedstone and Coyle, 2004; Fishbein et al., 2005).
Delay discounting task: Delay discounting provides an index of the relative value of immediate vs delayed rewards (Mazur, 1987). A classical DDT design, using hypothetical reward money, was presented on a computer screen (see also Dom et al., 2006b). The test consisted of ∼110 questions (trials), an example of which reads: ‘Which do you prefer: €10, 30 days from now, or €2 now?’ The questions are presented according to an adjusting-amount procedure (Richards et al., 1999), in which the amount of immediate certain money is adjusted across successive questions until an amount is reached that the participant judges to be equivalent to the delayed €10 reward. This amount thus provides a quantitative measure of the subjective value of the delayed rewards. The points of subjective equality are called indifference points, which were determined for three amounts, i.e. 10, 30 and €100, and for five different delays: 2, 30, 180, 365 and 730 days, respectively. On delay trials, participants could choose between varying amounts of instant money and between 10, 30 and €100 becoming available after a delay. The indifference points obtained at each of the delays were plotted and discount functions were derived through curve-fitting analysis (Matlab, version 5.3). Previous research has shown that an individual's discount curves are best described by a hyperbolic discount function (Mazur, 1987): V = A/(1 + kD).
This yields a parameter k, where V is the present value of the delayed reward A at delay D in days. As k increases, the person discounts the future reward more steeply. Therefore, k can be regarded as an impulsiveness parameter, with higher values corresponding to higher levels of impulsiveness (for details, see Richards et al., 1999).
Statistical analysis
For the comparison of group mean scores Student t-tests for independent groups were used, whereas χ2 tests were used for dichotomous variables.
If not specified otherwise, general linear model (GLM) procedures were used. Group was a fixed factor with two levels: alcoholics with cluster-B PD and alcoholics without PD. A GLM repeated measures analysis was used to analyse the DDT results. Because the discount rates had a positively skewed distribution statistical analyses were performed on the natural logarithmic transformation of these values: ln(k + 0.001). The DDT variable ‘amount’ had three levels (€10, €30 and €100; see also Dom et al., 2006b).
To control for potential confounding we ran an additional analysis on the behavioural data to adjust for factors that might have influenced motor performance, i.e. the current use of psychiatric medication (Y/N)[fixed factor], age and BDI depression scores [covariates].
Pearson correlations (with Bonferoni correction for multiple comparisons) were used to identify correlations between the self-report and behavioural measures of impulsivity.
All analyses were performed using SPSS-11 statistical software.
RESULTS
Patient characteristics
Baseline patient characteristics are presented in Table 1. Relative to the alcoholics without PD the alcoholics with a cluster-B PD had significantly higher EuropASI problem-severity scores on the domains work, illicit-drug, legal, social and psychiatric as well as significantly higher depression (BDI) and direct-aggression (BDHI-DA) scores and a higher incidence of nicotine dependence and lifetime history of illicit-drug abuse or dependence.
Measures of impulsivity
Self-report measures
The results of the self-report questionnaires are presented in Table 2. Alcoholics with a cluster-B PD had significantly higher scores on all BIS-11 subscales than those without PD and they also scored significantly higher on the SSS_ES. The two groups did not significantly differ in their scores on the other SSS subscales.
Differences between the alcoholics without PDs and those with a cluster-B PD on the subscales of the two self-report questionnaires, i.e. the BIS (BIS_C = cognitive, BIS_M = motor, BIS_NP = non-planning, BIS_T = total score) and the SSS (SSS_TAS = thrill and adventure seeking, SSS_ES = experience seeking, SSS_BS = boredom susceptibility, SSS_DIS = disinhibition, SSS_G = general sensation seeking)
. | Alcoholics without any PD (n = 40) . | Alcoholics with cluster-B (n = 22) . | F(P) . |
|---|---|---|---|
| BIS_C | 14.72 (5.78) | 18.10 (6.18) | 4.440 (P < 0.05) |
| BIS_M | 14.87 (6.34) | 18.86 (8.47) | 4.244 (P < 0.05) |
| BIS_NP | 20.23 (7.82) | 24.71 (8.75) | 4.128 (P < 0.05) |
| BIS_T | 49.82 (16.07) | 61.67 (17.09) | 7.095 (P < 0.01) |
| SSS_TAS | 6.43 (2.64) | 6.05 (3.19) | 0.245 (ns*) |
| SSS_ES | 6.62 (2.61) | 8.24 (2.32) | 4.669 (P < 0.05) |
| SSS_BS | 5.44 (2.99) | 6.62 (2.48) | 2.337 (ns) |
| SSS_DIS | 7.49 (2.25) | 8.48 (1.72) | 2.599 (ns) |
| SSS_G | 6.72 (2.50) | 7.95 (2.33) | 2.599 (0.113) |
. | Alcoholics without any PD (n = 40) . | Alcoholics with cluster-B (n = 22) . | F(P) . |
|---|---|---|---|
| BIS_C | 14.72 (5.78) | 18.10 (6.18) | 4.440 (P < 0.05) |
| BIS_M | 14.87 (6.34) | 18.86 (8.47) | 4.244 (P < 0.05) |
| BIS_NP | 20.23 (7.82) | 24.71 (8.75) | 4.128 (P < 0.05) |
| BIS_T | 49.82 (16.07) | 61.67 (17.09) | 7.095 (P < 0.01) |
| SSS_TAS | 6.43 (2.64) | 6.05 (3.19) | 0.245 (ns*) |
| SSS_ES | 6.62 (2.61) | 8.24 (2.32) | 4.669 (P < 0.05) |
| SSS_BS | 5.44 (2.99) | 6.62 (2.48) | 2.337 (ns) |
| SSS_DIS | 7.49 (2.25) | 8.48 (1.72) | 2.599 (ns) |
| SSS_G | 6.72 (2.50) | 7.95 (2.33) | 2.599 (0.113) |
*ns = non-significant (α = 0.05).
Differences between the alcoholics without PDs and those with a cluster-B PD on the subscales of the two self-report questionnaires, i.e. the BIS (BIS_C = cognitive, BIS_M = motor, BIS_NP = non-planning, BIS_T = total score) and the SSS (SSS_TAS = thrill and adventure seeking, SSS_ES = experience seeking, SSS_BS = boredom susceptibility, SSS_DIS = disinhibition, SSS_G = general sensation seeking)
. | Alcoholics without any PD (n = 40) . | Alcoholics with cluster-B (n = 22) . | F(P) . |
|---|---|---|---|
| BIS_C | 14.72 (5.78) | 18.10 (6.18) | 4.440 (P < 0.05) |
| BIS_M | 14.87 (6.34) | 18.86 (8.47) | 4.244 (P < 0.05) |
| BIS_NP | 20.23 (7.82) | 24.71 (8.75) | 4.128 (P < 0.05) |
| BIS_T | 49.82 (16.07) | 61.67 (17.09) | 7.095 (P < 0.01) |
| SSS_TAS | 6.43 (2.64) | 6.05 (3.19) | 0.245 (ns*) |
| SSS_ES | 6.62 (2.61) | 8.24 (2.32) | 4.669 (P < 0.05) |
| SSS_BS | 5.44 (2.99) | 6.62 (2.48) | 2.337 (ns) |
| SSS_DIS | 7.49 (2.25) | 8.48 (1.72) | 2.599 (ns) |
| SSS_G | 6.72 (2.50) | 7.95 (2.33) | 2.599 (0.113) |
. | Alcoholics without any PD (n = 40) . | Alcoholics with cluster-B (n = 22) . | F(P) . |
|---|---|---|---|
| BIS_C | 14.72 (5.78) | 18.10 (6.18) | 4.440 (P < 0.05) |
| BIS_M | 14.87 (6.34) | 18.86 (8.47) | 4.244 (P < 0.05) |
| BIS_NP | 20.23 (7.82) | 24.71 (8.75) | 4.128 (P < 0.05) |
| BIS_T | 49.82 (16.07) | 61.67 (17.09) | 7.095 (P < 0.01) |
| SSS_TAS | 6.43 (2.64) | 6.05 (3.19) | 0.245 (ns*) |
| SSS_ES | 6.62 (2.61) | 8.24 (2.32) | 4.669 (P < 0.05) |
| SSS_BS | 5.44 (2.99) | 6.62 (2.48) | 2.337 (ns) |
| SSS_DIS | 7.49 (2.25) | 8.48 (1.72) | 2.599 (ns) |
| SSS_G | 6.72 (2.50) | 7.95 (2.33) | 2.599 (0.113) |
*ns = non-significant (α = 0.05).
Behavioural tasks
The cluster-B alcoholics made more errors on the No-Go trials than the alcoholics without PD. On the Go trials, however, the two patient groups had similar RTs and MTs. The Stroop and the DDT did not reveal any significant group differences (see Table 3).
Differences between the alcoholics without PDs and those with a cluster-B PD on the selected behavioural tasks: Go/No-Go task, Stroop colour word task, and DDT
. | Alcoholics without any PD (n = 40) . | Alcoholics with cluster-B (n = 22) . | F(P) . |
|---|---|---|---|
| Number error No-Go | 8.3 (6.1) | 11.3 (5.7) | 4.062 (P < 0.05) |
| RT Go | 0.3267 (0.0447) | 0.3179 (0.0343) | 0.081 (ns*) |
| MT Go | 0.4977 (0.0667) | 0.5012 (0.0458) | 0.371 (ns) |
| IE | 36.74 (22.72) | 36.69 (12.38) | 0.554 (ns) |
| k10 | 0.0474 (0.1253) | 0.0395 (0.0898) | 0.285 (ns) |
| k30 | 0.0298 (0.0655) | 0.0306 (0.0561) | |
| k100 | 0.0298 (0.0719) | 0.0668 (0.1951) |
. | Alcoholics without any PD (n = 40) . | Alcoholics with cluster-B (n = 22) . | F(P) . |
|---|---|---|---|
| Number error No-Go | 8.3 (6.1) | 11.3 (5.7) | 4.062 (P < 0.05) |
| RT Go | 0.3267 (0.0447) | 0.3179 (0.0343) | 0.081 (ns*) |
| MT Go | 0.4977 (0.0667) | 0.5012 (0.0458) | 0.371 (ns) |
| IE | 36.74 (22.72) | 36.69 (12.38) | 0.554 (ns) |
| k10 | 0.0474 (0.1253) | 0.0395 (0.0898) | 0.285 (ns) |
| k30 | 0.0298 (0.0655) | 0.0306 (0.0561) | |
| k100 | 0.0298 (0.0719) | 0.0668 (0.1951) |
Reaction (RT) and movement times (MT) and interference effect (IE) are presented in seconds. The raw scores for the three discount rates (k10, k30 and k100) are presented (in the statistical analysis the natural log transformation Lnk was the repeated measure).
*ns = non-significant (α = 0.05).
Differences between the alcoholics without PDs and those with a cluster-B PD on the selected behavioural tasks: Go/No-Go task, Stroop colour word task, and DDT
. | Alcoholics without any PD (n = 40) . | Alcoholics with cluster-B (n = 22) . | F(P) . |
|---|---|---|---|
| Number error No-Go | 8.3 (6.1) | 11.3 (5.7) | 4.062 (P < 0.05) |
| RT Go | 0.3267 (0.0447) | 0.3179 (0.0343) | 0.081 (ns*) |
| MT Go | 0.4977 (0.0667) | 0.5012 (0.0458) | 0.371 (ns) |
| IE | 36.74 (22.72) | 36.69 (12.38) | 0.554 (ns) |
| k10 | 0.0474 (0.1253) | 0.0395 (0.0898) | 0.285 (ns) |
| k30 | 0.0298 (0.0655) | 0.0306 (0.0561) | |
| k100 | 0.0298 (0.0719) | 0.0668 (0.1951) |
. | Alcoholics without any PD (n = 40) . | Alcoholics with cluster-B (n = 22) . | F(P) . |
|---|---|---|---|
| Number error No-Go | 8.3 (6.1) | 11.3 (5.7) | 4.062 (P < 0.05) |
| RT Go | 0.3267 (0.0447) | 0.3179 (0.0343) | 0.081 (ns*) |
| MT Go | 0.4977 (0.0667) | 0.5012 (0.0458) | 0.371 (ns) |
| IE | 36.74 (22.72) | 36.69 (12.38) | 0.554 (ns) |
| k10 | 0.0474 (0.1253) | 0.0395 (0.0898) | 0.285 (ns) |
| k30 | 0.0298 (0.0655) | 0.0306 (0.0561) | |
| k100 | 0.0298 (0.0719) | 0.0668 (0.1951) |
Reaction (RT) and movement times (MT) and interference effect (IE) are presented in seconds. The raw scores for the three discount rates (k10, k30 and k100) are presented (in the statistical analysis the natural log transformation Lnk was the repeated measure).
*ns = non-significant (α = 0.05).
After current use of psychiatric medication (Y/N), age and depression scores (BDI) had been controlled for the group difference with respect to error remained significant (P = 0.024) and the between-subjects effects of the Go/No-Go task (RT and MT), Stroop and DDT remained non-significant.
Correlations
Results of the correlation (Pearson) analysis are presented in Table 4. Correlations were generally weak (ranging from 0.01 to 0.32) and after Bonferoni correction for multiple testing (P < 0.003 = 0.05/14) none of the correlations between the self-report measures and behavioural measures of impulsivity proved statistically significant.
Correlations [r (P)] between the number of errors (Errors) in the NoGo trials, the reaction (RT) and movement times (MT) on the Go trials, the natural log transformation of the discount rate for €100 (Lnk100) and the interference effect (IE) on the Stroop task on the one hand and the scores on the impulsivity subscales of the BIS [cognitive (BIS_C), motor (BIS_M), non-planning (BIS_NP) and total (BIS_T)], and the subscales thrill and adventure seeking (SSS_TAS), experience seeking (SSS_ES), boredom susceptibility (SSS_BS), disinhibition (SSS_DIS) and general sensation seeking (SSS_G) of the Sensation-Seeking Scale on the other
. | BIS_C . | BIS_M . | BIS_NP . | BIS_T . | SSS_TAS . | SSS_ES . | SSS_BS . | SSS_DIS . | SSS_G . |
|---|---|---|---|---|---|---|---|---|---|
| Error | 0.090 (ns*) | 0.065 (ns) | 0.105 (ns) | 0.111 (ns) | 0.013 (ns) | 0.054 (ns) | 0.024 (ns) | 0.047 (ns) | 0.057 (ns) |
| RT | 0.108 (ns) | 0.063 (ns) | 0.025 (ns) | 0.079 (ns) | −0.100 (ns) | 0.001 (ns) | −0.122 (ns) | −0.010 (ns) | −0.100 (ns) |
| MT | 0.276 (ns) | 0.246 (ns) | 0.076 (ns) | 0.244 (ns) | −0.144 (ns) | −0.106 (ns) | −0.120 (ns) | 0.086 (ns) | −0.111 (ns) |
| Lnk100 | 0.055 (ns) | 0.068 (ns) | −0.039 (ns) | 0.029 (ns) | −0.033 (ns) | 0.102 (ns) | 0.315 (ns) | 0.168 (ns) | 0.212 (ns) |
| IE | 0.053 (ns) | −0.155 (ns) | −0.085 (ns) | −0.090 (ns) | −0.248 (ns) | −0.198 (ns) | −0.155 (ns) | −0.215 (ns) | −0.247 (ns) |
. | BIS_C . | BIS_M . | BIS_NP . | BIS_T . | SSS_TAS . | SSS_ES . | SSS_BS . | SSS_DIS . | SSS_G . |
|---|---|---|---|---|---|---|---|---|---|
| Error | 0.090 (ns*) | 0.065 (ns) | 0.105 (ns) | 0.111 (ns) | 0.013 (ns) | 0.054 (ns) | 0.024 (ns) | 0.047 (ns) | 0.057 (ns) |
| RT | 0.108 (ns) | 0.063 (ns) | 0.025 (ns) | 0.079 (ns) | −0.100 (ns) | 0.001 (ns) | −0.122 (ns) | −0.010 (ns) | −0.100 (ns) |
| MT | 0.276 (ns) | 0.246 (ns) | 0.076 (ns) | 0.244 (ns) | −0.144 (ns) | −0.106 (ns) | −0.120 (ns) | 0.086 (ns) | −0.111 (ns) |
| Lnk100 | 0.055 (ns) | 0.068 (ns) | −0.039 (ns) | 0.029 (ns) | −0.033 (ns) | 0.102 (ns) | 0.315 (ns) | 0.168 (ns) | 0.212 (ns) |
| IE | 0.053 (ns) | −0.155 (ns) | −0.085 (ns) | −0.090 (ns) | −0.248 (ns) | −0.198 (ns) | −0.155 (ns) | −0.215 (ns) | −0.247 (ns) |
*Non-significant (Bonferoni corrected α = 0.003).
Correlations [r (P)] between the number of errors (Errors) in the NoGo trials, the reaction (RT) and movement times (MT) on the Go trials, the natural log transformation of the discount rate for €100 (Lnk100) and the interference effect (IE) on the Stroop task on the one hand and the scores on the impulsivity subscales of the BIS [cognitive (BIS_C), motor (BIS_M), non-planning (BIS_NP) and total (BIS_T)], and the subscales thrill and adventure seeking (SSS_TAS), experience seeking (SSS_ES), boredom susceptibility (SSS_BS), disinhibition (SSS_DIS) and general sensation seeking (SSS_G) of the Sensation-Seeking Scale on the other
. | BIS_C . | BIS_M . | BIS_NP . | BIS_T . | SSS_TAS . | SSS_ES . | SSS_BS . | SSS_DIS . | SSS_G . |
|---|---|---|---|---|---|---|---|---|---|
| Error | 0.090 (ns*) | 0.065 (ns) | 0.105 (ns) | 0.111 (ns) | 0.013 (ns) | 0.054 (ns) | 0.024 (ns) | 0.047 (ns) | 0.057 (ns) |
| RT | 0.108 (ns) | 0.063 (ns) | 0.025 (ns) | 0.079 (ns) | −0.100 (ns) | 0.001 (ns) | −0.122 (ns) | −0.010 (ns) | −0.100 (ns) |
| MT | 0.276 (ns) | 0.246 (ns) | 0.076 (ns) | 0.244 (ns) | −0.144 (ns) | −0.106 (ns) | −0.120 (ns) | 0.086 (ns) | −0.111 (ns) |
| Lnk100 | 0.055 (ns) | 0.068 (ns) | −0.039 (ns) | 0.029 (ns) | −0.033 (ns) | 0.102 (ns) | 0.315 (ns) | 0.168 (ns) | 0.212 (ns) |
| IE | 0.053 (ns) | −0.155 (ns) | −0.085 (ns) | −0.090 (ns) | −0.248 (ns) | −0.198 (ns) | −0.155 (ns) | −0.215 (ns) | −0.247 (ns) |
. | BIS_C . | BIS_M . | BIS_NP . | BIS_T . | SSS_TAS . | SSS_ES . | SSS_BS . | SSS_DIS . | SSS_G . |
|---|---|---|---|---|---|---|---|---|---|
| Error | 0.090 (ns*) | 0.065 (ns) | 0.105 (ns) | 0.111 (ns) | 0.013 (ns) | 0.054 (ns) | 0.024 (ns) | 0.047 (ns) | 0.057 (ns) |
| RT | 0.108 (ns) | 0.063 (ns) | 0.025 (ns) | 0.079 (ns) | −0.100 (ns) | 0.001 (ns) | −0.122 (ns) | −0.010 (ns) | −0.100 (ns) |
| MT | 0.276 (ns) | 0.246 (ns) | 0.076 (ns) | 0.244 (ns) | −0.144 (ns) | −0.106 (ns) | −0.120 (ns) | 0.086 (ns) | −0.111 (ns) |
| Lnk100 | 0.055 (ns) | 0.068 (ns) | −0.039 (ns) | 0.029 (ns) | −0.033 (ns) | 0.102 (ns) | 0.315 (ns) | 0.168 (ns) | 0.212 (ns) |
| IE | 0.053 (ns) | −0.155 (ns) | −0.085 (ns) | −0.090 (ns) | −0.248 (ns) | −0.198 (ns) | −0.155 (ns) | −0.215 (ns) | −0.247 (ns) |
*Non-significant (Bonferoni corrected α = 0.003).
DISCUSSION
The present study was the first to examine different aspects of impulsivity in abstinent alcohol-dependent patients with and without a cluster-B PD. The results showed that (i) compared with alcoholics without PDs, alcoholics with a cluster-B PD are characterized by a higher self-reported impulsivity and a weakened inhibitory control on the Go/No-Go task and (ii) the ability to delay gratification did not differ between the two groups.
Disinhibited behaviour may arise from either a weakened inhibitory-control system or a heightened activation system (Fillmore and Weafer, 2004). The Go/No-Go paradigm assesses behavioural control as the net effect of competing activating and inhibiting processes. It allows the source of impaired control to be identified by independently measuring the subject's ability to activate and to inhibit a response. Our Go/No-Go findings indicate that the cluster-B alcoholics primarily differed from the alcoholics without co-morbidity in terms of reduced inhibition rather than in terms of any over-activation or excessive drive.
In contrast with the disparities in behavioural control, the DDT did not reveal any differences between our two alcoholic subgroups. The DDT is thought to reflect the reward-sensitivity dimensions of impulsivity and as such may be hypothesized to be involved in Verheul's reward-sensitivity pathway and not the behavioural-disinhibition pathway. The delay discounting paradigm has been extensively studied in both clinical and non-clinical populations and showing elevated levels of discounting in nicotine-, alcohol- and illicit-drug-dependent individuals (e.g. Bickel et al., 1999; Kirby and Petry, 2004; Dom et al., 2006b). With the exception of studies into pathological gambling (Petry and Casarella, 1999), few studies explored this aspect of impulsivity in other psychiatric disorders characterized by high levels of impulsivity (like impulse-control disorders or cluster-B PDs). Our results suggest that discounting may be relatively unimpaired in cluster-B syndromes. However, since both our samples consisted of alcohol-dependent participants, i.e. a population that is by definition characterized by a low capacity to postpone gratification (ceiling effect), more research is needed that explores delay discounting in subjects with a cluster-B disorder without co-morbid substance abuse.
Salo et al. (2002) showed illicit-drug abusers to have increased interference, i.e. prolonged RTs, on a Stroop colour word task compared with controls. Others (like Swick and Jovanovic, 2002) propose longer RTs or higher error rates to reflect inefficient inhibition of task-irrelevant responses, response conflict and distractibility. The lack of differences in interference between our two alcoholic subgroups may be due to the Stroop's insensitivity to mild cognitive impairment (Bohnen et al., 1992). In the literature Stroop effects in alcohol-dependent patients tend to be conflicting. More consistent findings have been produced with an emotional or alcohol-specific Stroop task (i.e. including alcohol-related words), suggesting that specifically alcohol-related words are distracting to alcoholics (Bauer and Cox, 1998; Lusher et al., 2004).
That our cluster-B alcoholics had a higher subjective (BIS-11) impulsivity than the alcoholics without co-morbidity is in line with earlier studies reporting higher BIS scores for borderline patients relative to controls (e.g. Dougherty et al., 1999; Berlin et al., 2005). The BIS is a widely used validated self-report measure of impulsivity and Dawe et al. (2004) have proposed that it specifically taps rash impulsiveness.
Together, our findings are in line with the behavioural-disinhibition pathway as proposed by Verheul (2001) and indicate that specifically rash impulsiveness may play a prominent role in the co-morbidity of substance abuse and cluster-B PDs (Casillas and Clark, 2002).
In line with earlier studies (see e.g. Kirby and Petry, 2004; Reynolds et al., 2006), the correlations between the self-report and behavioural measures of impulsivity in our samples were weak (ranging from 0.01 to 0.3) and not statistically significant. This implies that the two types of tools reflect different aspects of the concept impulsivity. It is of interest in this respect that several recent studies revealed that the impairments on behavioural tasks and not the levels of self-reported impulsivity predicted relapse in addictive behaviours (Bowden-Jones et al., 2005; Marisssen et al., 2005; Goudriaan A.E., Oosterlaan J., de Beurs E., van den Brink W., manuscript submitted). These findings lend further support to the use of, not directly observable, behavioural (endophenotypic) characteristics in personality and addiction research and underscore the limitations of the use of directly observable or subjective clinical (phenotypic) traits (Dougerthy et al., 2005; Noël et al., 2005; Ooteman et al., 2005).
It is of clinical importance that the EuropASI problem severity of the alcoholics with cluster-B PD was higher in four of the seven substance-use-related domains than that of the alcoholics without co-morbidity. A similar relationship has been documented extensively in substance abusers with Axis-II co-morbidity. In addition, co-morbidity with Axis-II PDs and specifically with antisocial and borderline PD within drug-abusing or alcohol-dependent populations has been related to poor pre-treatment characteristics, treatment compliance and outcome (Verheul et al., 1998, 2000; Van Horn and Frank, 1998; Thomas et al., 1999; Ross et al., 2003; Haro et al., 2004; Wagner et al., 2004). Specifically high levels of aggression and impulsivity, key characteristics of our cluster-B alcoholics, were associated with poor treatment outcome (Moeller et al., 2001). Clearly, more targeted pharmacological and psychosocial interventions need to be developed that address impulsive and self-destructive behaviours and enhance treatment compliance (Ball, 2005). In alcoholics with a borderline personality, for instance, dialectical behaviour therapy (Linehan, 1987) has proven effective in reducing these behaviours (Linehan et al., 1999; van den Bosch et al., 2002; Verheul et al., 2003).
For a proper interpretation of our results, it needs to be taken into account that the samples in our study are only representative of alcohol-dependent subjects with a high problem severity on several life domains. Although one might argue that alcoholics that have chosen to undergo long-term inpatient therapy might represent a highly motivated group, most of the alcoholics in our sample remained in our clinic because of their problem severity and a lack of sufficient social or family support. Consequently, our sample may have been characterized by disproportionately high levels of co-morbidity and a disproportionately high number of participants with socially inappropriate behaviours.
We did not test a non-substance-abusing control group because the principal aim of the current study was to elucidate the differences between two clinically relevant subgroups of alcoholic inpatients. However, our findings merit corroboration in other populations and the different aspects of impulsivity warrant further scrutiny, both in healthy controls and in patients afflicted by other psychiatric disorders.
Finally, since this is essentially a clinical-descriptive study, it excludes any hypotheses about causes or consequences. Nevertheless, an interesting detail is that the alcoholics with a cluster-B disorder had an earlier age-of-onset than those without co-morbidity, which is in line with the findings Skodol et al. (1999) reported. This could either indicate that people who eventually develop PDs have problems in adolescence that tend to incite them to substance use at an early age or that an earlier use of substances leads to psychosocial problems that come to be diagnosed as PDs. Several longitudinal studies corroborated the first assumption by demonstrating that childhood conduct problems and disinhibitory behaviours were predictive of the development of both (cluster-B) PDs and substance-use-related disorders later in life (Bernstein et al., 1996; Masse and Tremblay, 1997; Tarter et al., 2004). More prospective, longitudinal studies are recommended to delineate the effects of different personality traits on the development of alcohol dependence and other psychiatric syndromes later in life.
CONCLUSIONS
Alcohol-dependent patients with and without a cluster-B PD differ in terms of behavioural inhibition and self-reported (rash) impulsiveness but not in terms of activation or their ability to delay gratification. Health professionals should take these differences into account when planning the psychosocial and pharmacological treatments of these two types of patients.
This study was funded by an internal grant of the Brothers of Charity and supported by the outpatient mental health care centre (cggz), VAGGA, Belgium.
REFERENCES
American Psychiatric Association (
Ball, S. A. (
Bauer, D. and Cox, W. M. (
Beck, A. T., Ward, C. H., Mendelson, M. et al. (
Berlin, H. A., Rolls, E. T. and Iversen, S. D. (
Bernstein, D. P., Cohen, P., Skodol, A. E. et al. (
Bickel, W. K., Odum, A. L. and Madden, G. J. (
Bjork, J. M., Hommer, D. W., Grant, S. J. et al. (
Bohnen, N., Jolles, J. and Twijnstra, A. (
Bosscher, R. J., Koning, H. and Van Meurs, R. (
Bouman, T. K., Luteijn, F., Albersnagel, F. A. et al. (
Bowden-Jones, O., Iqbal, M., Tyrer, P. et al. (
Bowden-Jones, H., McPhillips, M., Rogers, R. et al. (
Buss, A. H. and Durkee, A. (
Casillas, A. and Clark, L. A. (
Cloninger, C. R., Sigvardsson, S. and Bohman, M. (
Coffey, S. F., Gudleski, G. D., Saladin, M. E. et al. (
Dawe, S. and Loxton, N. J. (
Dawe, S., Matthew, J. G. and Loxton, N. J. (
De Jong, W. P., Hulstijn, W., Kosterman, B. J. M. et al. (
de Wit, H. and Richards, J. B. (
Dickman, S. J. (
Dom, G., Sabbe, B., Hulstijn, W. et al. (
Dom, G., Hulstijn, W. and Sabbe, B. (
Dom, G., D'Haene, P., Hulstijn, W. et al. (
Dougherty, D. M., Bjork, J. M., Huckabee, H. C. et al. (
Dougherty, D. M., Mathias, C. W., Marsh, D. M. et al. (
Feij, J. A., Dekker, P. H., Koopmans, J. R. et al. (
Fillmore, M. T. and Weafer, J. (
First, M. B., Gibbon, M., Spitzer, R. L. et al. (
Fishbein, D., Hyde, C., Eldreth, D. et al. (
Forman, S. D., Dougherty, G. G., Casey, B. J. et al. (
Haro, G., Mateu, C., Martinez-Raga, J. et al. (
Hester, R. and Garavan, H. (
Kalivas, P. W. and Volkow, N. D. (
Kaufman, J. N., Ross, T. J., Stein, E. A. et al. (
Kirby, K. N. and Petry, N. M. (
Kirby, K. N., Petry, M. N. and Bickel, W. K. (
Kirisci, L., Tarter, R. E., Vanyukov, M. et al. (
Kokkevi, A. and Hartgers, C. (
Kollins, S. H. (
Koob, G. F. and Le Moal, M. (
Lange, A., Hoogendoorn, M., Wiederspahn, A. et al. (
Lange, A., Pahlich, A., Sarucco, M. et al. (
Lee, T. M., Zhou, W. H., Luo, X. J. et al. (
Linehan, M. M. (
Linehan, M. M., Schmidt, H. III, Dimeff, L. A. et al. (
Lubman, D. I., Yücel, M. and Pantelis, C. (
Lusher, J., Chandler, C. and Ball, D. (
Lykouras, L., Moussas, G. and Botsis, A. (
Madden, G. J., Petry, N. M., Badger, G. J. et al. (
Madden, G. J., Bickel, W. K. and Jacobs, E. A. (
Mann, R. E., Sobell, L. C. and Sobell, M. B. (
Marissen, M. A. E., Franken, I. H. A., Waters, A. J. et al. (
Masse, L. C. and Tremblay, R. E. (
Mazur, J.E. (
Moeller, F. G., Dougherty, D. M., Barratt, E. S. et al. (
Moeller, F. G., Barratt, E. S., Fischer, C. J. et al. (
Moeller, F. G., Hasan, K. M., Steinberg, J. L. et al. (
Noël, X. Van der Linden, M., d'Acremont, M. et al. (
Ooteman, W., Verheul, R., Naassila, M. et al. (
Patton, J. H., Stanford, M. S. and Barratt, E. S. (
Petry, N. M. (
Petry, M. N. and Casarella, T. (
Raes, V. (
Reynolds, B., Richards, J. B., Horn, K. et al. (
Reynolds, B., Ortengren, A., Richards, J.B. et al. (
Richards, J. B., Zhang, L., Mitchell, S. H. et al. (
Ross, S., Dermatis, H., Levounis, P. et al. (
Salo, R., Nordahl, T. E., Possin, K. et al. (
Seguin, J. R. (
Skodol, A. E., Oldham, J. M. and Gallaher, P. E. (
Stroop, J. R. (
Swick, D. and Jovanovic, J. (
Tarter, R. E., Kirisci, L., Mezzich, A. et al. (
Tedstone, D. and Coyle, K. (
Thomas, V. H., Melchert, T. P. and Banken, J. A. (
van den Bosch, L. M. C., Verheul, R., Schippers, G. M. et al. (
Van Horn, D. H. and Frank, A. F. (
Verheul, R. (
Verheul, R., van den Brink, W. and Hartgers, C. (
Verheul, R., van den Brink, W. and Geerlings, P. (
Verheul, R., Kranzler, H. R., Poling, J. et al. (
Verheul, R., van den Bosch, L. C. M., Koeter, M. W. J. et al. (
Wagner, T., Krampe, H., Stawicki, S. et al. (
Weertman, A., Arntz, A. and Kerhofs, M. L. M. (
Whiteside, S. P. and Lynam, D. R. (
Winstanley, C. A., Dalley, J. W., Theobald, D. E. et al. (
Author notes
1Psychiatric Centre Alexian Brothers, Boechout, Belgium
2Collaborative Antwerp Psychiatric Research Institute (CAPRI), Antwerp, Belgium
3Academic Medical Centre (AMC) and Amsterdam Institute for Addiction Research (AIAR), Amsterdam, The Netherlands